Advanced Synthesis Strategy for High-Purity Sugammadex Sodium Impurity Reference Standards
Introduction to Patent CN111019016B and Strategic Importance
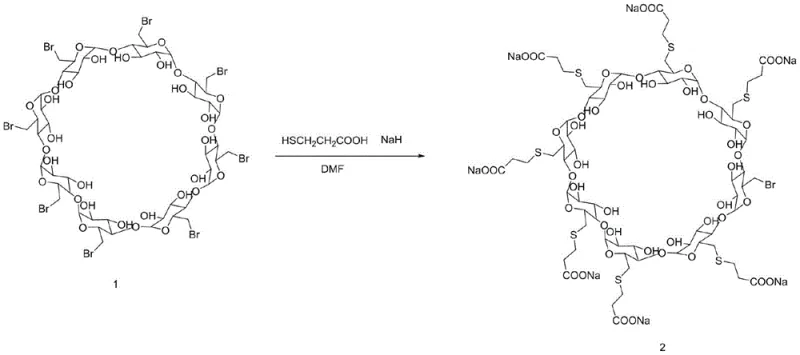
The pharmaceutical industry relies heavily on the availability of high-quality reference standards to ensure the safety and efficacy of active pharmaceutical ingredients (APIs), and patent CN111019016B addresses a critical gap in the supply chain for Sugammadex sodium quality control materials. Sugammadex sodium, known commercially as Bridion, represents a milestone in anesthesia reversal agents, functioning as a selective relaxant binding agent that encapsulates rocuronium bromide. To maintain rigorous quality assurance during the manufacturing of this complex cyclodextrin derivative, regulatory bodies require well-characterized impurities for method validation and stability testing. This patent discloses a novel synthesis method specifically targeting the hepta-substituted impurity of Sugammadex sodium, a compound where only seven of the eight available hydroxyl sites on the gamma-cyclodextrin ring are modified with 2-carboxyethylthio groups, leaving one bromine atom intact. By providing a robust and reproducible pathway to generate this specific structural variant, the technology enables analytical laboratories to accurately detect and quantify trace impurities in the final drug product, thereby safeguarding patient safety and ensuring compliance with global pharmacopoeia standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Sugammadex and its related substances has relied heavily on methodologies described in foundational patents such as US6670340B1, which typically utilize octa-(6-iodo-6-deoxy)-gamma-cyclodextrin as the starting material. While effective for producing the fully substituted API, these conventional iodo-based routes present significant challenges when attempting to isolate specific under-substituted impurities for reference purposes. The carbon-iodine bond is highly reactive, often leading to rapid and exhaustive substitution that makes it difficult to arrest the reaction at the hepta-substituted stage without generating a complex mixture of products. Furthermore, iodine-containing reagents are substantially more expensive than their bromine counterparts, driving up the raw material costs for non-API applications like reference standard production. The handling of iodo-intermediates also introduces additional safety and waste disposal concerns due to the potential release of elemental iodine and the generation of heavy halogenated waste streams, which complicates the environmental footprint of the manufacturing process and increases the burden on waste treatment facilities.
The Novel Approach
The methodology outlined in patent CN111019016B offers a transformative solution by shifting the synthetic strategy to utilize octa-(6-bromo-6-deoxy)-gamma-cyclodextrin as the key starting material, coupled with precise stoichiometric control to favor the formation of the target impurity. By reacting the bromo-cyclodextrin with 3-mercaptopropionic acid in the presence of an alkali base at moderate temperatures ranging from 40 to 50°C, the process achieves a controlled nucleophilic substitution that naturally limits the degree of substitution. The innovation lies in the specific molar ratios employed, where the amount of thiol and base is carefully restricted relative to the cyclodextrin substrate, effectively preventing the eighth substitution event and preserving the single bromine atom required for the impurity structure. This approach not only simplifies the purification workflow by reducing the formation of fully substituted byproducts but also leverages the lower cost and improved availability of bromo-precursors compared to iodo-analogues. The result is a streamlined, cost-efficient process that delivers the target hepta-substituted compound with exceptional purity, overcoming the selectivity issues inherent in previous high-energy substitution methods.
