Advanced Dithieno Pyrrole Hole Transport Materials for High-Efficiency Perovskite Solar Cell Manufacturing
Advanced Dithieno Pyrrole Hole Transport Materials for High-Efficiency Perovskite Solar Cell Manufacturing
The rapid evolution of third-generation photovoltaics has placed immense pressure on the supply chain to deliver hole transport materials that balance high efficiency with commercial viability. Patent CN113336772A introduces a groundbreaking class of organic small molecule hole transport materials utilizing a dithieno[3,2-b:2',3'-d]pyrrole core coupled with carbazole end groups. This innovation addresses the critical bottlenecks of thermal instability and exorbitant synthesis costs associated with industry-standard materials like Spiro-OMeTAD. By leveraging a robust Suzuki-Miyaura coupling strategy, this technology enables the production of materials with tunable HOMO energy levels and exceptional glass transition temperatures, positioning it as a premier solution for reliable electronic chemical supplier networks aiming to support the mass commercialization of perovskite solar cells.
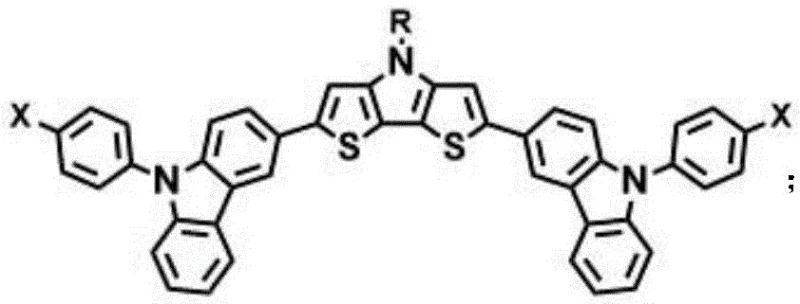
The structural versatility of these compounds allows for precise modulation of optoelectronic properties through the variation of alkyl chains (R) and terminal substituents (X). As illustrated in the general formula, the central dithienopyrrole unit provides a rigid, planar conjugated system that facilitates efficient hole mobility, while the peripheral carbazole units enhance film-forming capabilities and oxidative stability. This molecular architecture is specifically designed to overcome the morphological degradation often observed in amorphous small molecule films under prolonged thermal stress, making it an ideal candidate for high-purity OLED material and photovoltaic applications where long-term device integrity is paramount.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
For over a decade, the perovskite solar cell industry has been heavily reliant on Spiro-OMeTAD as the benchmark hole transport layer, despite its well-documented drawbacks in cost reduction in display & optoelectronic materials manufacturing. The synthesis of Spiro-OMeTAD involves a cumbersome multi-step spiral coupling process that requires expensive starting materials and rigorous purification protocols, leading to a supply chain that is both fragile and cost-prohibitive for gigawatt-scale deployment. Furthermore, the intrinsic thermal properties of Spiro-OMeTAD often necessitate the use of hygroscopic dopants to achieve acceptable conductivity, which inadvertently accelerates device degradation and limits the operational lifetime of the solar modules in real-world environmental conditions.
The Novel Approach
In stark contrast, the methodology disclosed in the patent employs a convergent synthetic route that dramatically simplifies the construction of the hole transport scaffold. By utilizing a pre-functionalized dithienopyrrole core and coupling it with readily accessible carbazole derivatives, the process eliminates the need for complex spiral bond formation. This approach not only streamlines the reaction sequence but also enhances the overall atom economy, allowing for cost reduction in electronic chemical manufacturing through reduced solvent usage and fewer purification cycles. The resulting materials, such as the TM series (TM-I to TM-VI), demonstrate photoelectric conversion efficiencies exceeding 18%, rivaling the performance of Spiro-OMeTAD while offering superior thermal resilience and easier processability.
