Advanced Chiral Oxazoline Phosphine Ligands: Enabling High-Precision Asymmetric Synthesis for Global Pharma
The landscape of asymmetric catalysis is continuously evolving, driven by the demand for more efficient and selective chiral ligands capable of managing complex radical intermediates. Patent CN116178431A introduces a groundbreaking class of nitrogen-nitrogen-phosphorus (N,N,P) ligands featuring a chiral oxazoline backbone integrated with a phosphine or phosphine oxide moiety. These structures represent a significant departure from conventional bisoxazoline systems, offering a unique trifunctional coordination environment that enhances stereocontrol in transition metal catalysis. The core innovation lies in the modular design, where the electronic and steric properties of both the phosphine group and the oxazoline substituents can be independently tuned to optimize performance in challenging transformations. 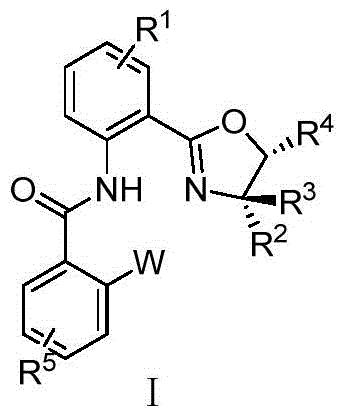
For R&D directors and process chemists, this patent provides a robust platform for developing next-generation catalytic systems, particularly for radical-based asymmetric cross-coupling reactions which have historically been difficult to control. The versatility of the general formula I allows for extensive derivatization, enabling the fine-tuning required for specific substrate classes in pharmaceutical intermediate synthesis. By addressing the limitations of existing ligand libraries, this technology opens new avenues for constructing chiral carbon-carbon triple bonds and other complex motifs with high fidelity.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional chiral ligands, such as bisoxazolines (BOX) or cinchona alkaloid derivatives, have served as the workhorses of asymmetric synthesis for decades. However, they often exhibit limited adaptability when applied to radical-mediated processes, where the transient nature of free radical intermediates demands precise spatial and electronic stabilization. Conventional P,N-ligands frequently suffer from rigid structural constraints that prevent optimal orbital overlap with the metal center during the critical bond-forming steps. Furthermore, many existing systems lack the modularity to adjust the electronic density at the phosphorus atom without completely redesigning the chiral backbone, leading to suboptimal enantioselectivity in oxidative coupling reactions. This rigidity often results in broad substrate scope limitations, requiring bespoke ligand development for each new reaction type, which significantly increases R&D timelines and costs.
The Novel Approach
The novel approach detailed in CN116178431A overcomes these hurdles by introducing a flexible yet robust N,N,P scaffold that decouples the steric bulk from the electronic tuning elements. By incorporating a phosphine group directly onto the benzamide side chain adjacent to the oxazoline ring, the ligand creates a hemilabile coordination sphere that can dynamically adjust during the catalytic cycle. This design allows for the simultaneous stabilization of the metal center and the radical intermediate, a crucial factor in achieving high enantiomeric excess (ee) values, with some embodiments reporting up to 92% ee in Sonogashira-type couplings. The ability to oxidize the phosphine to a phosphine oxide further expands the chemical space, allowing chemists to switch between soft and hard donor atoms to match the specific requirements of the metal catalyst, whether it be copper, palladium, or iridium.
Mechanistic Insights into Cu-Catalyzed Radical Asymmetric Coupling
The mechanistic superiority of these ligands stems from their ability to form stable yet reactive complexes with copper salts, facilitating the generation and control of radical species. In the context of oxidative Sonogashira cross-coupling, the N,N,P ligand coordinates to the copper center, creating a chiral pocket that dictates the trajectory of the incoming alkyne and the radical precursor. The phosphine moiety acts as a strong sigma-donor, increasing the electron density on the metal to promote single-electron transfer (SET) processes essential for radical generation. Simultaneously, the chiral oxazoline ring imposes strict steric constraints that differentiate between the pro-chiral faces of the intermediate, ensuring high stereoselectivity. This dual functionality minimizes background racemic reactions and suppresses side pathways such as homocoupling or beta-hydride elimination, which are common pitfalls in radical chemistry.
From an impurity control perspective, the well-defined coordination geometry reduces the formation of off-cycle catalyst species that often lead to difficult-to-remove metal residues or organic byproducts. The robustness of the ligand structure under oxidative conditions (as evidenced by the stability of the phosphine oxide variants) ensures that the catalyst remains active throughout the reaction duration, typically ranging from 24 to 120 hours at mild temperatures like 0°C to room temperature. This stability translates to cleaner reaction profiles, simplifying downstream purification processes such as silica gel chromatography or crystallization. For manufacturing teams, this means higher throughput and reduced solvent consumption, as fewer recrystallization steps are needed to meet stringent pharmaceutical purity specifications.
