Scalable Synthesis of Quinolinyloxydiphenylcyclopropanedicarboxamide for Oncology Drug Development
Introduction to Advanced c-Met Inhibitor Synthesis
The development of small molecule drugs targeting cell proliferation and angiogenesis remains a cornerstone of modern oncology research, with the kinase c-Met emerging as a particularly attractive therapeutic target. Patent CN102227164A discloses a sophisticated and robust methodology for the preparation of quinolinyloxydiphenylcyclopropanedicarboxamide derivatives, specifically designed to inhibit c-Met signaling pathways effectively. This technology represents a significant leap forward in the synthesis of complex heterocyclic architectures, offering a convergent route that joins a functionalized cyclopropane core with a substituted quinoline moiety. For pharmaceutical manufacturers and R&D teams, understanding the nuances of this synthetic pathway is critical, as it addresses common bottlenecks associated with forming ether linkages between electron-deficient heterocycles and phenolic substrates. The disclosed methods provide a clear roadmap for generating high-purity intermediates essential for downstream drug substance manufacturing, ensuring that the final active pharmaceutical ingredients meet stringent regulatory standards for impurity profiles and structural integrity.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional approaches to synthesizing biaryl ethers or heteroaryl ethers often suffer from harsh reaction conditions that can compromise sensitive functional groups present in complex drug candidates. Classical nucleophilic aromatic substitution typically requires extremely high temperatures and strong bases, which can lead to decomposition of the cyclopropane ring or hydrolysis of amide bonds frequently found in these inhibitor scaffolds. Furthermore, older methodologies often rely on stoichiometric amounts of copper salts, which introduce significant challenges in terms of heavy metal removal and environmental compliance during the scale-up phase. The presence of residual copper in the final API is strictly regulated, necessitating additional purification steps such as chromatography or specialized scavenging treatments that drastically reduce overall yield and increase production costs. Additionally, linear synthesis strategies where the quinoline and the diphenylcyclopropane components are built sequentially on the same molecule often result in poor atom economy and difficult purification of intermediates due to similar polarity profiles.
The Novel Approach
The methodology outlined in the patent introduces a highly efficient convergent strategy that mitigates these risks by preparing the two major fragments independently before joining them in a final, controlled coupling step. This approach utilizes a palladium-catalyzed etherification reaction, which operates under milder conditions compared to classical nucleophilic substitution, thereby preserving the integrity of the sensitive cyclopropane dicarboxamide core. By employing specific phosphine ligands and optimized solvent systems, the reaction achieves high selectivity for the desired ether bond formation while minimizing side reactions such as dehalogenation of the quinoline ring. This modularity allows for the independent optimization of each fragment's synthesis, ensuring that both the cyclopropane core and the quinoline tail are of the highest possible purity before the final assembly. The use of catalytic rather than stoichiometric metal species also simplifies the downstream purification process, aligning with green chemistry principles and reducing the burden on waste treatment facilities.
Mechanistic Insights into Pd-Catalyzed Etherification
The core of this synthetic innovation lies in the palladium-catalyzed cross-coupling reaction between a phenolic hydroxyl group on the cyclopropane derivative and a halogenated position on the quinoline ring. The mechanism likely proceeds through an oxidative addition of the palladium(0) species into the carbon-halogen bond of the quinoline intermediate, forming a reactive aryl-palladium complex. This is followed by the coordination and deprotonation of the phenol substrate by the base present in the reaction mixture, generating a phenoxide species that undergoes transmetallation with the palladium center. The final reductive elimination step releases the coupled product and regenerates the active palladium catalyst, allowing the cycle to continue. The choice of ligand is paramount in this transformation; bulky, electron-rich phosphine ligands such as racemic 2-(di-tert-butylphosphino)-1,1'-binaphthyl stabilize the palladium center and facilitate the difficult reductive elimination step required to form the C-O bond. This mechanistic understanding allows chemists to fine-tune reaction parameters, such as temperature and base strength, to maximize turnover numbers and minimize catalyst loading, which is a key driver for cost efficiency in large-scale manufacturing.
