Strategic Manufacturing of Novel 6-Fluoro-2,4-Diaminonitrobenzene for Advanced Pharmaceutical Intermediates
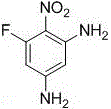
The pharmaceutical and fine chemical industries are constantly seeking novel building blocks that offer unique structural motifs for drug discovery and advanced material synthesis. Patent CN109096123B introduces a groundbreaking approach to synthesizing 6-fluoro-2,4-diaminonitrobenzene, a previously unreported compound with significant potential as a versatile intermediate. This innovation addresses critical bottlenecks in the production of fluorine-containing nitroanilines, which are essential precursors for herbicides, antibacterial agents, and specialized dyes. Unlike traditional methods that often rely on harsh conditions, this patent discloses a remarkably mild and efficient pathway that utilizes readily available reagents. For R&D directors and procurement strategists, this represents a pivotal shift towards more sustainable and cost-effective manufacturing protocols. The ability to access this specific substitution pattern—retaining a fluorine atom at the 6-position while introducing amino groups at the 2 and 4 positions—opens new avenues for molecular diversification in medicinal chemistry.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of poly-substituted fluorinated anilines has been plagued by significant technical and economic challenges that hinder large-scale adoption. Conventional routes typically necessitate the use of high-temperature and high-pressure reactors to drive nucleophilic aromatic substitution reactions, which drastically increases capital expenditure and energy consumption. Furthermore, many existing protocols depend on expensive transition metal catalysts to achieve acceptable conversion rates, introducing complex downstream purification requirements to remove trace metal residues that are strictly regulated in pharmaceutical applications. These legacy methods often suffer from poor selectivity, leading to difficult-to-separate impurity profiles that compromise the overall yield and quality of the final product. The reliance on hazardous reagents like anhydrous ammonia or strong bases also poses substantial safety risks and environmental compliance burdens for manufacturing facilities. Consequently, the high operational costs and safety liabilities associated with these traditional processes have severely limited the widespread application of valuable fluorine-containing aromatic compounds in commercial settings.
The Novel Approach
In stark contrast to these cumbersome legacy techniques, the method disclosed in patent CN109096123B leverages a sophisticated yet operationally simple strategy centered on the use of ammonium bicarbonate as a nitrogen source. This innovative protocol allows for the selective displacement of fluorine atoms on the 2,4,6-trifluoronitrobenzene scaffold under ambient conditions, specifically at temperatures ranging from 25°C to 35°C. By eliminating the need for extreme thermal energy or pressurized systems, this approach fundamentally transforms the risk profile and economic viability of producing 6-fluoro-2,4-diaminonitrobenzene. The reaction proceeds efficiently in industrial ethanol, a green and recyclable solvent, further enhancing the environmental footprint of the process. The simplicity of the workup procedure, involving merely suction filtration and recrystallization, ensures that the product can be isolated with high purity without the need for chromatographic separation. This paradigm shift not only reduces the barrier to entry for manufacturing this novel intermediate but also aligns perfectly with modern principles of green chemistry and process intensification.
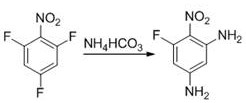
Mechanistic Insights into Ammonium Bicarbonate-Mediated Amination
The core of this technological breakthrough lies in the nuanced mechanistic pathway of the nucleophilic aromatic substitution (SnAr) facilitated by ammonium bicarbonate. In this system, ammonium bicarbonate acts as a controlled release source of ammonia, which serves as the nucleophile to attack the electron-deficient aromatic ring activated by the strong electron-withdrawing nitro group. The presence of three fluorine atoms on the starting material creates multiple potential sites for substitution; however, the reaction conditions are finely tuned to favor the displacement of the fluorines at the 2 and 4 positions while preserving the fluorine at the 6-position. This regioselectivity is crucial for maintaining the synthetic utility of the final molecule, as the remaining fluorine atom serves as a handle for further functionalization in downstream drug synthesis. The mild basicity provided by the bicarbonate ion likely assists in deprotonating the intermediate sigma-complex, driving the reaction forward without promoting excessive side reactions or decomposition of the sensitive nitro-aniline product. Understanding this delicate balance is essential for process chemists aiming to replicate or optimize this route for commercial scale-up.
From an impurity control perspective, the choice of ammonium bicarbonate and ethanol as the reaction medium plays a pivotal role in defining the quality of the final API intermediate. Traditional amination methods often generate complex mixtures of mono-, di-, and tri-substituted byproducts that are structurally similar and difficult to separate. However, the specific stoichiometry employed in this patent, utilizing a molar ratio of ammonium bicarbonate to substrate between 5:1 and 8:1, ensures complete conversion of the starting material while minimizing over-reaction or degradation. The subsequent recrystallization from an ethanol-water mixture exploits the differential solubility of the target diamino compound versus any potential impurities, effectively polishing the crude product to a high degree of purity. This robust purification capability is a critical advantage for supply chain managers, as it reduces the risk of batch failures due to out-of-specification impurity levels. The resulting pale yellow crystals exhibit consistent physical properties, facilitating reliable handling and formulation in subsequent manufacturing steps.
How to Synthesize 6-Fluoro-2,4-Diaminonitrobenzene Efficiently
Implementing this synthesis route requires careful attention to the specific operational parameters outlined in the patent to ensure optimal yield and reproducibility. The process begins with the dissolution of 2,4,6-trifluoronitrobenzene in 95% ethanol, followed by the controlled addition of ammonium bicarbonate to maintain the desired exotherm and mixing efficiency. The reaction mixture is then allowed to stand undisturbed for a period of 10 to 12 hours, a duration that is critical for allowing the slow, selective amination to reach completion without the need for aggressive agitation or heating. Following the reaction period, the product precipitates from the solution and is collected via standard suction filtration, washed to remove residual salts and mother liquor, and finally subjected to recrystallization using a 2:1 volume ratio of ethanol to water. This straightforward sequence of unit operations minimizes the need for specialized equipment, making it an ideal candidate for rapid technology transfer from the laboratory to pilot and commercial plants. For detailed standardized operating procedures and safety data sheets regarding this specific synthesis, please refer to the technical guide below.
