Advanced Porphyrin-Mesitrazine Conjugates: Technical Breakthroughs and Commercial Scalability for Nonlinear Optics
Advanced Porphyrin-Mesitrazine Conjugates: Technical Breakthroughs and Commercial Scalability for Nonlinear Optics
The landscape of organic nonlinear optical (NLO) materials is undergoing a significant transformation driven by the need for high-performance components in optoelectronics and photonics. Patent CN113816974B introduces a groundbreaking class of porphyrin covalently linked mesitrazine compounds that address critical limitations in current material science. These novel structures leverage a sophisticated Donor (D)-Acceptor (A) architecture, where the electron-rich porphyrin macrocycle acts as the donor and the electron-deficient s-triazacone (mesitrazine) serves as the acceptor. This strategic molecular design facilitates efficient intramolecular charge transfer, resulting in superior nonlinear optical absorption coefficients compared to their monomeric counterparts. The invention provides a robust synthetic pathway starting from inexpensive industrial feedstocks like 2,3-dichloronitrobenzene, ensuring that the production of these high-value electronic chemicals remains economically viable. By extending the pi-conjugated system through covalent carbon-carbon single and triple bonds, the material exhibits enhanced photochemical and thermal stability, making it an ideal candidate for next-generation optical limiting and switching applications.
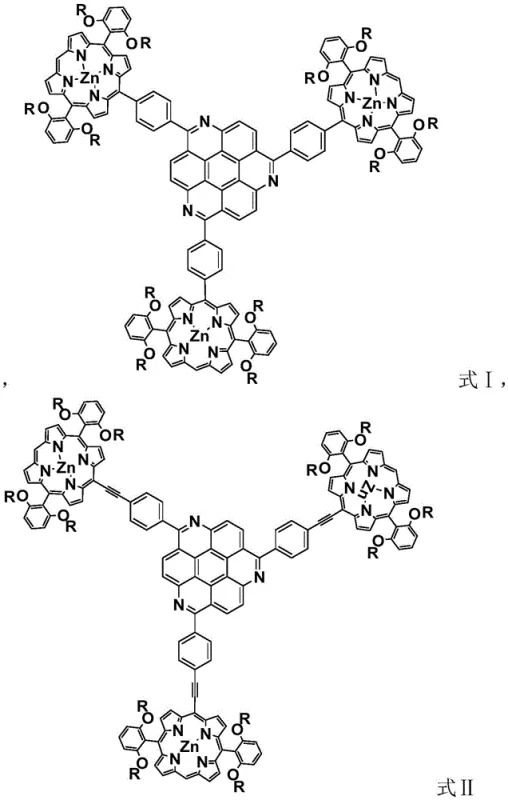
For procurement specialists and supply chain managers seeking a reliable nonlinear optical material supplier, understanding the structural integrity of these compounds is paramount. The molecules feature long alkyl chains (C6-C12) attached to the phenyl rings of the porphyrin unit, which drastically improves solubility in common organic solvents such as dichloromethane, chloroform, and DMF. This solubility enhancement is not merely a chemical curiosity; it is a critical processing parameter that allows for solution-based fabrication techniques like spin-coating or inkjet printing, which are essential for manufacturing large-area optical devices. The patent explicitly details two distinct structural variants, Formula I and Formula II, differentiated by the nature of the conjugated bridge connecting the donor and acceptor units. Formula II incorporates a carbon-carbon triple bond linker, which further extends the conjugation length and optimizes the flow of pi electrons, thereby yielding even higher nonlinear optical responses. This level of molecular tunability offers R&D directors the flexibility to tailor material properties for specific wavelength requirements without compromising on the fundamental stability of the core scaffold.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the development of polycyclic aromatic hydrocarbons (PAHs) for nonlinear optical applications has been hindered by the difficulty of balancing planarity with solubility. While planar structures are essential for effective pi-stacking and charge transport, they often lead to poor solubility and aggregation, which quenches fluorescence and reduces optical performance. Conventional doping strategies, where heteroatoms are simply introduced into the aromatic lattice, often fail to create a strong enough internal electric field to drive significant charge transfer. Furthermore, many existing synthetic routes for complex PAHs rely on harsh conditions that degrade the delicate porphyrin macrocycles or require expensive transition metal catalysts that are difficult to remove to ppm levels required for electronic grade materials. The lack of a defined Donor-Acceptor interface in simple PAHs limits their nonlinear absorption coefficients, necessitating higher concentrations or thicker films to achieve the desired optical limiting effect, which is impractical for miniaturized photonic circuits. Additionally, the thermal instability of many organic NLO materials restricts their operational lifetime in high-power laser environments, creating a bottleneck for commercial adoption in defense and telecommunications sectors.
