Advanced Synthesis of 1-Hydroxyaminoanthraquinone for High-Performance Dye Manufacturing
Advanced Synthesis of 1-Hydroxyaminoanthraquinone for High-Performance Dye Manufacturing
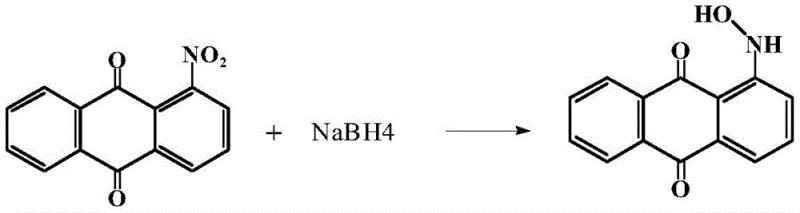
The global demand for high-performance anthraquinone dyes continues to drive innovation in the synthesis of key intermediates, specifically targeting methods that balance efficiency with environmental compliance. A pivotal advancement in this sector is detailed in patent CN113527134B, which discloses a novel, green synthesis route for 1-hydroxyaminoanthraquinone (CAS: 56613-39-9). This compound serves as a critical building block for a wide array of textile, plastic, and ink dyes, necessitating a production method that ensures exceptional purity and structural integrity. The patented technology leverages a sophisticated sodium borohydride reduction strategy within a binary solvent system, effectively addressing the longstanding challenges of selectivity and safety associated with traditional nitro-group reductions. By shifting away from hazardous reagents and complex catalytic systems, this methodology represents a significant leap forward for reliable dye intermediate suppliers aiming to modernize their manufacturing capabilities.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the preparation of 1-hydroxyaminoanthraquinone has relied on methodologies fraught with significant operational risks and inefficiencies that hinder large-scale commercial viability. Traditional literature often cites the use of hydrazine hydrate as a reducing agent, a substance known for its toxicity and potential explosiveness, requiring stringent safety protocols that inflate operational costs. Furthermore, alternative routes utilizing catalytic hydrogenation with palladium on carbon (Pd/C) introduce the complexity of handling high-pressure hydrogen gas and the subsequent necessity for rigorous removal of trace heavy metal residues to meet pharmaceutical or high-grade dye specifications. These conventional processes frequently suffer from poor selectivity, where the reaction inadvertently proceeds past the desired hydroxylamino stage to form 1-aminoanthraquinone as a dominant byproduct, thereby drastically reducing yield and complicating downstream purification efforts.
The Novel Approach
In stark contrast, the innovative approach outlined in the patent utilizes sodium borohydride (NaBH4) in a carefully tuned mixture of N,N-dimethylformamide (DMF) and water to achieve superior control over the reduction pathway. This method operates under atmospheric pressure and mild temperatures ranging from 10°C to 90°C, effectively eliminating the need for specialized high-pressure reactors or pyrophoric catalysts. The strategic selection of the DMF-water solvent system plays a dual role: it ensures the complete dissolution of the lipophilic 1-nitroanthraquinone substrate while simultaneously facilitating the reactivity of the hydrophilic borohydride species. This homogeneous reaction environment allows for precise kinetic control, enabling the reduction to halt selectively at the hydroxylamino stage with minimal formation of the over-reduced amine impurity, thus streamlining the entire production workflow.
Mechanistic Insights into Sodium Borohydride Reduction in Binary Solvents
The core mechanistic advantage of this synthesis lies in the unique interaction between the borohydride anion and the nitro group within the polar aprotic-protic solvent matrix. Unlike catalytic hydrogenation which relies on surface adsorption and can be difficult to stop at intermediate oxidation states, the chemical reduction by NaBH4 proceeds through a defined electron transfer mechanism that is highly sensitive to solvent polarity and proton availability. The presence of water in the DMF mixture provides the necessary protons to stabilize the intermediate nitroso and hydroxylamino species, preventing further reduction to the amine. The patent data indicates that optimizing the molar ratio of the reducing agent is critical; an excess of NaBH4 can drive the reaction towards the amine, whereas a controlled stoichiometric balance favors the accumulation of the target 1-hydroxyaminoanthraquinone. This delicate balance ensures that the electron density is managed precisely to achieve the desired functional group transformation without compromising the anthraquinone backbone.
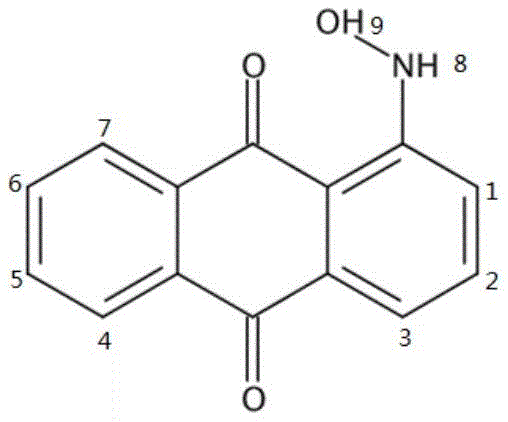
Structural verification of the product is paramount for R&D directors concerned with impurity profiles, and the patent provides robust analytical data confirming the successful synthesis. The use of advanced spectroscopic techniques, such as proton nuclear magnetic resonance (1H NMR), allows for the definitive assignment of the hydroxylamino protons distinct from the aromatic ring protons. As illustrated in the structural analysis, the characteristic signals for the NH and OH protons appear in the downfield region, clearly distinguishing the product from the starting nitro material or the amino byproduct. This level of analytical clarity is essential for quality control, ensuring that the final intermediate meets the rigorous purity standards required for the synthesis of complex anthraquinone dyes used in high-end textile and polymer applications.
