Advanced Synthesis of S-Type Chiral Sulfoxide Intermediates for Ticagrelor Production
Introduction to Next-Generation Chiral Sulfoxide Synthesis
The pharmaceutical industry continuously demands higher standards of stereochemical purity, particularly for complex anticoagulants like Ticagrelor. A pivotal advancement in this domain is detailed in patent CN110698482A, which discloses a robust preparation method for S-type chiral sulfoxide compounds. This technology addresses a critical gap in the synthesis of Ticagrelor-related substances, specifically targeting the production of single-enantiomer sulfoxide impurities that were previously only accessible as racemic mixtures. By leveraging a sophisticated metal-organic catalytic system, this process ensures the generation of high-purity intermediates essential for rigorous pharmacological evaluation and quality control. For global procurement teams and R&D directors, mastering this synthetic route represents a significant leap forward in securing reliable supply chains for next-generation antiplatelet therapies.
The strategic importance of this patent lies in its ability to distinguish between S-type and R-type isomers, a capability that was notably absent in earlier methodologies such as those described in CN105237540A. The inability to separate these isomers previously hindered the understanding of their distinct biological activities and toxicological profiles. This new method not only resolves the chirality issue but does so under mild reaction conditions that are highly amenable to industrial scale-up. As a result, manufacturers can now produce reference standards and active intermediates with unprecedented precision, thereby supporting the development of safer and more effective cardiovascular medications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Ticagrelor sulfoxide impurities has been plagued by significant stereochemical challenges. Traditional approaches often resulted in racemic mixtures, where the biologically active S-type isomer was inextricably mixed with its R-type counterpart. Separating these enantiomers typically requires preparative chiral HPLC, a technique that is prohibitively expensive and difficult to scale for commercial manufacturing. Furthermore, the lack of pure standards made it nearly impossible to fully characterize the toxicity or efficacy of individual isomers, creating regulatory hurdles for drug approval processes. The thermal instability of the sulfoxide bond under certain conditions also posed risks of racemization during downstream processing, further compromising product integrity.
The Novel Approach
The methodology outlined in CN110698482A introduces a paradigm shift by integrating asymmetric catalysis directly into the synthesis workflow. Instead of relying on post-synthesis separation, this route employs a chiral vanadium catalyst system to induce stereoselectivity during the oxidation step itself. Crucially, the process optimizes the reaction sequence by performing the nucleophilic substitution prior to the oxidation. This specific ordering prevents the exposure of the sensitive chiral sulfoxide moiety to the elevated temperatures required for substitution, thereby locking in the desired stereochemistry. The result is a streamlined process that delivers the target S-type compound with exceptional optical purity and yield, eliminating the need for costly chiral resolution steps.
Mechanistic Insights into Vanadium-Catalyzed Asymmetric Oxidation
The core innovation of this synthesis lies in the precise formulation of the catalytic system used in step three. The reaction utilizes Vanadyl acetylacetonate (VO(acac)2) as the metal center, coordinated with a chiral ligand such as (R,R)-diethyl tartrate and a controlled amount of water. This ternary system creates a chiral environment around the vanadium atom, which directs the attack of the peroxide oxidant (such as tert-butyl hydroperoxide) onto the sulfide sulfur atom from a specific face. The presence of water is critical, as it modulates the coordination sphere of the vanadium, enhancing both the reaction rate and the enantioselectivity. Experimental data within the patent demonstrates that deviating from the optimal molar ratio of catalyst to ligand to water (specifically 1:1.25:2.5) leads to a marked decrease in both yield and optical purity.
Furthermore, the mechanism relies heavily on temperature control to preserve the chiral integrity of the product. The oxidation is conducted at cryogenic temperatures, specifically between 0°C and 4°C. This low-temperature regime suppresses non-selective background oxidation and prevents the thermal racemization that plagues sulfoxide chemistry. By maintaining these strict thermal parameters, the process ensures that the kinetic product—the desired S-isomer—is formed exclusively. This level of mechanistic control is vital for R&D teams aiming to replicate the process, as even minor fluctuations in temperature or reagent stoichiometry can lead to the formation of unwanted diastereomers that complicate downstream purification.