The Novel Approach
The methodology outlined in CN113816974B overcomes these hurdles by covalently integrating the porphyrin donor directly onto the mesitrazine acceptor core. This approach creates a permanent, rigid D-A system that maximizes the dipole moment change upon excitation, a key factor in determining nonlinear optical efficiency. The synthesis utilizes a modular strategy where the mesitrazine core is constructed first via a copper-catalyzed coupling and subsequent solid-phase cyclization, ensuring a robust electron-deficient foundation. The porphyrin unit is separately synthesized with solubilizing hexyloxy chains, addressing the aggregation issue at the molecular level before the final coupling step. By employing palladium-catalyzed cross-coupling reactions (Suzuki and Sonogashira types) for the final assembly, the process ensures high regioselectivity and yield, minimizing the formation of regioisomers that could act as impurities. This covalent linkage strategy not only enhances the nonlinear optical response through synergistic effects but also locks the molecular conformation, preventing rotational relaxation that could dissipate energy non-radiatively. The result is a material that combines the high absorbance of porphyrins with the electron-accepting capability of nitrogen-doped PAHs, offering a comprehensive solution to the performance limitations of previous generations of organic optical materials.
Mechanistic Insights into Copper and Palladium Catalyzed Assembly
The synthesis of the mesitrazine core begins with a copper-catalyzed coupling of 2,3-dichloronitrobenzene in DMF at elevated temperatures (140-170°C). This Ullmann-type coupling forms the initial biaryl linkage, which is subsequently reduced to the amine using hydrazine hydrate and Pd/C. The critical step in forming the heterocyclic core is the solid-phase cyclization involving aluminum chloride and sodium chloride at 220°C. This high-temperature Friedel-Crafts-like cyclization closes the rings to form the rigid triazacone structure, a process that is thermodynamically driven and highly efficient for creating fused aromatic systems. The use of solid-phase reagents minimizes solvent waste and simplifies the workup, as the inorganic salts can be easily washed away with aqueous base. Following the core formation, the introduction of boronic ester groups via palladium catalysis (using dppf-PdCl2) activates the core for cross-coupling. This borylation step is crucial for the subsequent Suzuki coupling with the brominated porphyrin, allowing for the formation of the carbon-carbon single bond in Formula I. For Formula II, a Sonogashira coupling protocol is employed using cuprous iodide and palladium catalysts to install the alkyne linker, demonstrating the versatility of the synthetic platform.
