Advanced Synthesis of Nickel(II) Beta-Carboline Chelates for Oncology Drug Development
Advanced Synthesis of Nickel(II) Beta-Carboline Chelates for Oncology Drug Development
The landscape of oncology drug discovery is continuously evolving, with a significant resurgence of interest in metal-based therapeutics following the success of platinum drugs like cisplatin. Patent CN107446002B introduces a groundbreaking advancement in this field: a novel Nickel(II) nitrate chelate utilizing 1-pyridine-6-methoxy-β-carboline as a ligand. This innovation addresses critical limitations in current antitumor research by providing a structurally defined, synthetically accessible metal complex that exhibits superior biological activity compared to its organic precursors. For R&D directors and procurement specialists in the pharmaceutical sector, this technology represents a pivotal opportunity to access high-purity pharmaceutical intermediates with a robust and scalable supply chain. The structural integrity of this complex, featuring a central nickel ion coordinated with bioactive carboline ligands, offers a unique mechanism of action that could bypass resistance mechanisms common in traditional chemotherapy agents.
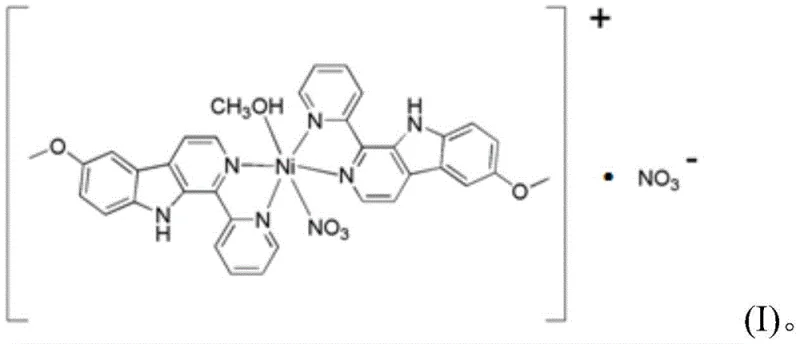
The core of this invention lies in the precise molecular architecture of the target compound, designated as Formula (I). Unlike naturally occurring beta-carboline alkaloids which suffer from variable composition and low abundance in plant sources such as Peganum harmala, this synthetic chelate provides a consistent and reproducible chemical entity. The complex is formed through the coordination of nickel(II) ions with two molecules of the specialized ligand 1-pyridine-6-methoxy-β-carboline (LKL), along with nitrate anions and a methanol molecule. This specific arrangement not only stabilizes the metal center but also enhances the lipophilicity and cellular uptake potential of the drug candidate. For manufacturers seeking a reliable pharmaceutical intermediate supplier, the ability to produce this well-characterized crystal structure eliminates the batch-to-batch variability that plagues botanical extracts, thereby ensuring stringent purity specifications required for clinical trials.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the acquisition of beta-carboline alkaloids for medicinal chemistry has been fraught with significant challenges that hinder large-scale drug development. The primary conventional method involves the extraction of these compounds from medicinal plants, a process that is inherently inefficient and environmentally taxing. The content of active beta-carboline alkaloids in natural sources is typically extremely low, necessitating the processing of massive quantities of biomass to obtain mere grams of pure material. Furthermore, the extraction process is chemically complex, often requiring multiple chromatographic steps to separate the target alkaloids from a myriad of structurally similar impurities and plant metabolites. This results in exorbitant costs and long lead times, making it economically unfeasible for the commercial scale-up of complex pharmaceutical intermediates. Additionally, the reliance on agricultural sources introduces supply chain vulnerabilities, where crop failures or seasonal variations can disrupt the continuity of raw material supply, posing a severe risk to pharmaceutical manufacturing timelines.