How to Synthesize Oxazoline Phosphine Ligands Efficiently
The synthesis of these high-value ligands follows a logical and scalable two-step sequence that avoids exotic reagents or extreme conditions, making it highly attractive for commercial production. The process begins with the condensation of substituted 2-aminobenzonitriles with chiral amino alcohols in the presence of a Lewis acid catalyst, typically zinc chloride, in chlorobenzene at elevated temperatures around 130°C. This cyclization step constructs the chiral oxazoline core with moderate to good yields. The subsequent step involves an amide coupling reaction between the resulting amine intermediate and various substituted benzoic acids containing the phosphine moiety. This coupling is mediated by standard carbodiimide reagents like EDCI and catalytic DMAP in dichloromethane at room temperature, ensuring the integrity of the sensitive phosphine group is maintained. 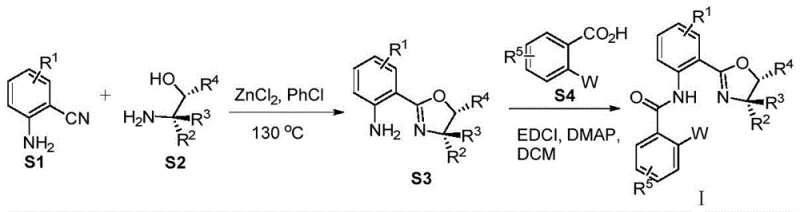
- React 2-aminobenzonitrile derivatives with chiral amino alcohols using ZnCl2 in chlorobenzene at 130°C to form the oxazoline intermediate.
- Couple the intermediate with substituted benzoic acids using EDCI and DMAP in dichloromethane at room temperature to yield the final ligand.
- Optionally oxidize the phosphine moiety to phosphine oxide using aqueous hydrogen peroxide for specific electronic tuning.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this ligand technology offers substantial strategic benefits beyond mere technical performance. The synthetic route relies on commodity chemicals such as chlorobenzene, dichloromethane, zinc chloride, and EDCI, all of which are readily available in the global market at stable prices. This reliance on established supply chains mitigates the risk of raw material shortages that often plague projects dependent on specialized or proprietary building blocks. Furthermore, the reaction conditions are operationally simple, utilizing standard glass-lined reactors and avoiding the need for cryogenic cooling or high-pressure equipment, which significantly lowers the capital expenditure (CAPEX) required for technology transfer and scale-up.
- Cost Reduction in Manufacturing: The streamlined two-step synthesis eliminates the need for multiple protection and deprotection sequences often seen in complex ligand synthesis, thereby reducing the overall number of unit operations. The use of room temperature conditions for the final coupling step drastically cuts energy consumption compared to processes requiring prolonged heating or cooling. Additionally, the high selectivity of the ligand in catalytic applications reduces the loading of expensive precious metal catalysts like copper or palladium required to achieve target conversion, leading to direct savings on metal costs and waste disposal fees associated with heavy metal removal.
- Enhanced Supply Chain Reliability: The structural modularity of the ligand library means that if a specific substituent becomes unavailable, alternative analogs with similar steric profiles can often be synthesized using interchangeable starting materials without redesigning the entire process. The robustness of the intermediates allows for potential stockpiling of the oxazoline core, decoupling the production timeline from the final customization step. This flexibility ensures continuous supply continuity even in the face of fluctuating market demands for specific chiral intermediates, providing a buffer against logistical disruptions.
- Scalability and Environmental Compliance: The process generates minimal hazardous waste, as the primary byproducts are water-soluble salts and urea derivatives from the coupling reagent, which are easily separated during aqueous workup. The avoidance of toxic solvents like DMF or NMP in favor of DCM and chlorobenzene (which can potentially be replaced with greener alternatives in future optimizations) aligns with increasingly strict environmental regulations. The high yields reported in the patent examples suggest that the process is atom-economical, reducing the volume of organic waste per kilogram of product and simplifying the environmental impact assessment for large-scale manufacturing facilities.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of these oxazoline phosphine ligands in industrial settings. These insights are derived directly from the experimental data and claims within the patent documentation, providing a factual basis for decision-making. Understanding these details helps stakeholders evaluate the feasibility of integrating this technology into existing production lines for chiral pharmaceutical ingredients.
Q: What represents the key structural advantage of these N,N,P ligands over traditional bisoxazolines?
A: The integration of a phosphine moiety allows for independent tuning of electronic and steric properties distinct from the oxazoline ring, offering superior control in radical asymmetric reactions.
Q: Are the synthesis conditions scalable for industrial production?
A: Yes, the process utilizes robust reagents like ZnCl2 and EDCI under standard thermal conditions (130°C) and room temperature coupling, avoiding cryogenic or ultra-high pressure requirements.
Q: What specific reactions do these ligands facilitate?
A: They are particularly effective in copper-catalyzed radical asymmetric oxidative Sonogashira cross-coupling reactions, achieving enantiomeric excess (ee) values up to 92%.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Ligand Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced chiral ligands play in accelerating drug discovery and process development. As a dedicated CDMO partner, we possess the technical expertise to not only supply these complex oxazoline phosphine ligands but also to customize them for your specific catalytic needs. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop discovery to full-scale manufacturing is seamless and compliant with cGMP standards. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced chiral HPLC and NMR capabilities to guarantee the quality of every batch.
We invite you to collaborate with us to leverage this cutting-edge ligand technology for your next asymmetric synthesis project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific process requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our reliable chiral ligand supplier capabilities can drive efficiency and innovation in your manufacturing operations.
