Scalable Synthesis of Aliskiren Intermediates via POBr3 for Commercial API Manufacturing
Scalable Synthesis of Aliskiren Intermediates via POBr3 for Commercial API Manufacturing
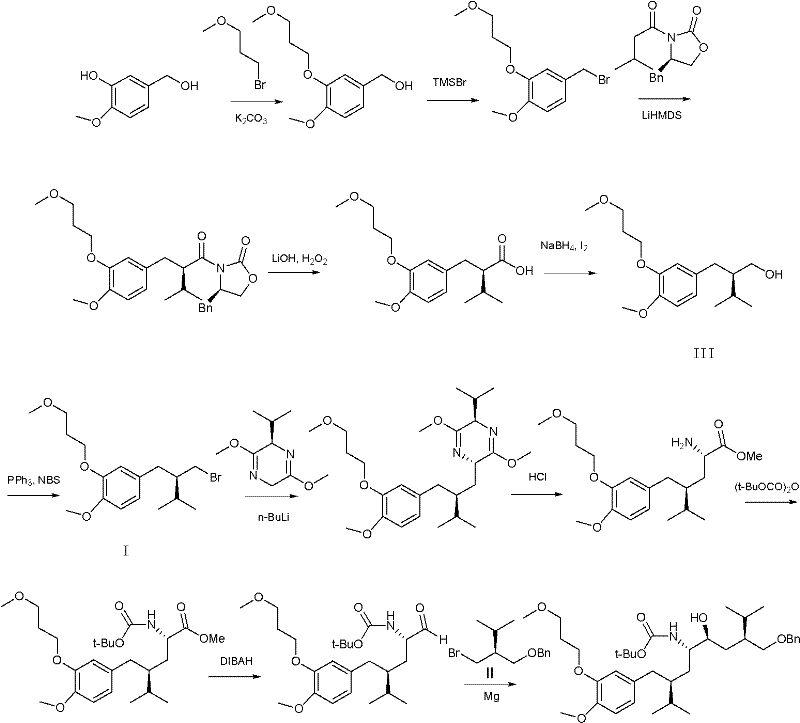
The global demand for effective antihypertensive therapies continues to drive innovation in the synthesis of Renin inhibitors, with Aliskiren standing out as a pivotal first-in-class non-peptide agent. The commercial viability of such complex small molecules relies heavily on the efficiency of producing key building blocks. Patent CN102001920A introduces a transformative preparation method for two critical intermediates, designated as Compound I and Compound II, which are essential precursors in the total synthesis of Aliskiren. This technology addresses the longstanding bottleneck of low yields and difficult purification associated with legacy routes. By shifting from phosphine-based halogenation reagents to tribromophosphine oxide (POBr3), the process achieves a dramatic improvement in operational simplicity and product quality. The following analysis details how this chemical innovation translates into tangible value for pharmaceutical manufacturers seeking robust supply chains.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methodologies, specifically those referenced in patent US5559111A, rely on the Appel reaction conditions using triphenylphosphine (Ph3P) and N-bromosuccinimide (NBS) in methylene chloride. While chemically feasible on a gram scale, this approach presents severe disadvantages for industrial application. The reaction typically proceeds with mediocre conversion rates, resulting in isolated yields ranging merely from 50% to 60%. More critically, the crude reaction mixture contains significant amounts of triphenylphosphine oxide and succinimide byproducts that are chemically similar to the target bromide. Consequently, the isolation of pure Intermediate I or II mandates the use of column chromatography on silica gel. In a commercial setting, column chromatography is economically prohibitive due to high solvent consumption, low throughput, and the generation of massive solid waste, rendering the process unsuitable for multi-kilogram or ton-scale production required for API manufacturing.
