Advanced Thienothiadiazole-Based Organic Photoelectric Materials for Next-Gen NIR Detectors
Advanced Thienothiadiazole-Based Organic Photoelectric Materials for Next-Gen NIR Detectors
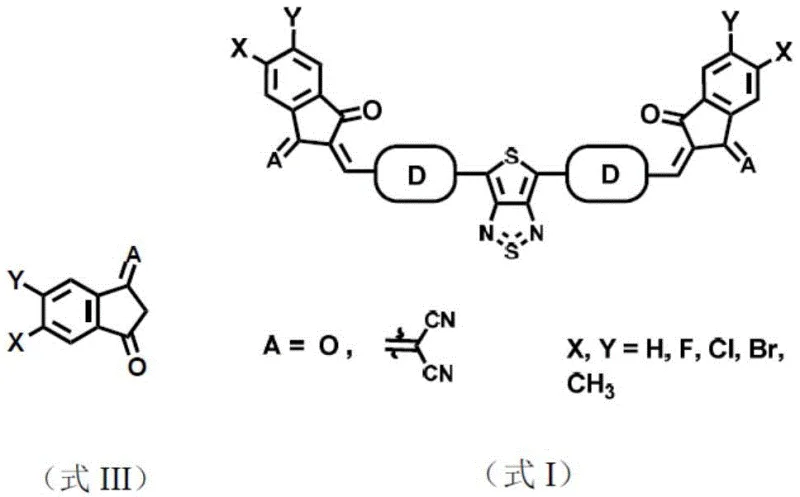
The rapid evolution of optoelectronic technology has intensified the demand for high-performance organic near-infrared (NIR) detectors that can surpass the limitations of traditional inorganic counterparts. Patent CN114057774A introduces a groundbreaking class of organic photoelectric molecular materials centered around a thienothiadiazole structure, specifically engineered to address the critical challenges of spectral range and charge transport efficiency. This innovation leverages a unique A-D-A (Acceptor-Donor-Acceptor) architecture where the thienothiadiazole unit serves as a powerful electron-deficient core, flanked by thiophene-based connecting units and capped with specialized end groups like 2-(2-methylene-3-oxo-2,3-dihydro-1H-inden-1-ylidene)malononitrile. The resulting molecular design ensures exceptional planarity and extended conjugation, which are paramount for delocalizing molecular orbitals and maximizing the extinction coefficient in the near-infrared region.
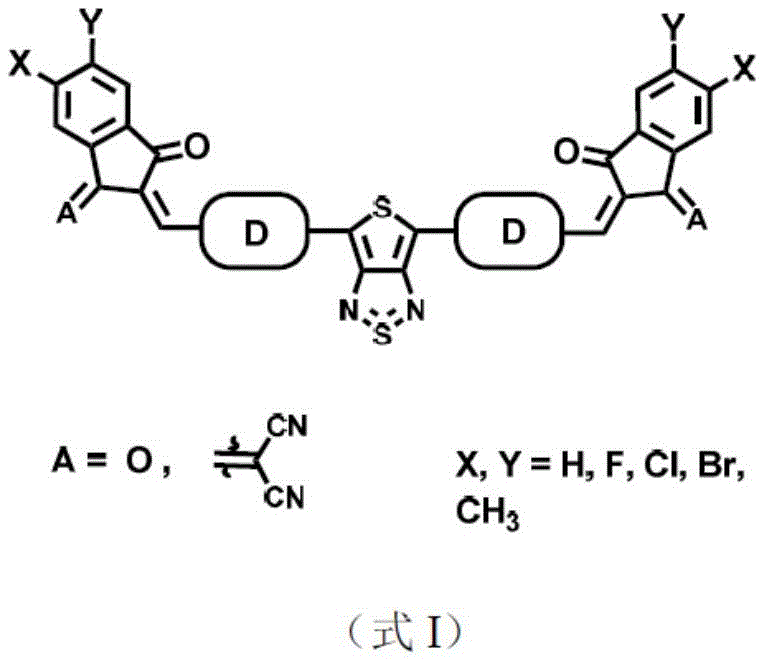
For R&D directors and procurement specialists seeking a reliable electronic chemical supplier, this patent represents a significant leap forward in material science. The disclosed compounds are not merely theoretical constructs but are synthesized through a robust, scalable pathway that yields materials with absorption spectra extending beyond 1.1 μm. This broad spectral coverage is essential for applications ranging from biomedical imaging and optical communication to night vision and environmental monitoring. Furthermore, the inherent solubility of these molecules in standard organic solvents allows for the fabrication of high-quality thin films using cost-effective solution processing techniques, thereby challenging the dominance of rigid, expensive inorganic detectors in flexible substrate applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the development of organic near-infrared light detectors has been hindered by the reliance on polymer donor materials that often suffer from batch-to-batch variability and limited control over molecular weight distribution. While polymer-based systems have attracted attention, their performance metrics, particularly regarding external quantum efficiency and specific detectivity in the NIR band, frequently fall short of the rigorous standards required for commercial deployment. Additionally, many existing small molecule receptor materials utilize conjugated or condensed ring thiophene units as cores, which restricts the structural diversity and limits the absorption spectrum to ranges below the critical 1.1 μm threshold. This spectral limitation severely constrains the application scenarios for organic detectors, preventing them from fully competing with inorganic technologies in fields requiring deep infrared sensitivity.