Mechanistic Insights into Nucleophilic Substitution on Cyclodextrin
The core chemical transformation driving this synthesis is a classic nucleophilic substitution reaction where the thiolate anion, generated from 3-mercaptopropionic acid, attacks the electrophilic carbon centers at the 6-position of the gamma-cyclodextrin ring. In the presence of a strong base such as sodium hydride, sodium methoxide, or potassium carbonate, the thiol group of 3-mercaptopropionic acid is deprotonated to form a highly reactive thiolate species. This nucleophile then displaces the bromine leaving group on the cyclodextrin scaffold through an SN2 mechanism, forming a stable thioether linkage. The reaction kinetics are finely tuned by the choice of solvent, with polar aprotic solvents like dimethylformamide (DMF) or dimethyl sulfoxide (DMSO) playing a crucial role in solvating the cationic counter-ions while leaving the nucleophilic thiolate relatively unsolvated and thus more reactive. This solvent effect enhances the rate of substitution at the primary hydroxyl positions of the cyclodextrin, ensuring efficient conversion while maintaining the structural integrity of the macrocyclic ring system.
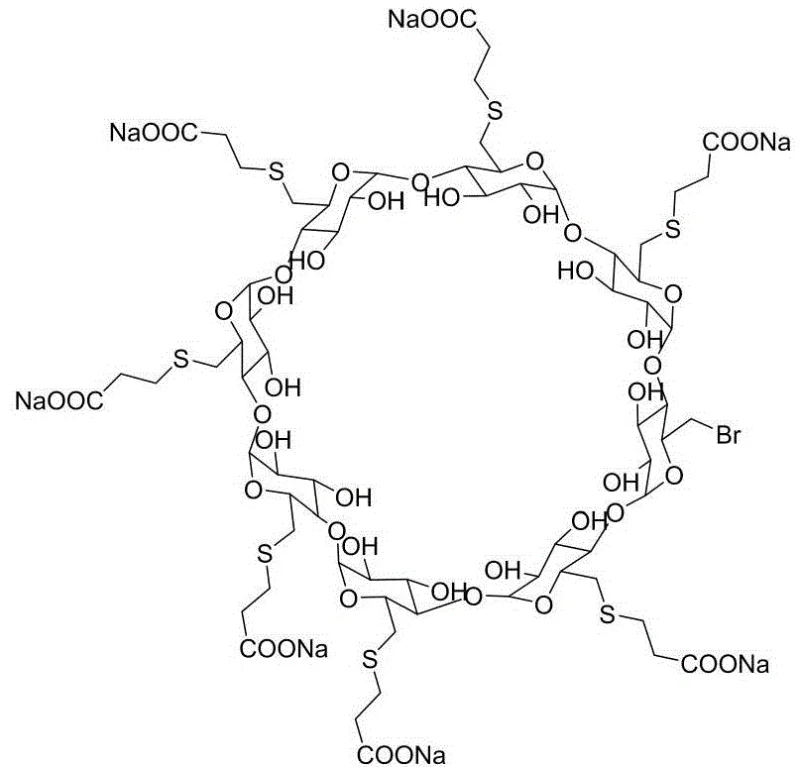
Controlling the impurity profile in this reaction is fundamentally a exercise in stoichiometric precision and thermodynamic management. Unlike the synthesis of the parent API which drives towards complete octa-substitution, the goal here is to halt the reaction after seven substitutions have occurred. The patent achieves this by employing a molar ratio of cyclodextrin to 3-mercaptopropionic acid to alkali of approximately 1:(3.5-4.5):(7.5-8.5). This deliberate deficit of nucleophile ensures that statistically, not all eight bromine sites are attacked, enriching the reaction mixture with the desired hepta-substituted species. Furthermore, the moderate reaction temperature of 40-50°C provides sufficient energy to overcome the activation barrier for substitution without promoting excessive side reactions or degradation of the sensitive thioether bonds. The subsequent purification steps, involving pulping with absolute ethanol and recrystallization from water-organic solvent mixtures, exploit the subtle solubility differences between the hepta-substituted impurity, the unreacted starting material, and the fully substituted octa-product, allowing for the isolation of the target compound with purity exceeding 99%.
How to Synthesize Sugammadex Impurity Efficiently
The practical execution of this synthesis requires careful attention to reaction conditions and purification protocols to ensure the high quality of the final reference standard. The process begins with the preparation of the reaction mixture under an inert nitrogen atmosphere to prevent oxidation of the thiol reagent, followed by the controlled addition of reagents to manage exotherms and ensure homogeneity. Once the substitution reaction is complete, the workup involves a series of crystallization and washing steps designed to remove inorganic salts and organic byproducts. For a comprehensive understanding of the operational parameters and specific equipment requirements, please refer to the detailed standardized synthesis steps provided in the guide below.
- Prepare the reaction mixture by adding solvent A (such as DMF or DMSO) and 3-mercaptopropionic acid, followed by batch addition of alkali under nitrogen protection.
