Advanced Star-shaped D-A Conjugated Molecules for High-Efficiency Organic Photovoltaic Manufacturing
The rapid evolution of organic photovoltaics demands materials that balance high energy conversion efficiency with manufacturing feasibility. Patent CN109956955B introduces a groundbreaking class of star-shaped D-A structure conjugated molecules based on benzotris(cyclopentadienobis five-membered aromatic heterocycles). These molecules are engineered with a C3 symmetric π-conjugated rigid plane core, directly connected to strong electron-withdrawing end groups, creating a novel architecture for organic solar cells. Unlike traditional polymer donors or fullerene acceptors, this star-shaped design offers precise molecular weight control and superior batch reproducibility, addressing critical pain points in the electronic chemical supply chain. The patent details a robust synthesis pathway that enables the production of materials with strong absorption, high mobility, and suitable electronic energy levels, positioning them as ideal candidates for next-generation electron donor or acceptor layers. 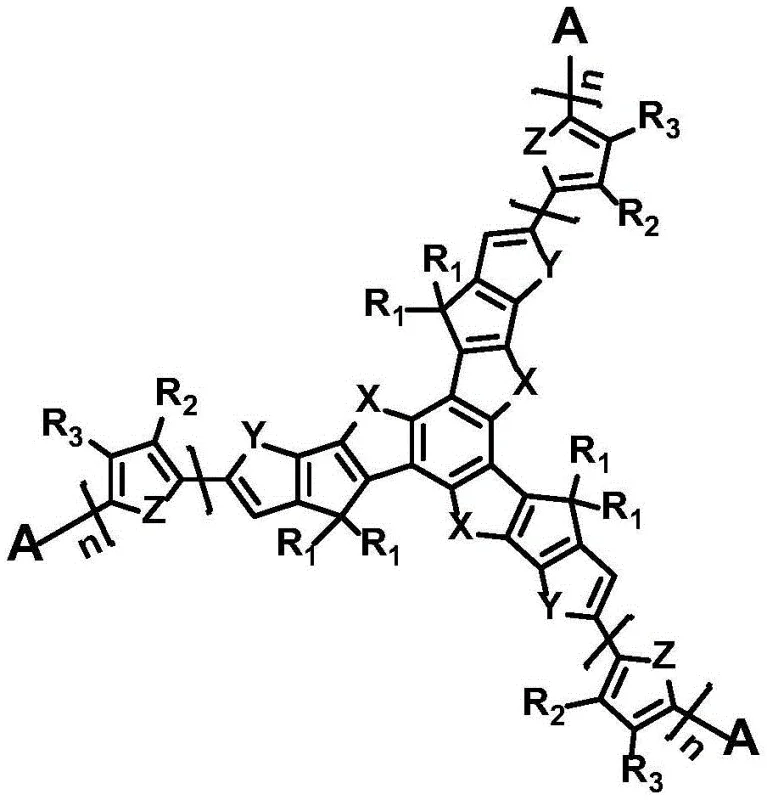
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional organic solar cell materials have long relied on conjugated polymers or fullerene derivatives, each presenting significant drawbacks for large-scale commercialization. Conjugated polymers, while offering good film-forming properties, suffer from inherent polydispersity in molecular weight distribution, making purification difficult and leading to poor batch-to-batch reproducibility. This variability complicates the quality control process for procurement teams seeking consistent performance in photovoltaic modules. On the other hand, fullerene derivatives, though possessing high electron mobility, exhibit poor absorption in the visible region and limited chemical adjustability. Their high cost and purification difficulties further hinder the cost reduction in electronic chemical manufacturing required for widespread solar energy adoption. These limitations create a bottleneck for R&D directors aiming to push energy conversion efficiencies beyond current thresholds while maintaining supply chain stability.
The Novel Approach
The novel approach described in the patent leverages a star-shaped small-molecule architecture to overcome these historical constraints. By utilizing a benzo-tris(cyclopenta-bi-five-membered aromatic heterocycle) core, the material achieves a larger π-conjugated plane compared to linear analogues, facilitating stronger pi-pi accumulation interactions. This structural rigidity translates to higher carrier mobility and more effective charge transmission within the active layer. Furthermore, the small-molecule nature ensures a definite structure and molecular weight, eliminating the polydispersity issues associated with polymers. The ability to tune the peripheral electron-withdrawing groups allows for precise adjustment of electronic energy levels, optimizing the match with donor materials for maximum photon capture. This design not only enhances device performance but also simplifies the purification process, offering a clear pathway for cost reduction in organic solar cell manufacturing through improved yield and consistency.