The Novel Approach
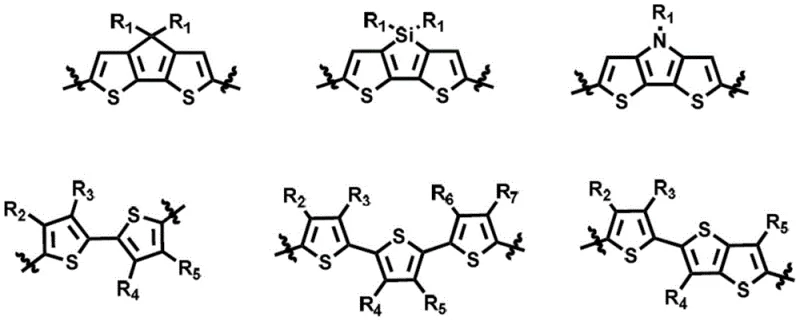
The novel approach detailed in the patent overcomes these hurdles by introducing a thienothiadiazole core, a strong receptor unit with a distinct quinoid structure effect that significantly reduces the molecular band gap. Unlike traditional cores, the double five-membered ring structure of thienothiadiazole offers superior planarity and minimal steric hindrance, which remarkably enhances the degree of conjugation and improves overall solubility. By coupling this central unit with electron-donating thiophene derivatives and capping the ends with strong acceptor groups, the new materials achieve a balanced energy level alignment that promotes efficient charge separation and transport. This strategic molecular engineering results in a material that retains high crystallinity while offering the processability needed for large-area manufacturing, effectively bridging the gap between laboratory performance and industrial viability.
Mechanistic Insights into Thienothiadiazole-Core Assembly and Functionalization
The synthesis of these advanced materials relies on a sophisticated sequence of organometallic and condensation reactions that ensure precise control over the molecular architecture. The process begins with the construction of the central backbone through a palladium-catalyzed cross-coupling reaction, likely a Stille coupling, between a halogenated thienothiadiazole derivative and a stannylated thiophene unit. This step is critical for establishing the conjugated bridge between the core and the donor units, setting the foundation for the material's electronic properties. Following the backbone assembly, the nitro groups on the thienothiadiazole ring are selectively reduced to amines using stannous chloride under acidic conditions, a transformation that prepares the molecule for the subsequent cyclization step which locks in the heterocyclic core structure.
Once the amine intermediates are generated, they undergo a cyclization reaction with N-sulfinylaniline in the presence of trimethylsilyl chloride to form the stable thienothiadiazole ring system. This is followed by a Vilsmeier-Haack formylation using phosphorus oxychloride and DMF, which installs the necessary aldehyde groups at the terminal positions of the donor units. The final and perhaps most crucial step involves a Knoevenagel condensation between the dialdehyde intermediate and the active methylene compounds, such as indene-dione derivatives. This condensation extends the conjugation length to the molecular termini, creating the final A-D-A structure responsible for the ultra-narrow band gap and the redshifted absorption characteristics that define the material's superior performance in NIR detection applications.
How to Synthesize Thienothiadiazole-Based Organic Photoelectric Material Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for producing these high-value intermediates with high purity and yield. The process is designed to be modular, allowing for the variation of substituents on the thiophene units and the terminal acceptor groups to fine-tune the optical and electronic properties for specific device requirements. From the initial coupling to the final purification via column chromatography and recrystallization, each step is optimized to minimize impurities that could act as charge traps in the final photodetector device. For detailed operational parameters and safety considerations, please refer to the standardized synthesis guide below.