How to Synthesize S-Type Chiral Sulfoxide Efficiently
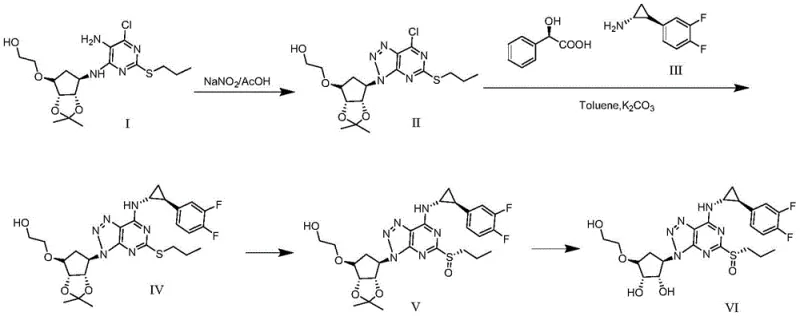
Implementing this synthesis route requires strict adherence to the sequential logic defined in the patent. The process begins with the formation of the triazole core, followed by the introduction of the chiral cyclopropylamine side chain. Once the sulfide precursor is established, the critical asymmetric oxidation is performed, followed by a final deprotection step. The following guide outlines the standardized operational parameters derived from the patent examples, ensuring reproducibility and high yield. For detailed laboratory protocols and safety data sheets regarding the specific reagents mentioned below, please refer to the technical documentation provided by your chemical supplier.
- Cyclization of Compound I using sodium nitrite in glacial acetic acid to form the triazole ring structure (Compound II).
- Nucleophilic substitution of Compound II with a chiral cyclopropylamine derivative in toluene to yield Compound IV.
- Enantioselective oxidation of the sulfide group in Compound IV using a vanadyl acetylacetonate catalyst system with a chiral tartrate ligand.
- Acidic hydrolysis of the protecting groups in Compound V using ethanol and hydrochloric acid to finalize the S-type chiral sulfoxide (Compound VI).
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers substantial strategic benefits beyond mere chemical elegance. The elimination of chiral chromatography steps translates directly into reduced manufacturing costs and shorter production cycles. By achieving high enantiomeric excess directly through catalysis, manufacturers can bypass the capital-intensive infrastructure typically required for preparative separation. This efficiency gain allows for more competitive pricing models and faster time-to-market for critical drug intermediates. Additionally, the use of commercially available catalysts and reagents ensures that the supply chain remains resilient against raw material shortages.
- Cost Reduction in Manufacturing: The primary driver for cost optimization in this process is the removal of chiral resolution steps. Traditional methods often lose up to half of the material during separation, whereas this catalytic approach maximizes atom economy by producing the desired isomer directly. Furthermore, the mild reaction conditions reduce energy consumption associated with heating and cooling, contributing to lower utility costs. The high purity of the crude product also minimizes the solvent and silica gel usage required for final purification, resulting in significant waste reduction and disposal cost savings.
- Enhanced Supply Chain Reliability: The reagents utilized in this protocol, such as vanadyl acetylacetonate and diethyl tartrate, are commodity chemicals with stable global supply chains. Unlike proprietary chiral auxiliaries that may be subject to single-source bottlenecks, these catalysts are readily available from multiple vendors. This diversity in sourcing mitigates the risk of production delays due to raw material scarcity. Moreover, the robustness of the reaction conditions means that the process is less susceptible to variations in reagent quality, ensuring consistent batch-to-batch performance.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial production is facilitated by the absence of extreme conditions. The reactions proceed at near-ambient pressures and moderate temperatures, reducing the engineering complexity of the reactor systems. From an environmental perspective, the high selectivity of the catalyst reduces the generation of hazardous byproducts. The ability to recycle solvents like toluene and ethyl acetate further aligns the process with green chemistry principles, helping manufacturers meet increasingly stringent environmental regulations without compromising output.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this synthesis method. These insights are derived directly from the experimental data and comparative examples provided in the patent literature. Understanding these nuances is essential for process chemists aiming to adapt this technology for large-scale manufacturing environments.
Q: Why is the reaction sequence critical in this synthesis?
A: Performing substitution before oxidation is crucial. If oxidation occurs first, the subsequent high-temperature substitution step can cause racemization of the chiral sulfoxide center, drastically reducing optical purity and yield.
Q: What catalyst system provides the highest enantioselectivity?
A: The patent data indicates that Vanadyl acetylacetonate (VO(acac)2) combined with (R,R)-diethyl tartrate and water in a specific molar ratio (1:1.25:2.5) yields the highest purity (>99%) and optimal conversion rates.
Q: How does this method improve upon prior art for Ticagrelor impurities?
A: Unlike previous methods that produced racemic mixtures requiring difficult separation, this novel approach directly synthesizes the specific S-type isomer with high stereochemical control, facilitating accurate biological activity studies and quality control.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable S-Type Chiral Sulfoxide Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-purity intermediates play in the development of life-saving medications. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with unwavering consistency. We specialize in the manufacture of complex pharmaceutical intermediates, adhering to stringent purity specifications and utilizing rigorous QC labs to verify every batch. Our commitment to quality assurance means that every gram of S-type chiral sulfoxide we deliver meets the exacting standards required for global regulatory submissions.
We invite you to collaborate with us to optimize your supply chain for Ticagrelor and related anticoagulant projects. Our experts are ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Please contact our technical procurement team today to request specific COA data and route feasibility assessments. By partnering with us, you gain access to a reliable source of advanced chiral building blocks that will accelerate your R&D timelines and secure your commercial manufacturing future.