- Dropwise add the mixture of octa-(6-bromo-6-deoxy)-gamma-cyclodextrin and solvent A, then heat to 40-50°C and react for 14-20 hours.
- Isolate the crude product via suction filtration, then purify through pulping with absolute ethanol and multiple recrystallization steps using water and solvent B.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this bromo-based synthesis route offers substantial strategic advantages over traditional iodo-based methods, particularly regarding cost structure and raw material security. The shift from iodine to bromine chemistry significantly reduces the direct material costs, as bromo-cyclodextrin derivatives are generally more accessible and less expensive to manufacture on a large scale than their iodo-counterparts. This cost reduction in pharmaceutical intermediate manufacturing is further amplified by the simplified purification process; because the reaction selectivity is higher, fewer downstream processing steps are required to isolate the target impurity, leading to reduced solvent consumption and lower utility costs. Additionally, the use of common industrial solvents like DMF and ethanol ensures that the supply chain is not dependent on exotic or restricted chemicals, enhancing the reliability of supply and reducing the risk of production delays due to raw material shortages.
- Cost Reduction in Manufacturing: The elimination of expensive iodine reagents and the optimization of stoichiometric ratios lead to a drastic simplification of the bill of materials. By avoiding the need for excess reagents to drive full conversion, the process minimizes waste generation and lowers the cost of goods sold (COGS). The ability to achieve high purity through simple recrystallization rather than complex chromatographic separation further drives down operational expenses, making the production of high-purity reference standards economically viable for large-scale quality control programs.
- Enhanced Supply Chain Reliability: Utilizing widely available reagents such as 3-mercaptopropionic acid and standard alkali bases ensures a robust supply chain that is less susceptible to geopolitical or logistical disruptions. The mild reaction conditions (40-50°C) reduce the energy intensity of the process and allow for the use of standard glass-lined or stainless steel reactors, facilitating easier technology transfer between manufacturing sites. This flexibility supports a resilient supply network capable of meeting the fluctuating demands of the global pharmaceutical market without compromising on delivery timelines.
- Scalability and Environmental Compliance: The process is inherently scalable, moving seamlessly from laboratory benchtop synthesis to commercial production volumes without requiring specialized high-pressure or cryogenic equipment. The reduced reliance on heavy halogens like iodine aligns with modern green chemistry principles, simplifying waste treatment and disposal procedures. This environmental compatibility not only reduces regulatory compliance burdens but also supports corporate sustainability goals, making the manufacturing process more attractive to environmentally conscious stakeholders and regulatory agencies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of Sugammadex sodium impurities. These answers are derived directly from the technical specifications and experimental data disclosed in patent CN111019016B, providing clarity on the feasibility and benefits of this novel manufacturing route. Understanding these details is essential for quality assurance teams and procurement specialists evaluating new suppliers or methods for reference standard production.
Q: Why is the bromo-substituted route preferred over the iodo-substituted route for this impurity?
A: The bromo-substituted route utilizes octa-(6-bromo-6-deoxy)-gamma-cyclodextrin, which is generally more cost-effective and easier to handle than the iodo-analogues described in earlier patents, while still achieving high conversion rates under mild conditions.
Q: How is the specific hepta-substituted impurity selectively synthesized?
A: Selectivity is achieved by strictly controlling the molar ratios of the reactants, specifically using a deficit of 3-mercaptopropionic acid and alkali relative to the cyclodextrin substrate, preventing full octa-substitution.
Q: What purity levels can be achieved with this purification method?
A: Through a combination of ethanol pulping and repeated recrystallization using solvent systems like water/DMF or water/methanol, the final impurity purity consistently reaches over 99%, making it suitable as a reference standard.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Sugammadex Impurity Supplier
As a leader in the fine chemical and pharmaceutical intermediate sector, NINGBO INNO PHARMCHEM is uniquely positioned to leverage advanced technologies like patent CN111019016B to deliver superior value to our global partners. Our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production ensures that we can meet the rigorous volume requirements of major pharmaceutical manufacturers while maintaining stringent purity specifications. Our state-of-the-art rigorous QC labs are equipped to perform comprehensive impurity profiling and structural characterization, guaranteeing that every batch of Sugammadex impurity meets the highest international standards for reference materials. We understand that consistency and reliability are paramount in the supply of critical quality control substances, and our dedicated technical team is committed to supporting your regulatory filings and quality assurance initiatives.
We invite you to collaborate with us to optimize your supply chain for Sugammadex-related materials and explore the cost-saving potential of this innovative synthesis method. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume needs and quality requirements. We encourage you to reach out today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that enhance both the efficiency and compliance of your pharmaceutical manufacturing operations.