The Novel Approach
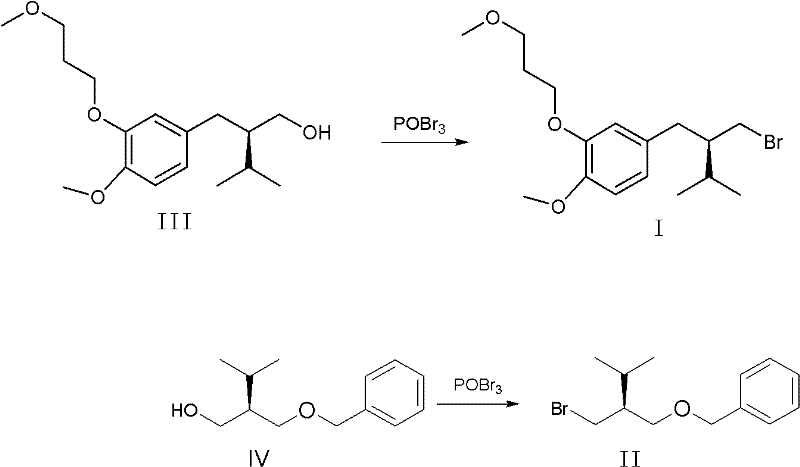
The methodology disclosed in CN102001920A fundamentally re-engineers the halogenation step by employing tribromophosphine oxide as the brominating agent. This reagent activates the hydroxyl group of the precursor alcohols (Compound III or IV) more efficiently, facilitating a cleaner substitution reaction. As illustrated in the reaction scheme below, this switch eliminates the formation of bulky phosphine oxide byproducts that complicate purification. The result is a crude product of sufficient purity that can be processed using standard unit operations. For Intermediate I, simple recrystallization from methanol or ethyl acetate/hexane mixtures suffices, while Intermediate II can be purified via reduced pressure distillation. This transition from chromatographic separation to crystallization or distillation represents a paradigm shift in process chemistry, enabling high-throughput manufacturing with significantly reduced environmental footprint and operational costs.
Mechanistic Insights into POBr3-Mediated Bromination
The core of this technological advancement lies in the mechanistic efficiency of tribromophosphine oxide compared to the Ph3P/NBS system. In the conventional Appel reaction, the activation of the alcohol involves the formation of an oxyphosphonium intermediate, followed by nucleophilic attack by bromide. However, the stoichiometric generation of triphenylphosphine oxide creates a highly polar impurity that is notoriously difficult to separate from non-polar organic bromides without chromatography. In contrast, the POBr3 mechanism likely proceeds through the formation of a dibromophosphite ester intermediate, which is subsequently displaced by bromide ion. The byproduct of this reaction is phosphoric acid derivatives or HBr, which are water-soluble and easily removed during the aqueous workup phase described in the patent. This inherent difference in byproduct polarity is the key enabler for the simplified purification strategy, allowing the organic phase to be washed clean with aqueous sodium bicarbonate and brine, leaving the target molecule in the organic layer with high fidelity.
Furthermore, the preservation of stereochemical integrity is paramount for chiral drugs like Aliskiren. The patent data explicitly confirms that the POBr3-mediated transformation proceeds without racemization, maintaining chiral purity above 99.9%. This suggests that the reaction conditions—specifically the temperature control between 20°C and 80°C and the choice of aprotic solvents like toluene or DMF—are optimized to favor an SN2-type displacement with inversion (or double inversion depending on the specific activation mode) that does not allow for the formation of a planar carbocation intermediate. Such control over the stereocenter ensures that the downstream Grignard coupling reactions, which are sensitive to steric and electronic properties of the halide, proceed with high diastereoselectivity, ultimately securing the quality of the final active pharmaceutical ingredient.
How to Synthesize Aliskiren Intermediates Efficiently
The implementation of this synthesis route requires precise control over reaction parameters to maximize the benefits of the POBr3 chemistry. The process begins with the dissolution of the chiral alcohol precursor in a dry, aprotic solvent, followed by the controlled addition of the brominating agent. The subsequent workup leverages liquid-liquid extraction to remove inorganic salts and acidic byproducts, followed by a physical separation technique tailored to the specific physical properties of the target intermediate. This approach bypasses the need for specialized chromatographic equipment, making it accessible for standard multipurpose chemical plants.
- Dissolve the hydroxy-precursor (Compound III or IV) in an aprotic solvent such as toluene or DMF and slowly add tribromophosphine oxide while controlling temperature between 20-80°C.
- Quench the reaction mixture with water, extract the organic phase using hexane or petroleum ether, and wash sequentially with saturated sodium bicarbonate and brine.