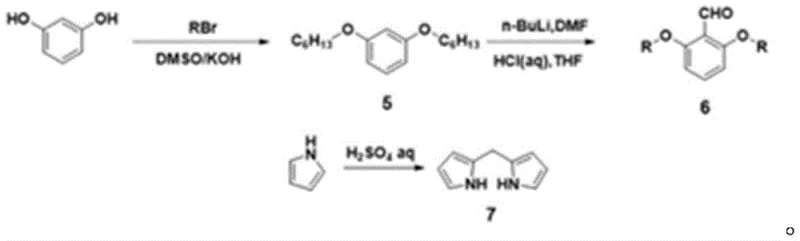
Impurity control is meticulously managed throughout the synthetic sequence, particularly during the porphyrin functionalization steps. The bromination of the porphyrin precursor using N-bromosuccinimide (NBS) is conducted at 0°C to prevent poly-bromination, ensuring that only the mono-bromo species reacts in the final coupling stage. This selectivity is vital because unreacted dibromo species or homocoupled porphyrin dimers would act as fluorescent impurities, degrading the optical clarity of the final film. The patent specifies rigorous purification protocols, including column chromatography with specific eluent systems (e.g., dichloromethane/petroleum ether/THF), to isolate the target compounds with high purity. The metallation of the porphyrin with zinc acetate is performed under mild conditions to preserve the integrity of the macrocycle while introducing the metal center, which influences the electronic properties of the donor unit. The final purification steps involve recrystallization and silica gel chromatography, which effectively remove residual palladium and copper catalysts, meeting the stringent metal content specifications required for electronic applications. This attention to detail in the mechanistic execution ensures that the final product possesses the consistent batch-to-batch quality necessary for industrial scale-up.
How to Synthesize Porphyrin Covalently Linked Mesitrazine Efficiently
The efficient production of these advanced materials relies on a sequential workflow that prioritizes the stability of intermediates and the optimization of coupling yields. The process begins with the preparation of the mesitrazine core, followed by the independent synthesis of the functionalized porphyrin donor, and concludes with the convergent coupling of these two fragments. Detailed standard operating procedures for each reaction step, including precise molar ratios and temperature controls, are essential for reproducibility. For instance, maintaining the coupling reaction temperature at 90°C for 12 hours ensures complete conversion of the starting materials while minimizing side reactions. The use of inert atmosphere techniques (nitrogen protection) throughout the synthesis prevents oxidation of sensitive intermediates like the boronic esters and phosphine ligands. Operators must adhere to strict quenching and extraction protocols to ensure safe handling of reagents like aluminum chloride and hydrazine hydrate.
- Synthesize the mesitrazine core intermediate via copper-catalyzed coupling of 2,3-dichloronitrobenzene, followed by reduction and solid-phase cyclization at 220°C.
- Prepare the porphyrin donor unit by condensing 2,6-dihexyloxybenzaldehyde with dipyrromethane, followed by bromination and zinc metallation.
- Couple the mesitrazine acceptor and porphyrin donor using Suzuki or Sonogashira cross-coupling reactions catalyzed by palladium complexes to form the final conjugated system.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the synthetic route described in this patent offers substantial advantages in terms of cost reduction in electronic chemical manufacturing and supply chain reliability. The reliance on commodity chemicals such as 2,3-dichloronitrobenzene and standard porphyrin building blocks means that the raw material supply is not subject to the volatility associated with exotic or proprietary precursors. This accessibility translates directly into lower input costs and reduced risk of supply disruption, which is a critical consideration for procurement managers planning long-term production schedules. Furthermore, the elimination of complex transition metal catalysts in the core formation step (using copper powder instead of expensive palladium for the initial coupling) significantly lowers the catalyst cost burden. Although palladium is used in the final coupling steps, the loadings are optimized, and the catalysts can potentially be recovered or scavenged, further enhancing the economic viability of the process. The high yields reported in the examples (e.g., 80% for the core cyclization, 61-95% for porphyrin steps) indicate a material-efficient process that minimizes waste generation and maximizes output per batch.
- Cost Reduction in Manufacturing: The process utilizes a solid-phase cyclization method that avoids the use of large volumes of organic solvents during the core formation, leading to significant savings in solvent purchase and disposal costs. By replacing solution-phase cyclization with a melt reaction using inorganic salts, the energy consumption for solvent recovery is drastically reduced. Additionally, the use of readily available copper powder for the initial coupling step instead of homogeneous palladium catalysts reduces the expense associated with precious metal procurement. The high atom economy of the cross-coupling reactions ensures that the majority of the mass of the starting materials ends up in the final product, reducing the cost of goods sold (COGS). These factors combined create a lean manufacturing profile that allows for competitive pricing in the high-value nonlinear optical material market without sacrificing quality.