Mechanistic Insights into Palladium-Catalyzed Star-Shaped Core Synthesis
The synthesis of the central core involves a sophisticated palladium-catalyzed coupling reaction that establishes the C3 symmetric foundation of the molecule. Specifically, 2,5,8-tri(trimethylstannyl) benzotrithiophene reacts with 2-bromo-3-formate five-membered aromatic heterocyclic compounds in a toluene solvent at temperatures between 100°C and 120°C. This step is critical for forming the rigid planar structure that defines the material's electronic properties. The use of palladium catalysts such as tetrakis(triphenylphosphine)palladium ensures high selectivity and conversion, minimizing the formation of structural defects that could act as charge traps. Following the coupling, an addition reaction with active metal reagents and subsequent ring closure solidifies the core structure. This multi-step process is designed to maximize the conjugation length while maintaining solubility, a key factor for solution-processed device fabrication. 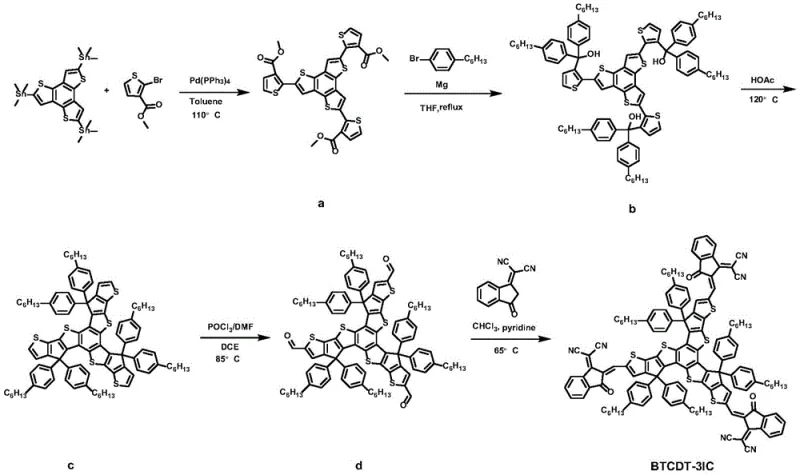
Impurity control is paramount in the production of high-purity organic semiconductors, and the patent outlines specific purification protocols to achieve this. After the initial coupling and ring closure, the intermediates undergo rigorous extraction, washing, and column chromatography purification. The use of specific eluent systems, such as petroleum ether mixed with ethyl acetate or dichloromethane, allows for the precise separation of the target star-shaped core from unreacted starting materials and side products. This attention to detail in the synthesis route ensures that the final conjugated molecules meet stringent purity specifications required for commercial scale-up of complex organic semiconductors. By eliminating transition metal residues and organic impurities early in the process, the method reduces the burden on downstream processing, thereby enhancing overall supply chain reliability and reducing lead time for high-purity organic photovoltaic materials.
How to Synthesize BTCDT-3IC Efficiently
The synthesis of the specific embodiment BTCDT-3IC demonstrates the practical application of the general method, involving a sequence of coupling, formylation, and condensation reactions. The process begins with the construction of the central core, followed by Vilsmeier-Haack formylation to introduce aldehyde groups necessary for the final extension. The detailed standardized synthesis steps see the guide below, which outlines the precise molar ratios, temperatures, and reaction times required to achieve optimal yields. This structured approach allows manufacturing teams to replicate the results consistently, ensuring that the commercial advantages of the material are fully realized in production environments.
- Perform palladium-catalyzed coupling of benzotrithiophene derivatives with brominated heterocycles in toluene at 100-120°C to form the central core.
- Execute aldehyde functionalization on the central core using Vilsmeier-Haack conditions to prepare the trialdehyde intermediate.