- Concentrate the organic layer and purify the crude product via recrystallization (for Intermediate I) or vacuum distillation (for Intermediate II) to achieve >99% purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of the POBr3 route described in CN102001920A offers compelling economic and logistical advantages over the legacy Ph3P/NBS method. The primary driver of value is the elimination of column chromatography, which is a major cost center in fine chemical manufacturing. By replacing this batch-intensive, solvent-hungry process with recrystallization or distillation, manufacturers can drastically reduce solvent procurement costs and waste disposal fees. Additionally, the increase in yield from approximately 50% to nearly 80% means that less raw material is required to produce the same amount of intermediate, directly lowering the cost of goods sold (COGS). This efficiency gain is compounded by the reduction in processing time, as crystallization and distillation are significantly faster than running and processing large silica columns, thereby increasing plant capacity and throughput without capital expenditure.
- Cost Reduction in Manufacturing: The shift to POBr3 chemistry removes the dependency on expensive silica gel and the vast volumes of eluent solvents required for column chromatography. This qualitative change in the purification workflow translates to substantial cost savings in both material consumption and waste treatment. Furthermore, the higher reaction yield reduces the burden on upstream synthesis steps, as less starting material is needed to meet production targets, creating a ripple effect of efficiency throughout the entire supply chain.
- Enhanced Supply Chain Reliability: Reliance on column chromatography often introduces bottlenecks due to the limited loading capacity of columns and the variability of manual packing processes. By transitioning to scalable unit operations like crystallization and distillation, the manufacturing process becomes more robust and predictable. This stability ensures consistent delivery schedules and reduces the risk of supply disruptions caused by purification failures. The use of common, commodity chemicals like toluene and POBr3 also mitigates the risk of raw material shortages compared to more specialized reagents.
- Scalability and Environmental Compliance: The new method aligns perfectly with green chemistry principles by minimizing waste generation. The absence of silica gel waste and the reduction in solvent usage lower the environmental impact of the manufacturing process, simplifying regulatory compliance and permitting. From a scalability perspective, the ability to purify tons of material via distillation or crystallization in standard reactors allows for seamless scale-up from pilot plant to commercial production, ensuring that supply can meet market demand for this critical cardiovascular medication.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These insights are derived directly from the experimental data and claims within patent CN102001920A, providing a factual basis for evaluating the technology's fit within your existing manufacturing framework. Understanding these nuances is essential for R&D teams planning technology transfer and for procurement teams assessing supplier capabilities.
Q: Why is the POBr3 method superior to the traditional Ph3P/NBS method for Aliskiren intermediates?
A: The traditional method utilizing triphenylphosphine and N-bromosuccinimide typically yields only 50-60% and necessitates column chromatography for purification, which is impractical for large-scale production. The POBr3 method achieves yields around 80% and allows for purification via simple recrystallization or distillation, significantly enhancing scalability and reducing waste.
Q: Does the use of tribromophosphine oxide affect the chiral purity of the intermediates?
A: No, the process described in patent CN102001920A maintains excellent stereochemical integrity. The reaction conditions are mild enough to prevent racemization, consistently delivering products with chiral purity exceeding 99.9%, which is critical for the biological activity of the final antihypertensive drug.
Q: What are the preferred solvents for this bromination reaction?
A: The patent specifies the use of aprotic solvents to ensure reaction efficiency. Preferred options include toluene, N,N-dimethylformamide (DMF), acetonitrile, or dichloromethane. Toluene and DMF are highlighted as particularly effective for balancing reaction kinetics and ease of downstream workup.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aliskiren Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and robust infrastructure. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the efficiencies promised by patent CN102001920A are fully realized in practice. We operate stringent purity specifications and maintain rigorous QC labs equipped to verify the chiral integrity and chemical purity of every batch, guaranteeing that our Aliskiren intermediates meet the exacting standards required for global API registration.
We invite you to collaborate with us to optimize your supply chain for cardiovascular therapeutics. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis that quantifies the potential benefits of switching to this POBr3-based route for your specific volume requirements. Contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our advanced manufacturing capabilities can support your long-term strategic goals.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