How to Synthesize 1-Hydroxyaminoanthraquinone Efficiently
Implementing this synthesis route requires careful attention to solvent ratios and temperature control to maximize yield and selectivity. The process begins with the dissolution of the substrate in the optimized DMF-water mixture, followed by the controlled addition of the reducing agent. Maintaining the reaction temperature within the specified window is crucial to prevent thermal degradation or runaway exotherms, although the mild nature of NaBH4 generally offers a wide safety margin. For a comprehensive understanding of the operational parameters, including specific mass ratios and workup procedures, please refer to the detailed technical guide below which outlines the standardized protocol derived from the patent examples.
- Dissolve 1-nitroanthraquinone in a mixed solution of N,N-dimethylformamide (DMF) and water with a volume ratio optimized between 0.5: 1 and 3:1.
- Add sodium borohydride (NaBH4) to the solution at a molar ratio of 0.5: 1 to 10:1 relative to the substrate, maintaining a reaction temperature between 10°C and 90°C.
- After reaction completion (5-60 min), perform aqueous workup followed by dichloromethane extraction to isolate the solid 1-hydroxyaminoanthraquinone product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this sodium borohydride-based methodology offers substantial strategic benefits beyond mere chemical efficiency. The elimination of noble metal catalysts like palladium removes a significant variable cost component and mitigates the supply chain risks associated with fluctuating precious metal prices. Additionally, the avoidance of high-pressure hydrogenation equipment reduces the capital expenditure required for plant infrastructure, allowing for more flexible manufacturing setups that can be rapidly scaled to meet market demand. The simplified workup procedure, which involves standard solvent extraction and distillation, minimizes the generation of hazardous waste streams, aligning with increasingly strict global environmental regulations and reducing the overhead costs associated with waste disposal and treatment.
- Cost Reduction in Manufacturing: The replacement of expensive catalytic systems with commodity-grade sodium borohydride results in a direct reduction in raw material costs. By removing the need for catalyst recovery and regeneration steps, the overall process throughput is increased, leading to lower unit costs per kilogram of produced intermediate. Furthermore, the high selectivity of the reaction minimizes the loss of valuable starting material to byproducts, ensuring that the theoretical yield is closely approached in practical operations, which is a key driver for profitability in high-volume dye intermediate manufacturing.
- Enhanced Supply Chain Reliability: Utilizing widely available reagents such as DMF, water, and sodium borohydride ensures a robust and resilient supply chain that is less susceptible to geopolitical disruptions compared to specialized catalysts or high-purity hydrogen sources. The mild reaction conditions also reduce the risk of unplanned shutdowns due to equipment failure or safety incidents, guaranteeing consistent delivery schedules for downstream customers. This reliability is critical for maintaining continuous production lines in the textile and coating industries, where delays in intermediate supply can have cascading effects on finished goods availability.
- Scalability and Environmental Compliance: The inherent safety of operating at atmospheric pressure and moderate temperatures makes this process exceptionally easy to scale from pilot batches to multi-ton commercial production without significant engineering hurdles. The reduced environmental footprint, characterized by the absence of heavy metal contamination and lower energy consumption, facilitates easier regulatory approval and permitting in various jurisdictions. This compliance advantage accelerates time-to-market for new dye formulations and secures long-term operational licenses, positioning manufacturers as leaders in sustainable chemical production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for decision-making. Understanding these nuances helps stakeholders evaluate the feasibility of adopting this route for their specific production needs and quality requirements.
Q: What are the primary advantages of the NaBH4 reduction method over traditional catalytic hydrogenation?
A: The sodium borohydride method described in patent CN113527134B eliminates the need for expensive and potentially hazardous noble metal catalysts like Pd/C. It operates under milder conditions without high-pressure hydrogen equipment, significantly enhancing operational safety and reducing capital expenditure for reactor infrastructure while maintaining high selectivity for the hydroxylamino group.
Q: How does the solvent system impact the selectivity of 1-hydroxyaminoanthraquinone?
A: The patent highlights that the volume ratio of DMF to water is critical. A ratio between 1:1 and 3:1 optimizes the solubility of both the organic substrate and the inorganic reducing agent. This specific polarity balance suppresses the over-reduction to 1-aminoanthraquinone, ensuring the reaction stops selectively at the hydroxylamino stage, which is crucial for downstream dye synthesis.
Q: Is this synthesis method scalable for industrial production of dye intermediates?
A: Yes, the process is highly scalable due to its simplicity and lack of specialized high-pressure equipment. The short reaction time (5-60 minutes) and straightforward workup procedure involving standard solvent extraction make it ideal for large-scale batch processing, offering a reliable supply chain solution for manufacturers of anthraquinone-type dyes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1-Hydroxyaminoanthraquinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of the global dye and fine chemical industries. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless and efficient. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of 1-hydroxyaminoanthraquinone performs consistently in your downstream applications. Our dedication to process optimization allows us to offer competitive pricing without compromising on the structural integrity or coloristic properties of the final material.
We invite you to collaborate with us to leverage this advanced synthesis technology for your supply chain. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can enhance your operational efficiency and product quality. Let us be your partner in driving innovation and sustainability in the dye intermediate sector.