Mechanistic Insights into Palladium-Catalyzed Cross-Coupling
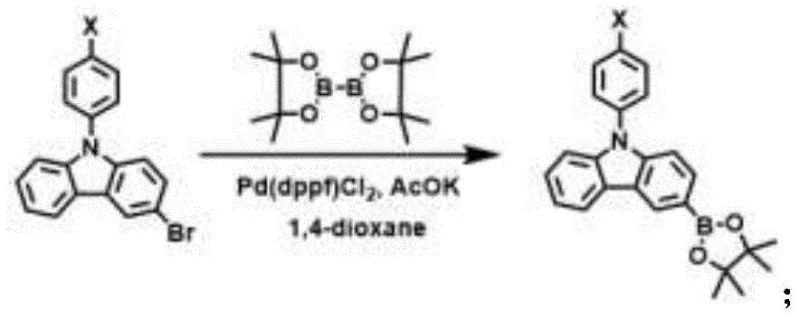
The synthesis of these advanced hole transport materials relies on a sophisticated two-stage palladium-catalyzed protocol that ensures high regioselectivity and yield. The first stage involves the borylation of 3-bromo-9-phenyl-9H-carbazole derivatives, where the bromine atom is replaced by a pinacol boronate ester group. This transformation is critical as it activates the carbazole unit for subsequent cross-coupling, utilizing Pd(dppf)Cl2 as the catalyst and potassium acetate as the base in anhydrous 1,4-dioxane. The reaction proceeds under nitrogen protection at temperatures between 90°C and 110°C, ensuring complete conversion while minimizing side reactions that could compromise the purity of the intermediate.
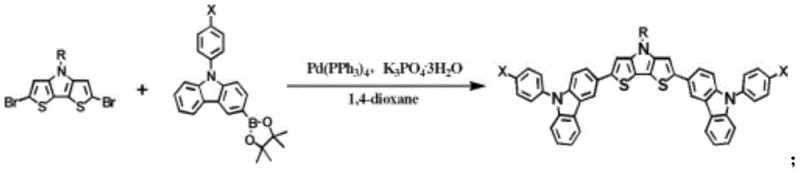
Following the preparation of the boronate intermediates, the second stage executes the key carbon-carbon bond formation via Suzuki-Miyaura coupling. In this step, the boronate ester reacts with 2,6-dibromo-4-alkyl-dithieno[3,2-b:2',3'-d]pyrrole in the presence of Pd(PPh3)4 and potassium phosphate tribasic. This catalytic cycle effectively links the electron-rich carbazole donors to the electron-deficient dithienopyrrole acceptor, creating the push-pull electronic structure necessary for efficient hole extraction. The use of mild bases and standard phosphine ligands allows this reaction to proceed smoothly at 100°C to 120°C, facilitating the commercial scale-up of complex polymer additives and small molecule semiconductors without requiring exotic reagents or extreme conditions.
How to Synthesize Dithieno Pyrrole Hole Transport Materials Efficiently
The practical implementation of this synthesis requires strict adherence to anhydrous conditions and precise stoichiometric control to maximize yield and minimize palladium residue. The process begins with the activation of the carbazole precursor, followed by the coupling reaction which forms the final conjugated backbone. Detailed standard operating procedures regarding reagent grades, solvent drying methods, and workup protocols are essential for reproducing the high purity levels reported in the patent examples. For a comprehensive guide on executing these steps with industrial-grade reproducibility, please refer to the standardized synthesis protocol below.
- Perform palladium-catalyzed borylation of 3-bromo-9-phenyl-9H-carbazole derivatives using bis(pinacolato)diboron and Pd(dppf)Cl2 in 1,4-dioxane at 90-110°C.
- Conduct Suzuki-Miyaura cross-coupling between the resulting boronate ester intermediates and 2,6-dibromo-4-alkyl-dithieno[3,2-b: 2',3'-d]pyrrole using Pd(PPh3)4 catalyst.
