Advanced Synthesis of Allylated Monophosphine Ligands for Commercial Catalysis Applications
Introduction to Next-Generation Phosphine Ligand Technology
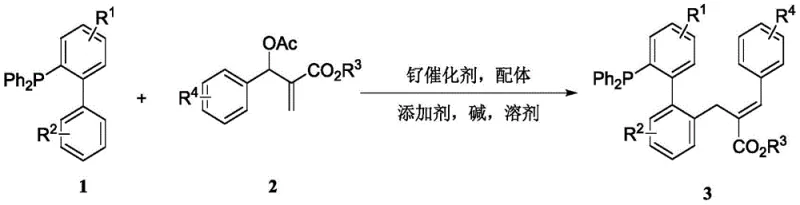
The landscape of homogeneous catalysis is continuously evolving, driven by the demand for more efficient and cost-effective ligand systems. Patent CN114478627B introduces a significant breakthrough in the synthesis of allylated monophosphine ligands, specifically focusing on functionalized biphenyl phosphines. These compounds are critical components in transition metal-catalyzed cross-coupling reactions, which are foundational to the manufacturing of active pharmaceutical ingredients (APIs) and advanced fine chemicals. The disclosed technology addresses the longstanding challenge of introducing allyl groups onto the biphenyl backbone with high regioselectivity and yield, bypassing the limitations of traditional methods that often rely on scarce and expensive noble metals.
This innovation provides a robust pathway for producing high-purity pharmaceutical intermediates and specialty catalysts. By leveraging a ruthenium-catalyzed system, the method ensures that the resulting ligands possess the necessary steric and electronic properties to enhance catalyst activity in downstream applications. For procurement and supply chain leaders, this represents a shift towards more sustainable and economically viable sourcing strategies for complex organophosphorus compounds, ensuring continuity in the production of high-value chemical entities.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the functionalization of biphenyl monophosphine ligands has been constrained by the reliance on rhodium-based catalytic systems for C-H activation. Literature precedents, such as those published in Angewandte Chemie and Nature Communications, describe ortho-alkylation processes that require expensive rhodium catalysts and specific olefin coupling reagents. These conventional approaches often suffer from high operational costs due to the price volatility of rhodium and the complexity of the reaction conditions required to achieve acceptable selectivity. Furthermore, the multi-step nature of traditional functionalization sequences introduces additional purification burdens, leading to increased waste generation and longer lead times for final product delivery.
The dependency on precious metals like rhodium not only inflates the cost of goods sold but also introduces supply chain vulnerabilities associated with the mining and refining of these scarce resources. Additionally, the harsh conditions sometimes required for C-H activation can limit the tolerance of sensitive functional groups, thereby restricting the structural diversity of the ligand library available for drug discovery and process development. These factors collectively hinder the rapid scale-up and commercial adoption of new ligand architectures in industrial settings.
The Novel Approach
In contrast, the methodology described in patent CN114478627B utilizes a ruthenium-catalyzed allylation strategy that fundamentally simplifies the synthesis of these valuable ligands. The process employs readily available biphenyl phosphines and acrylate derivatives as starting materials, reacting them in a single step under relatively mild conditions. This one-pot transformation eliminates the need for pre-functionalized substrates or complex protecting group strategies, drastically reducing the number of unit operations required. The use of ruthenium, a more abundant and cost-effective metal compared to rhodium, significantly lowers the raw material cost basis while maintaining high catalytic efficiency.
The reaction system is highly versatile, accommodating a wide range of substituents on both the phosphine and the acrylate components, as evidenced by the diverse array of examples provided in the patent data. This flexibility allows for the rapid generation of ligand libraries tailored for specific catalytic applications without compromising on yield or purity. The streamlined workflow, combined with the use of common organic solvents and standard inorganic bases, makes this approach exceptionally well-suited for commercial scale-up, offering a clear advantage in terms of both economic efficiency and operational simplicity for manufacturers of fine chemical intermediates.
Mechanistic Insights into Ruthenium-Catalyzed Allylation
The core of this technological advancement lies in the unique catalytic cycle facilitated by the ruthenium complex in conjunction with specific amino acid-derived additives. The reaction mechanism likely involves the activation of the allylic acetate precursor by the ruthenium center, forming a reactive pi-allyl ruthenium intermediate. This species then undergoes nucleophilic attack by the electron-rich biphenyl phosphine substrate, guided by the steric environment created by the ancillary ligands and additives. The presence of additives such as Boc-protected amino acids plays a crucial role in modulating the electronic properties of the metal center, thereby enhancing regioselectivity and preventing unwanted side reactions such as isomerization or polymerization of the allyl group.