- Conduct Knoevenagel condensation with electron-withdrawing end groups in chloroform with pyridine catalyst to finalize the star-shaped structure.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this star-shaped D-A architecture offers substantial strategic benefits beyond mere performance metrics. The synthesis route relies on commercially available reagents and standard organic synthesis techniques, which significantly simplifies the sourcing of raw materials. This accessibility reduces the risk of supply disruptions often associated with specialized or proprietary catalysts. Furthermore, the small-molecule nature of the product eliminates the need for complex polymerization control, streamlining the manufacturing process and reducing operational costs. The high purity achievable through the described purification methods means less waste and higher effective yield, contributing to significant cost savings in the long run. These factors combine to create a robust supply chain framework that supports the growing demand for organic photovoltaic materials.
- Cost Reduction in Manufacturing: The elimination of complex polymerization steps and the use of standard palladium-catalyzed coupling reactions drastically simplify the production workflow. By avoiding the need for specialized polymerization equipment and stringent molecular weight control measures, manufacturers can leverage existing infrastructure, leading to substantial cost savings. Additionally, the high yield and purity reduce the volume of waste solvents and reagents required for purification, further lowering the environmental and financial burden of production. This efficiency translates directly into a more competitive pricing structure for the final electronic chemical products.
- Enhanced Supply Chain Reliability: The reliance on widely available starting materials such as benzotrithiophene derivatives and common heterocyclic compounds ensures a stable supply base. Unlike materials requiring rare or custom-synthesized precursors, this route minimizes the risk of bottlenecks caused by raw material shortages. The robustness of the reaction conditions, which tolerate standard industrial solvents like toluene and chloroform, also means that production can be easily scaled across different facilities without significant requalification. This flexibility enhances the overall reliability of the supply chain, ensuring consistent delivery of high-purity organic photovoltaic materials to global partners.
- Scalability and Environmental Compliance: The synthesis process is inherently scalable, moving seamlessly from gram-scale laboratory synthesis to multi-kilogram commercial production. The use of standard workup procedures like extraction and column chromatography can be adapted to industrial-scale purification techniques such as crystallization or preparative HPLC. Moreover, the high efficiency of the reaction reduces the generation of hazardous by-products, aligning with increasingly strict environmental regulations. This compliance not only mitigates regulatory risk but also appeals to end-users seeking sustainable and eco-friendly electronic materials for their green energy applications.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this star-shaped D-A technology. These answers are derived directly from the patent's technical disclosures and beneficial effects, providing clarity on performance, synthesis, and application potential. Understanding these details is crucial for stakeholders evaluating the integration of these materials into their existing product lines or research pipelines.
Q: What are the advantages of star-shaped D-A molecules over linear polymers in organic solar cells?
A: Star-shaped D-A molecules offer definite molecular weights, high purity, and excellent batch-to-batch reproducibility compared to polymers, which often suffer from polydispersity and purification challenges.
Q: How does the C3 symmetric core improve charge transport performance?
A: The C3 symmetric benzo-tris(cyclopenta-bi-five-membered aromatic heterocycle) core provides a rigid planar structure that enhances pi-pi stacking interactions, leading to higher carrier mobility and effective charge transmission.
Q: Is this synthesis route scalable for commercial production of photovoltaic materials?
A: Yes, the synthesis utilizes standard palladium-catalyzed coupling and condensation reactions with commercially available reagents, facilitating scalable manufacturing and supply chain reliability for electronic chemical applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Star-shaped D-A Conjugated Molecules Supplier
NINGBO INNO PHARMCHEM stands at the forefront of translating advanced patent technologies like CN109956955B into commercial reality. As a specialized CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory curiosity to industrial staple is seamless. Our commitment to stringent purity specifications and rigorous QC labs guarantees that every batch of star-shaped D-A conjugated molecules meets the exacting standards required for high-efficiency organic solar cells. We understand the critical nature of material consistency in photovoltaic applications and have optimized our processes to deliver the reliability your projects demand.
We invite you to collaborate with us to unlock the full potential of this novel material class. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and integration needs. By partnering with us, you gain access to specific COA data and route feasibility assessments that will accelerate your development timeline. Contact us today to discuss how our expertise in organic electronic materials can support your supply chain and drive innovation in your renewable energy solutions.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