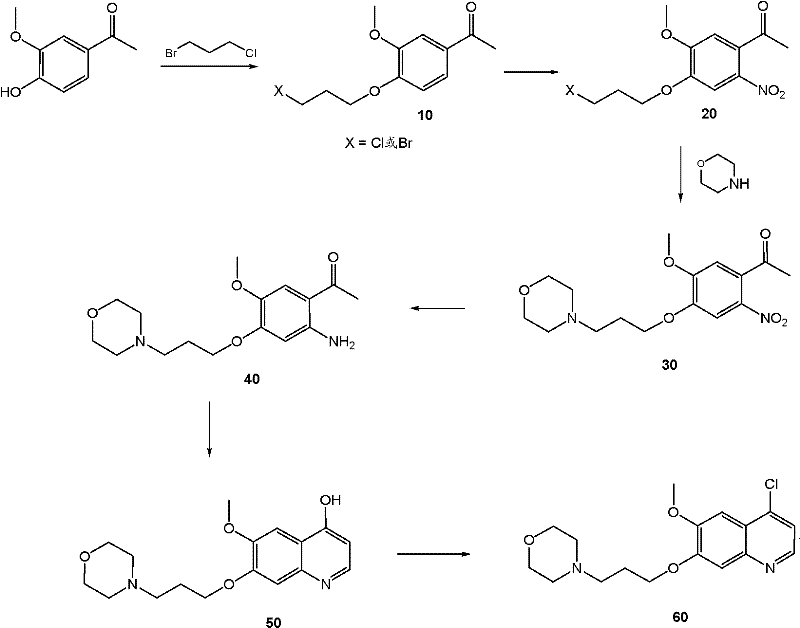
Impurity control is another critical aspect addressed by the detailed reaction conditions provided in the patent. The formation of the quinoline intermediate itself involves a multi-step sequence including nitration, reduction, and cyclization, each of which has the potential to generate regioisomers or over-reacted byproducts. The patent specifies the use of specific chlorinating agents like POCl3 and controlled temperatures during the cyclization of the aminoacetophenone precursor to ensure the chlorine atom is installed exclusively at the 4-position of the quinoline ring. Any deviation in this regioselectivity would render the intermediate useless for the subsequent coupling reaction, as the wrong isomer would not react or would form an inactive analog. Furthermore, the purification of the quinoline intermediate as a dihydrochloride salt prior to the coupling step serves as a powerful tool for removing non-basic impurities and unreacted starting materials, ensuring that only the highest quality electrophile enters the final coupling vessel. This rigorous control over intermediate quality directly translates to a cleaner reaction profile in the final step, reducing the complexity of the final purification and increasing the overall yield of the target molecule.
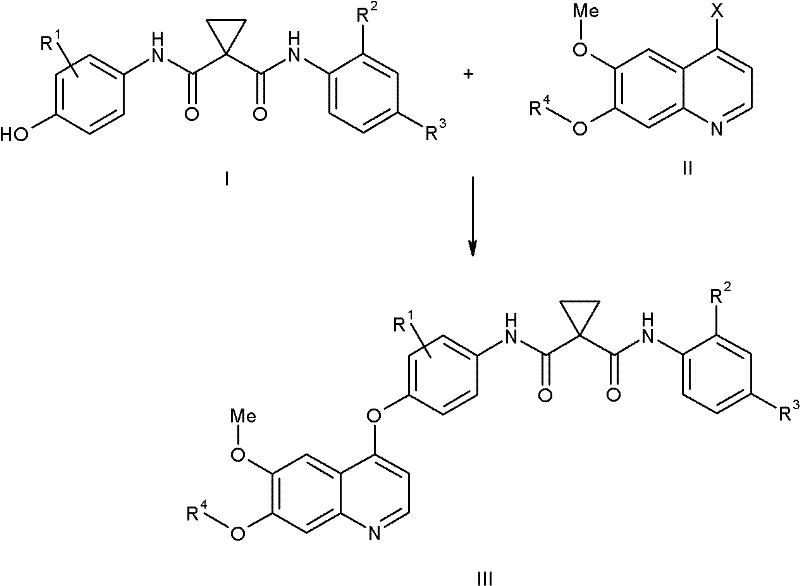
How to Synthesize Quinolinyloxydiphenylcyclopropanedicarboxamide Efficiently
The synthesis of these potent c-Met inhibitors requires a disciplined approach to process chemistry, balancing reaction kinetics with practical handling considerations. The patent provides a comprehensive guide that begins with the preparation of the cyclopropane dicarboxamide fragment, involving the activation of cyclopropane dicarboxylic acid followed by amidation with substituted anilines. Parallel to this, the quinoline fragment is constructed from readily available acetophenone derivatives through a sequence of alkylation, nitration, and reductive cyclization. The convergence of these two streams occurs in the final stage, where the phenolic hydroxyl of the cyclopropane fragment attacks the chloro-quinoline under palladium catalysis. Detailed protocols for workup and purification are included, emphasizing techniques like crystallization and pH-controlled extractions that are scalable and cost-effective. For a complete breakdown of the standardized operating procedures and specific reagent ratios required for this transformation, please refer to the technical guide below.
- Preparation of the cyclopropane dicarboxamide core (Formula I) via acylation of aminophenols with cyclopropane acid chlorides.
- Synthesis of the 4-chloroquinoline intermediate (Formula II) through nitration, reduction, and cyclization of acetophenone derivatives.
