Advanced Biocatalytic Synthesis of Phenylephrine Intermediates for Commercial Scale-up
Advanced Biocatalytic Synthesis of Phenylephrine Intermediates for Commercial Scale-up
The pharmaceutical industry is constantly seeking more sustainable and efficient pathways for the production of critical active pharmaceutical ingredients (APIs) and their precursors. A significant breakthrough in this domain is detailed in patent CN105985990B, which outlines a novel production method for phenylephrine intermediates utilizing a highly optimized biocatalytic system. This technology represents a paradigm shift from traditional chemical synthesis, leveraging the specificity of ketoreductase (KRED) enzymes to achieve superior stereocontrol and environmental compatibility. The core innovation lies in the design of a self-circulating coenzyme system driven by isopropanol, coupled with the strategic addition of antioxidants to maintain reaction integrity. For global procurement and R&D teams, understanding this methodology is crucial for securing a reliable pharmaceutical intermediates supplier capable of delivering high-purity materials with a reduced environmental footprint.

The structural complexity of phenylephrine, specifically the chiral hydroxyl center, necessitates precise synthetic control to ensure therapeutic efficacy and safety. As illustrated in the chemical structures, the transformation involves the reduction of a ketone precursor (Formula III) to the corresponding chiral alcohol (Formula II), which serves as the benzyl-protected intermediate. This intermediate is subsequently debenzylated to yield the final active compound (Formula I). The patent emphasizes that while direct reduction without amino protection is theoretically economic, the benzyl-protected route remains the industrial mainstream due to its robustness. Therefore, optimizing the conversion of Formula III to Formula II is the pivotal step in the entire value chain, dictating the overall yield, purity, and cost-efficiency of the final API production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the preparation of chiral hydroxyl centers in phenylephrine intermediates has relied heavily on chemical hydrogenation methods. These conventional processes typically employ chiral catalysts, often involving heavy metals such as palladium, under rigorous conditions of high pressure and elevated temperature. While effective, these methods present substantial drawbacks for modern manufacturing, including significant safety hazards associated with high-pressure hydrogen gas and the environmental toxicity of heavy metal catalysts. Furthermore, the removal of trace palladium residues from the final product requires complex and costly purification steps to meet stringent regulatory limits for elemental impurities. Additionally, previous biocatalytic attempts, such as those utilizing glucose dehydrogenase for coenzyme regeneration, suffered from inefficiencies; they required the addition of equivalent amounts of alkali to neutralize generated gluconic acid and consumed excessive amounts of enzyme, reaching up to 30% of the substrate mass, which drastically inflated production costs.
The Novel Approach
In stark contrast, the novel approach disclosed in the patent utilizes a ketoreductase (KRED) catalyzed reduction that operates under mild, ambient conditions, effectively eliminating the need for high-pressure equipment and toxic metal catalysts. The genius of this method lies in its self-circulating coenzyme system, where isopropanol acts as the hydrogen donor, regenerating the reduced coenzyme (NADPH) from its oxidized form (NADP+) without the need for auxiliary enzymes. This simplification reduces the total enzyme consumption to merely 5% of the substrate mass, a dramatic improvement over prior biological methods. Moreover, the integration of specific antioxidants into the reaction matrix addresses a previously overlooked instability issue, preventing substrate oxidation and ensuring high conversion rates. This holistic optimization results in a greener, safer, and more economically viable process for the commercial scale-up of complex pharmaceutical intermediates.
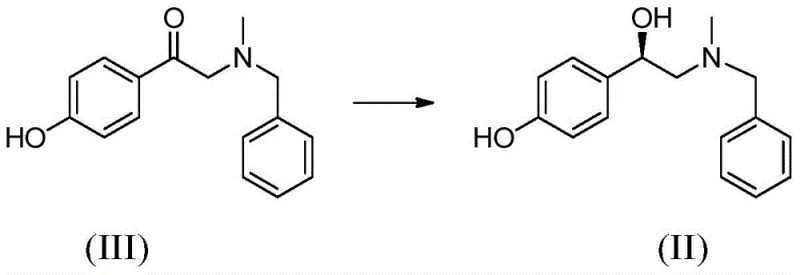
Mechanistic Insights into KRED-Catalyzed Asymmetric Reduction
The mechanistic foundation of this process rests on the stereoselective reduction of the prochiral ketone substrate (Formula III) by the ketoreductase enzyme. The enzyme facilitates the transfer of a hydride ion from the reduced coenzyme (NADPH) to the carbonyl carbon of the substrate, generating the desired (R)-configured chiral alcohol. Crucially, the patent highlights the discovery that the KRED enzyme possesses the intrinsic ability to oxidize isopropanol to acetone, thereby regenerating the NADPH required for the reduction cycle. This self-sustaining loop eliminates the stoichiometric requirement for expensive external reducing agents or secondary enzyme systems. The reaction is conducted in a buffered aqueous environment, preferably using a phosphate buffer at a pH of 7.0 ± 0.5, which maintains the optimal ionization state for enzyme activity and substrate solubility. The use of isopropanol not only drives the equilibrium towards product formation by Le Chatelier's principle but also serves as a cosolvent, enhancing the solubility of the organic substrate within the aqueous phase.
A critical, yet often underestimated, aspect of this mechanism is the role of the antioxidant. During the enzymatic reaction, the phenolic substrate is susceptible to oxidation by dissolved oxygen or reactive oxygen species present in the aqueous medium, leading to the formation of quinone-like byproducts. These oxidative side reactions not only consume the starting material but can also potentially inhibit the enzyme or complicate downstream purification. The patent data demonstrates that the addition of antioxidants such as mercaptoethanol, dithiothreitol (DTT), or vitamin C effectively scavenges these oxidative species. Experimental results indicate that without these additives, the reaction mixture turns black, signaling extensive degradation and low conversion. Conversely, with the antioxidant present, the solution remains faint yellow, correlating with conversion rates exceeding 99%. This mechanistic insight underscores the importance of managing the redox environment within the bioreactor to maximize yield and optical purity.