The Novel Approach
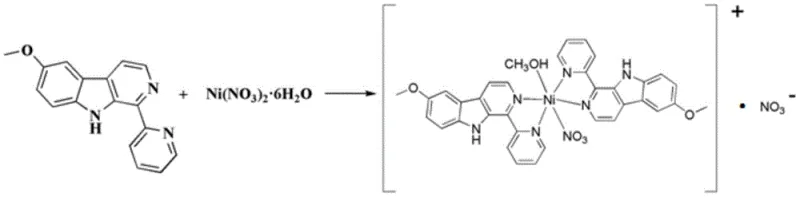
The methodology disclosed in the patent offers a transformative solution by shifting from extraction to total synthesis and coordination chemistry. Instead of relying on unpredictable natural sources, the process begins with the chemical synthesis of the ligand 1-pyridine-6-methoxy-β-carboline (LKL) from readily available starting materials like 5-methoxytryptamine and pyridine-2-carbaldehyde. This synthetic route allows for precise control over substitution patterns and purity levels that are impossible to achieve through extraction. Subsequently, the ligand undergoes a straightforward coordination reaction with nickel nitrate hexahydrate in a polar solvent system. This approach drastically simplifies the production workflow, reducing the number of unit operations and eliminating the need for extensive purification of natural extracts. The result is a high-yield process that produces golden-yellow crystalline solids with defined stoichiometry, facilitating cost reduction in API manufacturing and enabling a more predictable and stable supply of critical oncology intermediates.
Mechanistic Insights into Coordination and Cyclization
The synthesis mechanism involves two distinct chemical phases: the construction of the organic ligand and the subsequent metal coordination. The ligand synthesis utilizes a Pictet-Spengler condensation reaction, where the amine group of 5-methoxytryptamine reacts with the aldehyde group of pyridine-2-carbaldehyde under acidic catalysis to form a Schiff base intermediate. This intermediate then undergoes an intramolecular electrophilic aromatic substitution to close the ring, forming the tetrahydro-beta-carboline skeleton. Crucially, this is followed by an oxidative dehydrogenation step using Palladium on Carbon (Pd/C) at elevated temperatures (140-160°C), which aromatizes the pyridine ring to yield the fully conjugated beta-carboline system. This conjugated planar structure is essential for DNA intercalation potential, a key feature of its antitumor activity. The rigorous control of reaction conditions, such as maintaining pH levels between 7 and 9 during workup, ensures the minimization of side products and maximizes the yield of the pure ligand.
Following ligand preparation, the coordination mechanism proceeds through the interaction of the nickel(II) center with the nitrogen atoms of the ligand. The nickel ion, acting as a Lewis acid, accepts electron pairs from the pyridine nitrogen and the indole nitrogen of the beta-carboline framework. The patent details that the reaction can be driven either by ambient pressure reflux or by high-pressure solvothermal methods. In the solvothermal approach, the use of a sealed borosilicate glass tube allows the reaction mixture to reach temperatures above the boiling point of the solvent without evaporation, promoting the formation of high-quality single crystals. The inclusion of methanol in the coordination sphere suggests a labile binding site that could be displaced in a biological environment, potentially serving as a prodrug mechanism where the active species is released upon hydrolysis within the tumor microenvironment. This mechanistic understanding is vital for optimizing the process to ensure the correct isomer and hydration state are achieved consistently.
How to Synthesize 1-Pyridine-6-Methoxy-β-Carboline Nickel Chelate Efficiently
The efficient production of this high-value chelate requires strict adherence to the optimized reaction parameters outlined in the patent data. The process is designed to be flexible, accommodating both standard laboratory glassware for initial development and specialized pressure vessels for scaled-up crystallization. The key to success lies in the purity of the precursor ligand and the precise control of the solvent composition during the coordination step. By utilizing a mixed solvent system, typically dominated by methanol with minor co-solvents like chloroform or acetone, the solubility of both the organic ligand and the inorganic nickel salt is balanced to favor nucleation and crystal growth. Detailed standardized synthetic steps for replicating this high-yield process are provided in the technical guide below, ensuring that manufacturing teams can achieve the reported 85% yield and high crystallinity.
- Synthesize the ligand 1-Pyridine-6-Methoxy-β-Carboline (LKL) via Pictet-Spengler condensation of 5-methoxytryptamine and pyridine-2-carbaldehyde, followed by Pd/C oxidative dehydrogenation.
- Dissolve the purified LKL ligand and nickel nitrate hexahydrate in a polar solvent mixture, typically methanol with minor amounts of chloroform or acetone.