- Coupling of Formula I and Formula II using a Palladium catalyst and phosphine ligand in polar aprotic solvents to form the final ether linkage.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this synthetic route offers substantial strategic benefits that extend beyond simple yield improvements. The modular nature of the synthesis allows for the decoupling of supply chains for the two main fragments, reducing the risk of single-point failures in the production of the final API. Since the cyclopropane core and the quinoline tail can be manufactured at different sites or by different vendors, companies can diversify their supplier base and negotiate better pricing for bulk raw materials like acetophenones and cyclopropane acids. This flexibility is crucial for maintaining business continuity in the face of global supply chain disruptions or raw material shortages. Furthermore, the reliance on commodity chemicals and standard reagents such as thionyl chloride, potassium carbonate, and palladium acetate ensures that the process is not dependent on exotic or hard-to-source catalysts that might have long lead times or volatile pricing structures.
- Cost Reduction in Manufacturing: The elimination of stoichiometric heavy metal reagents and the use of catalytic palladium systems significantly lowers the cost of goods sold by reducing waste disposal fees and metal scavenging expenses. The ability to purify intermediates via crystallization rather than column chromatography further drives down operational costs, making the process economically viable for commercial-scale production. By optimizing the catalyst loading and ligand selection, the process minimizes the consumption of expensive palladium resources, which is a major cost driver in fine chemical synthesis. Additionally, the high purity of the intermediates reduces the need for extensive reprocessing or recycling of off-spec material, leading to a more streamlined and cost-efficient manufacturing workflow.
- Enhanced Supply Chain Reliability: The use of robust, scalable reaction conditions ensures that the manufacturing process can be reliably transferred from pilot plant to commercial production without significant re-optimization. The intermediates described are stable and can be stored for extended periods, allowing manufacturers to build up safety stock and buffer against demand fluctuations. The synthetic route avoids the use of hazardous reagents that require special handling permits or infrastructure, simplifying the logistics of raw material transport and storage. This stability and ease of handling contribute to a more predictable supply schedule, enabling pharmaceutical companies to plan their clinical trial material production and commercial launches with greater confidence.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing solvents and reaction conditions that are compatible with large-scale reactor equipment. The waste streams generated are primarily aqueous and organic phases that can be treated using standard effluent treatment protocols, reducing the environmental footprint of the manufacturing process. The avoidance of persistent organic pollutants and the minimization of heavy metal waste align with increasingly strict environmental regulations, future-proofing the supply chain against regulatory changes. This commitment to sustainable manufacturing practices not only reduces compliance risks but also enhances the corporate social responsibility profile of the final drug product.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthetic technology. These answers are derived directly from the experimental data and embodiments disclosed in the patent documentation, providing a reliable basis for process evaluation. Understanding these details is essential for technical teams assessing the feasibility of adopting this route for their specific development programs. The information covers critical aspects ranging from catalyst selection to impurity management, ensuring a comprehensive overview of the technology's capabilities.
Q: What are the critical parameters for the Pd-catalyzed coupling step?
A: The reaction requires precise control of temperature (typically 95-110°C), the use of specific phosphine ligands like rac-BINAP derivatives, and polar aprotic solvents such as NMP or toluene mixtures to ensure high conversion and minimize dehalogenation byproducts.
Q: How is the quinoline intermediate purified effectively?
A: The quinoline intermediate can be purified via crystallization from solvent mixtures like acetonitrile and water, or by forming salt forms such as dihydrochloride dihydrates, which significantly improves purity to over 99% before the final coupling step.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the patent details kilogram-scale preparations for key intermediates, utilizing robust reagents like thionyl chloride and POCl3, and standard workup procedures like filtration and distillation that are easily transferable to industrial reactors.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Quinolinyloxydiphenylcyclopropanedicarboxamide Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the race to bring life-saving oncology therapies to market. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from benchtop discovery to full-scale manufacturing. We are equipped with state-of-the-art rigorous QC labs and analytical capabilities to meet stringent purity specifications, guaranteeing that every batch of quinolinyloxydiphenylcyclopropanedicarboxamide intermediate we deliver meets the highest industry standards. Our commitment to quality and consistency makes us the ideal partner for pharmaceutical companies seeking a reliable source for complex c-Met inhibitor building blocks.
We invite you to contact our technical procurement team to discuss your specific requirements and explore how our manufacturing capabilities can support your supply chain goals. By requesting a Customized Cost-Saving Analysis, you can gain valuable insights into how optimizing this synthetic route can reduce your overall production costs. We are ready to provide specific COA data and route feasibility assessments tailored to your project's unique needs, helping you accelerate your development timeline and secure a competitive advantage in the marketplace.