Furthermore, the choice of base and phosphine additives contributes to the stabilization of the active catalytic species and the neutralization of acidic byproducts generated during the transformation. This careful balancing of reaction components ensures that the C-C bond formation occurs selectively at the desired position on the biphenyl ring, preserving the integrity of the phosphine moiety. The result is a clean reaction profile with minimal impurity formation, which is critical for downstream applications where trace metal contamination or organic impurities can poison subsequent catalytic cycles. This level of control underscores the sophistication of the patented process and its suitability for producing high-specification materials.
How to Synthesize Allylated Monophosphine Ligands Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for reproducing these high-value ligands in a laboratory or pilot plant setting. The procedure involves charging a reactor with the biphenyl phosphine substrate and the acrylate derivative, followed by the addition of the ruthenium catalyst precursor, such as p-cymene ruthenium dichloride dimer. Essential to the success of the reaction is the inclusion of specific additives like Boc-1-aminocyclohexane-1-carboxylic acid and phosphine ligands like tris(4-methoxyphenyl)phosphine, which optimize the catalytic turnover. The mixture is then heated in a solvent like n-hexane under an inert argon atmosphere to drive the reaction to completion.
- Combine biphenyl phosphine substrate and acrylate derivative with a ruthenium catalyst precursor.
- Add specific amino acid-based ligands, inorganic bases, and phosphine additives to the reaction mixture.
- Heat the mixture in an organic solvent under inert atmosphere at 60-140°C to achieve high-yield allylation.
Commercial Advantages for Procurement and Supply Chain Teams
For organizations focused on cost reduction in pharmaceutical intermediate manufacturing, this patented process offers substantial strategic benefits. The shift from rhodium to ruthenium catalysis represents a direct material cost saving, as ruthenium salts are generally more affordable and less subject to extreme market fluctuations than their rhodium counterparts. Moreover, the consolidation of the synthesis into a single step reduces labor costs, energy consumption, and solvent usage, all of which contribute to a lower overall cost of production. The high yields reported in the patent examples, reaching up to 94% for certain derivatives, further enhance the economic viability by maximizing the output from each batch of raw materials.
From a supply chain reliability perspective, the use of commodity chemicals as starting materials ensures a stable and secure supply base. Unlike specialized reagents that may have long lead times or single-source dependencies, the biphenyl phosphines and acrylates used in this process are widely available from multiple global suppliers. This diversification mitigates the risk of production delays caused by raw material shortages. Additionally, the simplified workup procedure, which typically involves filtration and distillation, reduces the dependency on complex chromatographic purification equipment, allowing for faster turnaround times and increased throughput in existing manufacturing facilities.
Environmental compliance and scalability are also key advantages of this technology. The process generates minimal waste, aligning with green chemistry principles and reducing the burden on waste treatment infrastructure. The absence of hazardous reagents and the use of relatively benign solvents facilitate easier regulatory approval and safer operating conditions for personnel. These factors collectively make the technology highly scalable, enabling manufacturers to transition smoothly from gram-scale development to tonne-scale commercial production without significant process re-engineering, thus ensuring a reliable supply of high-purity catalysts for the global market.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this allylation technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for evaluating the process feasibility. Understanding these details is essential for R&D teams planning to integrate these ligands into their catalytic workflows or for procurement specialists assessing the long-term viability of the supply chain.
Q: What are the advantages of this Ru-catalyzed method over traditional Rh-catalyzed C-H activation?
A: The patented Ru-catalyzed method eliminates the need for expensive rhodium catalysts and complex multi-step functionalization, significantly reducing raw material costs and simplifying the operational workflow for large-scale production.
Q: What is the typical yield range for these allylated ligands?
A: According to the patent data, laboratory-scale yields vary depending on substituents but can reach up to 94% for specific ester derivatives, demonstrating high efficiency and atom economy suitable for industrial optimization.
Q: Is this process scalable for commercial manufacturing?
A: Yes, the process is designed for scalability, utilizing readily available raw materials, standard organic solvents, and simple workup procedures like filtration and distillation, which minimizes equipment requirements and waste generation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Allylated Monophosphine Ligand Supplier
NINGBO INNO PHARMCHEM stands at the forefront of custom synthesis and contract development, possessing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our technical team is well-versed in the nuances of organometallic chemistry and is fully equipped to adapt the ruthenium-catalyzed allylation process described in CN114478627B to meet your specific volume and purity requirements. We maintain stringent purity specifications and operate rigorous QC labs to ensure that every batch of allylated monophosphine ligand meets the exacting standards required for sensitive pharmaceutical and agrochemical applications.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis route can optimize your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the potential economic benefits tailored to your specific project needs. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your next generation of catalytic processes is built on a foundation of reliability, quality, and cost-efficiency.