- Conduct the coordination reaction under heating (65-80°C) or solvothermal conditions (80°C, sealed tube) for 24-72 hours to precipitate the golden-yellow crystalline product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition from natural extraction to this synthetic coordination chemistry offers profound strategic benefits that extend beyond simple cost metrics. The primary advantage is the decoupling of raw material supply from agricultural variables. By synthesizing the ligand from commodity chemicals like tryptamine derivatives and pyridine aldehydes, manufacturers can secure a stable and continuous flow of inputs that are not subject to seasonal harvest cycles or geopolitical disruptions affecting botanical sourcing. This reliability is crucial for maintaining uninterrupted production schedules for downstream API manufacturing. Furthermore, the synthetic route allows for significant inventory optimization, as the starting materials are shelf-stable and widely available from multiple global chemical suppliers, reducing the risk of single-source dependency.
- Cost Reduction in Manufacturing: The elimination of labor-intensive extraction and purification steps associated with natural products leads to substantial operational savings. The synthetic pathway utilizes common reagents and standard reaction vessels, avoiding the need for specialized extraction equipment or large volumes of organic solvents required for biomass processing. Additionally, the high yield of the coordination reaction minimizes waste generation and maximizes atom economy, directly contributing to lower cost of goods sold (COGS). The ability to recycle solvents and the use of heterogeneous catalysts like Pd/C, which can be filtered and reused, further enhances the economic viability of the process for large-scale commercial production.
- Enhanced Supply Chain Reliability: The robustness of the chemical synthesis ensures consistent quality and quantity of output, which is a critical factor for regulatory compliance in the pharmaceutical industry. Unlike botanical extracts which can vary in potency and impurity profiles, this synthetic chelate offers a defined chemical structure that simplifies quality control testing and regulatory filing. The process scalability, demonstrated by the patent's coverage of both gram-scale and potential tonnage production via solvothermal methods, means that supply can be rapidly ramped up to meet clinical trial demands or commercial launch requirements without the long lead times associated with cultivating new crop cycles.
- Scalability and Environmental Compliance: The synthesis method aligns well with green chemistry principles by reducing the overall solvent footprint compared to extraction methods. The use of polar solvents like methanol and water mixtures, which are easier to recover and treat than the complex hydrocarbon mixtures often used in extraction, simplifies waste management. The high purity of the final crystalline product reduces the need for energy-intensive recrystallization steps, lowering the overall carbon footprint of the manufacturing process. This environmental efficiency not only reduces disposal costs but also positions the supply chain favorably against increasingly stringent environmental regulations governing pharmaceutical manufacturing facilities.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this nickel chelate. These insights are derived directly from the experimental data and claims within the patent documentation, providing clarity on the feasibility and performance of the technology. Understanding these details is essential for technical teams evaluating the integration of this intermediate into their drug development pipelines.
Q: What are the primary advantages of this nickel chelate over natural beta-carbolines?
A: The synthetic nickel chelate demonstrates significantly enhanced antitumor activity compared to the free ligand and avoids the low yields and complex purification associated with extracting beta-carbolines from natural plant sources.
Q: Is the synthesis process scalable for industrial production?
A: Yes, the patent describes both ambient pressure solution methods and high-pressure solvothermal methods, offering flexibility for scaling from laboratory grams to multi-kilogram commercial batches.
Q: What is the purity profile of the final crystalline product?
A: The process yields high-purity golden-yellow rod-shaped crystals, characterized by single-crystal X-ray diffraction, ensuring a well-defined structure suitable for pharmaceutical development.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1-Pyridine-6-Methoxy-β-Carboline Nickel Chelate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of having a dependable partner for complex pharmaceutical intermediates. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop discovery to clinical supply is seamless. We are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of Nickel(II) chelate meets the exacting standards required for oncology drug development. Our commitment to technical excellence means we can replicate the patented solvothermal and ambient pressure methods with precision, delivering the high-quality crystalline material necessary for your research and manufacturing needs.
We invite you to collaborate with us to optimize your supply chain for this innovative antitumor agent. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can support your project timelines and budget goals. Let us be your strategic partner in bringing next-generation metal-based therapeutics to market.