- Enhanced Supply Chain Reliability: The starting materials identified in the patent, such as 2,3-dichloronitrobenzene and resorcinol derivatives, are produced on a multi-ton scale by major chemical suppliers globally, ensuring a stable and continuous supply chain. This contrasts with custom-synthesized intermediates that may have long lead times or single-source dependencies. The synthetic route is robust and tolerant to minor variations in reaction conditions, which reduces the risk of batch failures and production delays. The ability to synthesize the porphyrin and mesitrazine components independently allows for parallel processing, shortening the overall production lead time for high-purity organic intermediates. This modularity also enables inventory management strategies where key intermediates can be stocked, allowing for rapid response to fluctuating market demand for the final conjugated products.
- Scalability and Environmental Compliance: The reaction conditions described, such as reflux temperatures in common solvents like DMF and THF, are easily transferable from laboratory glassware to industrial stainless steel reactors. The absence of cryogenic conditions or ultra-high pressure requirements simplifies the engineering controls needed for scale-up, facilitating the commercial scale-up of complex organic intermediates. The workup procedures primarily involve filtration and crystallization, which are unit operations that scale linearly and are well-understood in process engineering. From an environmental standpoint, the reduction in solvent usage during the solid-phase step and the potential for solvent recycling in the coupling steps align with green chemistry principles. This compliance with environmental regulations reduces the regulatory burden and permitting time for new manufacturing facilities, accelerating time-to-market for these advanced optical materials.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these porphyrin-mesitrazine conjugates. The answers are derived directly from the experimental data and technical specifications provided in the patent documentation. Understanding these details is crucial for stakeholders evaluating the feasibility of integrating these materials into their product lines.
Q: What is the primary advantage of covalently linking porphyrin to mesitrazine?
A: The covalent linkage creates an effective Donor (D)-Acceptor (A) system that promotes intramolecular charge transfer, significantly enhancing the nonlinear optical absorption coefficient compared to monomeric compounds.
Q: How does the solid-phase cyclization step impact product purity?
A: The solid-phase reaction at 220°C using aluminum chloride facilitates the formation of the rigid triazacone core without requiring complex solvent systems, simplifying downstream purification and improving thermal stability.
Q: Are the raw materials for this synthesis commercially available?
A: Yes, the process utilizes cheap and easily obtained starting materials such as 2,3-dichloronitrobenzene and standard porphyrin precursors, ensuring a reliable supply chain for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Porphyrin Covalently Linked Mesitrazine Supplier
NINGBO INNO PHARMCHEM stands at the forefront of custom synthesis and manufacturing for advanced electronic materials, possessing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our technical team is uniquely qualified to adapt the synthetic routes described in CN113816974B to meet your specific volume and purity requirements. We understand that the successful deployment of nonlinear optical materials depends on stringent purity specifications and rigorous QC labs to ensure consistent performance in optical devices. Our state-of-the-art facilities are equipped to handle the specialized reaction conditions, such as high-temperature solid-phase cyclizations and air-sensitive palladium couplings, with the highest standards of safety and quality control. We are committed to delivering high-purity porphyrin derivatives that meet the exacting standards of the optoelectronics industry.
We invite you to contact our technical procurement team to discuss your specific project needs and request a Customized Cost-Saving Analysis tailored to your production volumes. Whether you require specific COA data for qualification purposes or detailed route feasibility assessments for process optimization, our experts are ready to provide the support you need. Partnering with us ensures not only access to cutting-edge chemical technology but also a supply chain partner dedicated to your long-term success in the competitive field of organic nonlinear optics. Reach out today to secure your supply of these next-generation optical materials.