- Dissolve 2,4,6-trifluoronitrobenzene in industrial ethanol and add ammonium bicarbonate at a molar ratio of 5: 1 to 8:1.
- Allow the mixture to stand at room temperature (25-35°C) for 10 to 12 hours to facilitate selective amination.
- Perform suction filtration, wash the solids, and recrystallize from an ethanol-water solution to obtain high-purity pale yellow crystals.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this novel synthesis method translates directly into tangible strategic advantages that enhance overall business resilience. The elimination of expensive transition metal catalysts and high-pressure reactor requirements results in a drastic simplification of the manufacturing infrastructure, thereby lowering both capital investment and ongoing maintenance costs. Furthermore, the use of commodity chemicals like ammonium bicarbonate and ethanol ensures a stable and secure supply of raw materials, mitigating the risks associated with sourcing specialized or hazardous reagents that are subject to volatile market fluctuations. The mild reaction conditions also contribute to a safer working environment, reducing insurance premiums and regulatory compliance burdens associated with handling high-energy chemical processes. These factors collectively create a more robust and cost-efficient supply chain capable of meeting the demanding timelines of the global pharmaceutical market.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the removal of costly catalytic systems and the reduction of energy intensity. By operating at room temperature, the facility eliminates the substantial utility costs associated with heating large reactor volumes or maintaining cryogenic conditions for alternative reagents. Additionally, the simplified workup procedure, which relies on filtration and crystallization rather than complex distillation or chromatography, significantly reduces solvent consumption and waste disposal fees. The high yields reported in the patent examples, reaching up to 94.5%, further maximize the utilization of raw materials, ensuring that every kilogram of input generates maximum value. This holistic reduction in operational overhead allows for a more competitive pricing structure for the final intermediate without compromising on quality margins.
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by the reliance on single-source suppliers for exotic reagents or the logistical challenges of transporting hazardous gases. This method circumvents those risks by utilizing solid ammonium bicarbonate, which is stable, easy to transport, and widely available from multiple global vendors. The robustness of the reaction against minor variations in conditions also means that production schedules are less likely to be disrupted by batch failures or the need for re-processing. For supply chain heads, this predictability is invaluable, as it allows for more accurate forecasting and inventory management. The ability to produce this key intermediate reliably ensures that downstream customers can maintain their own production schedules for finished drugs or agrochemicals without interruption.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces unforeseen engineering challenges, but the linear nature of this synthesis makes it exceptionally amenable to expansion. The absence of exothermic runaway risks associated with high-pressure ammonia additions allows for safe scale-up from kilogram batches to multi-ton annual production capacities. From an environmental standpoint, the process aligns with increasingly stringent global regulations by minimizing the generation of hazardous waste and avoiding the use of persistent organic pollutants or heavy metals. The aqueous ethanol mother liquor can be easily recovered and recycled, further reducing the environmental footprint of the manufacturing site. This commitment to sustainability not only satisfies regulatory requirements but also enhances the corporate social responsibility profile of the manufacturing partner, a key consideration for modern multinational corporations.
Frequently Asked Questions (FAQ)
To assist technical teams in evaluating the feasibility of integrating this novel intermediate into their pipelines, we have compiled answers to common inquiries based on the patent data. These questions address critical aspects of the synthesis, including reagent selection, purity control, and scalability potential. Understanding these technical nuances is essential for making informed decisions regarding process adoption and supplier qualification. The following insights are derived directly from the experimental data and claims presented in the intellectual property documentation.
Q: What are the primary advantages of using ammonium bicarbonate over traditional amination reagents?
A: Ammonium bicarbonate serves as a mild, solid nitrogen source that eliminates the need for high-pressure ammonia gas or hazardous liquid ammonia, significantly improving operational safety and reducing equipment costs.
Q: How does this novel synthesis method impact the purity profile of the final intermediate?
A: The mild reaction conditions (25-35°C) minimize side reactions and thermal degradation, while the simple ethanol-water recrystallization step effectively removes impurities, ensuring high chemical purity suitable for sensitive pharmaceutical applications.
Q: Is this process scalable for commercial production of fluorine-containing nitroanilines?
A: Yes, the process relies on standard unit operations like dissolution, static reaction, and filtration, avoiding complex catalytic cycles or extreme conditions, which makes it highly amenable to scale-up from kilogram to multi-ton production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 6-Fluoro-2,4-Diaminonitrobenzene Supplier
As the demand for specialized fluorinated intermediates continues to grow, partnering with a manufacturer that possesses deep technical expertise and scalable infrastructure is paramount. NINGBO INNO PHARMCHEM stands at the forefront of this industry, leveraging extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production to deliver consistent quality. Our state-of-the-art facilities are equipped to handle the specific requirements of this novel synthesis, ensuring that stringent purity specifications are met through our rigorous QC labs and advanced analytical capabilities. We understand that the successful commercialization of new drug candidates depends on a supply chain that is both agile and reliable, and we are committed to providing that stability to our global partners.
We invite you to engage with our technical team to explore how this cost-effective synthesis route can be integrated into your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain a clearer understanding of the economic benefits tailored to your volume needs. We encourage potential partners to contact our technical procurement team to索取 specific COA data and route feasibility assessments, ensuring that your transition to this novel intermediate is seamless and scientifically sound. Let us collaborate to bring your next generation of pharmaceutical innovations to market faster and more efficiently.