- Purify the final crude product via column chromatography using petroleum ether and dichloromethane mixtures to obtain high-purity yellow solid hole transport materials.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement perspective, the adoption of this dithienopyrrole-based technology offers substantial benefits in terms of supply chain security and total cost of ownership. Unlike the proprietary and tightly controlled supply chains associated with spiral fluorene derivatives, the starting materials for this synthesis—such as carbazole and thiophene derivatives—are commodity chemicals available from multiple global vendors. This diversification of the raw material base significantly mitigates the risk of supply disruptions and provides procurement managers with greater leverage in negotiating favorable pricing terms, ultimately driving down the bill of materials for perovskite module assembly.
- Cost Reduction in Manufacturing: The streamlined two-step synthetic route eliminates the need for expensive chiral auxiliaries or complex spiral coupling reagents, which are major cost drivers in traditional hole transport material production. By reducing the number of synthetic steps and utilizing robust, non-precious metal bases like potassium phosphate, the overall consumption of high-value reagents is drastically lowered. Furthermore, the simplified purification process, which relies on standard column chromatography rather than preparative HPLC, reduces solvent waste and energy consumption, leading to significant operational expenditure savings in large-scale manufacturing facilities.
- Enhanced Supply Chain Reliability: The reliance on widely available building blocks ensures that production schedules are not held hostage by the lead times of niche intermediates. The robustness of the Suzuki coupling chemistry means that reaction yields are consistently high, reducing the need for re-processing batches and ensuring a steady flow of finished goods. This reliability is crucial for reducing lead time for high-purity electronic chemicals, allowing downstream device manufacturers to maintain just-in-time inventory levels without the fear of stockouts that often plague the supply of specialized photovoltaic materials.
- Scalability and Environmental Compliance: The synthesis operates in common organic solvents like 1,4-dioxane and dichloromethane, which are well-understood in industrial waste management systems, facilitating easier compliance with environmental regulations. The absence of highly toxic or unstable reagents simplifies the safety protocols required for plant operation, enabling safer commercial scale-up of complex organic semiconductors. Additionally, the high thermal stability of the final products reduces the failure rate of solar cells in the field, lowering the warranty liabilities and replacement costs for module integrators over the lifespan of the energy asset.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel hole transport material technology. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation, providing a clear understanding of the material's performance envelope and manufacturing feasibility. Understanding these nuances is vital for R&D teams evaluating the integration of these materials into existing device architectures.
Q: What are the thermal stability advantages of these new hole transport materials compared to Spiro-OMeTAD?
A: The dithieno[3,2-b:2',3'-d]pyrrole core materials exhibit significantly higher glass transition temperatures (Tg ranging from 91°C to 106°C) and thermal decomposition temperatures exceeding 410°C, ensuring superior morphological stability in device operation compared to traditional small molecules.
Q: How does the synthetic route impact the cost structure for large-scale production?
A: The synthesis utilizes widely available carbazole derivatives and standard palladium catalysts in a convergent two-step sequence, eliminating the complex multi-step spiral synthesis required for Spiro-OMeTAD, thereby drastically reducing raw material complexity and processing costs.
Q: Can the energy levels of these materials be tuned for different perovskite compositions?
A: Yes, by modifying the substituent X on the phenyl ring (e.g., methoxy or fluorine groups) and the alkyl chain R on the pyrrole nitrogen, the HOMO energy levels can be precisely adjusted between -5.11 eV and -5.17 eV to match various perovskite absorbers.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Dithieno Pyrrole Derivative Supplier
As the global demand for high-efficiency perovskite solar cells accelerates, the need for a partner who can bridge the gap between laboratory innovation and industrial reality has never been more critical. NINGBO INNO PHARMCHEM stands at the forefront of this transition, leveraging our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production to bring these advanced hole transport materials to market. Our state-of-the-art facilities are equipped with stringent purity specifications and rigorous QC labs capable of detecting trace impurities that could affect device performance, ensuring that every batch meets the exacting standards required by top-tier photovoltaic manufacturers.
We invite forward-thinking enterprises to collaborate with us to unlock the full potential of this technology. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific production volumes and quality requirements. We encourage you to reach out today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that will secure your supply chain and enhance the competitiveness of your next-generation energy products.