How to Synthesize Benzyl-protected Phenylephrine Efficiently
The implementation of this biocatalytic route requires precise control over reaction parameters to ensure reproducibility and high performance on an industrial scale. The process begins with the preparation of a reaction mixture containing the ketone substrate and isopropanol in a buffered solution, followed by the sequential addition of the biocatalytic components. Maintaining the correct pH and temperature throughout the reaction is vital for preserving enzyme stability and activity. The following guide outlines the standardized operational procedure derived from the patent examples, providing a clear roadmap for technical teams aiming to adopt this technology for large-scale manufacturing. Detailed standard operating procedures for this synthesis are provided in the section below.
- Mix the ketone substrate (Formula III) with isopropanol and phosphate buffer at pH 7.0, stirring at 30°C.
- Add the antioxidant (such as mercaptoethanol or DTT), ketoreductase KRED enzyme, and NADP coenzyme to the reaction mixture.
- Adjust pH to 5.0-6.0 during reaction, then filter and crystallize the product by adjusting pH to 9-10 and cooling.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this biocatalytic methodology offers profound strategic advantages that extend beyond mere technical feasibility. The elimination of heavy metal catalysts and high-pressure hydrogenation equipment fundamentally alters the cost structure and risk profile of the manufacturing process. By removing the need for expensive palladium catalysts and the associated rigorous purification steps required to meet residual metal specifications, the overall cost of goods sold (COGS) is significantly reduced. Furthermore, the mild reaction conditions enhance operational safety, reducing the insurance and compliance costs associated with handling hazardous chemicals and high-pressure vessels. This process optimization translates directly into a more competitive pricing structure for the final intermediate, allowing buyers to achieve substantial cost savings in API manufacturing without compromising on quality or regulatory compliance.
- Cost Reduction in Manufacturing: The primary driver for cost reduction in this process is the drastic decrease in biocatalyst loading. By utilizing a self-circulating coenzyme system powered by isopropanol, the method avoids the need for auxiliary enzymes like glucose dehydrogenase, which were required in previous generations of biocatalytic routes. This reduction in enzyme consumption, dropping from roughly 30% of substrate mass in older methods to just 5% in this new process, represents a massive decrease in raw material costs. Additionally, the simplified downstream processing, which avoids complex metal scavenging steps, further lowers operational expenditures. The use of commodity chemicals like isopropanol as both solvent and reagent ensures that raw material sourcing remains stable and inexpensive, insulating the supply chain from volatility in specialty chemical markets.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the robustness and simplicity of the reaction design. The reliance on commercially available, stable enzymes and common buffer systems reduces the risk of supply disruptions associated with exotic or proprietary catalysts. The mild operating conditions (ambient temperature and pressure) mean that the reaction can be performed in standard stainless steel or glass-lined reactors found in most multipurpose chemical plants, eliminating the need for specialized high-pressure infrastructure that often creates bottlenecks in production scheduling. This flexibility allows for faster turnaround times and easier scale-up from pilot to commercial production, ensuring that suppliers can meet fluctuating demand patterns with greater agility and reliability.
- Scalability and Environmental Compliance: From an environmental and regulatory standpoint, this biocatalytic route aligns perfectly with the industry's shift towards green chemistry. The absence of toxic heavy metals simplifies waste treatment and disposal, reducing the environmental burden and associated compliance costs. The aqueous nature of the reaction medium minimizes the use of volatile organic compounds (VOCs), contributing to a safer workplace and lower emissions. This environmental friendliness is not just a corporate social responsibility metric; it is a tangible commercial asset that facilitates smoother regulatory approvals and enhances the marketability of the final product to eco-conscious pharmaceutical partners. The process is inherently scalable, as demonstrated by successful amplification from milligram to multi-gram scales in the patent examples, proving its viability for industrial tonnage production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this KRED-catalyzed synthesis. These insights are derived directly from the experimental data and process descriptions within the patent, providing clarity on critical process parameters and troubleshooting strategies. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: Why is an antioxidant necessary in the KRED-catalyzed reduction of phenylephrine intermediates?
A: The addition of antioxidants like mercaptoethanol or dithiothreitol prevents the oxidation of the substrate and reaction components, specifically avoiding the formation of quinone byproducts which can stagnate the enzymatic reaction and lower conversion rates.
Q: How does this biocatalytic method improve upon traditional chemical hydrogenation?
A: Unlike traditional methods requiring high-pressure hydrogenation with toxic palladium catalysts, this enzymatic route operates at ambient temperature and pressure, eliminating heavy metal residues and simplifying downstream purification processes.
Q: What is the advantage of using isopropanol in this coenzyme circulation system?
A: Isopropanol serves as both the solvent and the hydrogen donor, enabling a self-circulating coenzyme system that eliminates the need for auxiliary enzymes like glucose dehydrogenase, thereby significantly reducing total enzyme consumption.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Phenylephrine Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the biocatalytic technologies described in patent CN105985990B for the production of high-value pharmaceutical intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent and reliable supply of critical materials. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including the precise control of chiral purity and residual solvent levels required for API intermediates. We are committed to leveraging advanced enzymatic processes to deliver products that meet the highest standards of quality and sustainability.
We invite global pharmaceutical partners to collaborate with us to optimize their supply chains through this innovative technology. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out today to obtain specific COA data and comprehensive route feasibility assessments, ensuring that your project moves forward with the most efficient and economically advantageous manufacturing strategy available in the market.