- Perform Stille coupling between dibromo-thienothiadiazole derivative and stannyl-thiophene unit using Pd catalyst.
- Reduce nitro groups to amines using stannous chloride and hydrochloric acid.
- Cyclize diamine with N-sulfinylaniline to form the central thienothiadiazole core.
- Formylate the core using Vilsmeier-Haack conditions (POCl3/DMF).
- Final Knoevenagel condensation with indene-dione or malononitrile derivative to complete the A-D-A structure.
Commercial Advantages for Procurement and Supply Chain Teams
From a supply chain and procurement perspective, the adoption of this thienothiadiazole-based synthesis route offers substantial strategic benefits over legacy manufacturing methods. The use of commercially available starting materials and standard organometallic catalysts ensures that the raw material supply chain is robust and less susceptible to geopolitical disruptions often associated with exotic reagents. Furthermore, the solution-processable nature of the final product means that downstream device manufacturers can utilize established coating and printing technologies, drastically reducing the capital expenditure required for equipment upgrades. This compatibility with existing infrastructure accelerates the time-to-market for new NIR detector products, providing a competitive edge in the rapidly evolving optoelectronics sector.
- Cost Reduction in Manufacturing: The synthetic pathway eliminates the need for complex, multi-step protection and deprotection strategies often seen in similar conjugated systems, leading to a streamlined process with fewer unit operations. By utilizing efficient catalytic cycles and avoiding expensive transition metals in the final steps, the overall cost of goods sold is significantly optimized. The high solubility of the intermediates also facilitates easier purification through standard crystallization techniques, reducing solvent consumption and waste disposal costs associated with more laborious purification methods.
- Enhanced Supply Chain Reliability: The reliance on fundamental chemical building blocks such as thiophene derivatives and indene diones ensures a stable supply base, as these commodities are produced by multiple global vendors. The robustness of the reaction conditions, which tolerate a range of temperatures and solvent systems, adds a layer of resilience to the manufacturing process, minimizing the risk of batch failures due to minor fluctuations in reaction parameters. This reliability translates directly into consistent lead times and the ability to scale production volumes in response to market demand without compromising quality.
- Scalability and Environmental Compliance: The process is inherently scalable, moving seamlessly from gram-scale laboratory synthesis to kilogram and ton-scale commercial production without requiring fundamental changes to the reaction engineering. The use of common organic solvents like toluene and chloroform, which have well-established recovery and recycling protocols, supports sustainable manufacturing practices. Additionally, the high atom economy of the condensation steps minimizes the generation of hazardous byproducts, aligning with increasingly stringent environmental regulations and corporate sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these thienothiadiazole derivatives. Understanding these details is crucial for integrating this material into your supply chain and product development pipeline. The answers are derived directly from the technical disclosures and experimental data provided in the patent documentation.
Q: What is the absorption range of this thienothiadiazole material?
A: The material exhibits an absorption spectrum range exceeding 1.1 μm, making it highly suitable for near-infrared and wide-spectrum photodetectors.
Q: Is this material soluble for solution processing?
A: Yes, the material is designed to be soluble in common organic solvents such as chloroform, tetrahydrofuran, and toluene, facilitating high-quality thin film preparation via solution methods.
Q: What are the key structural features enhancing performance?
A: The molecule features a thienothiadiazole central unit with a quinoid structure effect, providing good planarity and strong electron deficiency, which reduces the band gap and enhances charge transport.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Thienothiadiazole Supplier
As the global demand for high-performance organic photoelectric materials continues to surge, partnering with an experienced CDMO like NINGBO INNO PHARMCHEM is essential for securing a stable supply of these critical intermediates. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the volumetric requirements of major display and sensor manufacturers. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of thienothiadiazole material meets the exacting standards required for high-efficiency NIR detectors.
We invite you to engage with our technical procurement team to discuss how we can support your specific project needs. Whether you require a Customized Cost-Saving Analysis for your current sourcing strategy or need specific COA data and route feasibility assessments for new product development, we are ready to provide the expertise and capacity you need. Contact us today to explore how our advanced synthesis capabilities can drive innovation and efficiency in your organic optoelectronic supply chain.
