Advanced Solid Acid Catalysis for Commercial Scale-Up of High-Purity Mexidole Intermediates
The pharmaceutical industry continuously seeks robust synthetic pathways for critical cardiovascular intermediates, and patent CN101891677B represents a significant technological leap in the production of 6-methyl-2-ethyl-3-hydroxypyridine, widely known as Mexidole. This antioxidant drug candidate has garnered substantial attention for its efficacy in treating myocardial ischemia and improving cerebral metabolism, yet its commercial availability has historically been constrained by inefficient manufacturing processes. The disclosed invention introduces a streamlined, two-step synthesis that replaces hazardous homogeneous catalysts with recoverable solid superacids and utilizes a specialized phase transfer catalyst system to drive the critical ring-expansion reaction. By shifting away from the traditional four-step chlorination sequences that plague legacy supply chains, this methodology offers a compelling value proposition for global procurement teams seeking reliable pharmaceutical intermediate supplier partnerships. The integration of green chemistry principles, specifically the recyclability of the solid acid catalyst and the minimization of toxic by-products, aligns perfectly with modern environmental compliance standards while simultaneously enhancing the economic viability of large-scale production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of 6-methyl-2-ethyl-3-hydroxypyridine has relied heavily on a cumbersome four-step sequence initiating from 2-picoline, a pathway fraught with significant operational and safety challenges that hinder cost reduction in API manufacturing. The initial chlorination step necessitates the use of elemental chlorine gas and large quantities of strong acids, creating severe corrosion risks and generating unavoidable ortho-position by-products that are chemically similar to the desired intermediate, making separation extremely difficult and energy-intensive. As illustrated in the reaction scheme below, the cumulative yield of this traditional route is abysmal, often hovering around a mere 10% when calculated from the starting material, due to successive losses in acetylation, reduction, and hydrolysis steps. Furthermore, the reliance on stoichiometric metal reducing agents in the third step introduces heavy metal contamination risks, requiring expensive downstream purification to meet stringent pharmacopeial limits. These inefficiencies not only inflate the cost of goods sold but also create supply chain bottlenecks, as the complex waste treatment required for chlorinated effluents can delay production schedules and increase regulatory scrutiny.
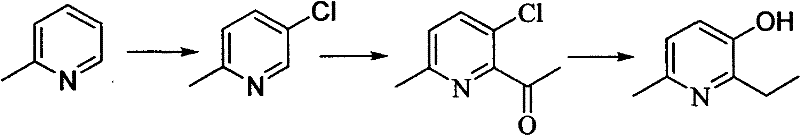
The Novel Approach
In stark contrast to the legacy picoline route, the novel approach detailed in the patent leverages 2-methylfuran as a more reactive and atom-economical starting material, fundamentally restructuring the synthesis into a concise two-step process that dramatically improves throughput. The first step involves the acylation of 2-methylfuran with propionic anhydride, but crucially, it employs a solid superacid catalyst such as sulfated zirconia instead of corrosive liquid acids, which preserves the integrity of the acid-sensitive furan ring and boosts the yield to over 80%. The second step utilizes a high-pressure ammonia-mediated ring expansion, enhanced by the addition of a quaternary ammonium resin acting as a three-phase transfer catalyst, which accelerates the reaction kinetics and suppresses the formation of pyrrole by-products. This strategic shift eliminates the need for hazardous chlorination entirely and reduces the number of isolation steps, thereby simplifying the plant operations and reducing the overall carbon footprint. For supply chain leaders, this translates to a more resilient production model where raw material volatility is minimized, and the risk of batch failure due to complex multi-step impurity profiles is significantly mitigated.
Mechanistic Insights into Solid Superacid-Catalyzed Acylation and Ring Expansion
The core innovation of this process lies in the sophisticated application of heterogeneous catalysis to overcome the inherent instability of the furan ring during electrophilic substitution. Traditional liquid acid catalysts tend to protonate the furan oxygen excessively, leading to ring opening and polymerization, which caps the yield at approximately 50%; however, solid superacids like SO4 2-/ZrO2 provide a controlled acidic environment on the surface of the catalyst particles. This surface confinement allows for the generation of the acylium ion from propionic anhydride while preventing the bulk solution from becoming overly acidic, thus maintaining the furan structure intact for the subsequent nucleophilic attack. The result is a highly selective acylation that produces 2-propionyl-5-methylfuran with a purity exceeding 98% by GC analysis, eliminating the need for complex chromatographic separations that would otherwise erode profit margins. Moreover, the solid nature of the catalyst allows for simple filtration and regeneration, turning a consumable reagent cost into a fixed capital asset that depreciates over hundreds of cycles, offering substantial long-term cost savings.
Following acylation, the ring expansion mechanism is critically dependent on the synergistic effect of ammonia and the phase transfer catalyst resin under high-temperature and high-pressure conditions. The reaction requires the opening of the furan ring and the incorporation of nitrogen to form the pyridine core, a transformation that is thermodynamically challenging and prone to side reactions like pyrrole formation if not carefully managed. The addition of the resin facilitates the transport of ammonium ions into the organic phase where the substrate resides, effectively increasing the local concentration of the nucleophile at the reaction interface and lowering the activation energy barrier. This acceleration allows the reaction to proceed to completion in a shorter timeframe, typically around 10 hours at 170°C and 3.0 MPa, compared to the prolonged heating required in non-catalyzed systems which often leads to thermal degradation. The visual representation of this streamlined pathway highlights the elegance of the design, where high yield and high purity are achieved through precise catalytic control rather than brute force reaction conditions.
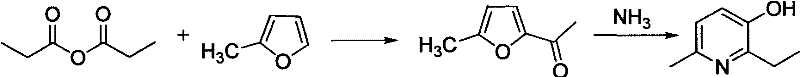
How to Synthesize 6-Methyl-2-Ethyl-3-Hydroxypyridine Efficiently
Implementing this advanced synthesis protocol requires careful attention to the preparation of the solid acid catalyst and the precise control of the autoclave conditions during the ring expansion phase to ensure consistent quality. The process begins with the activation of the solid superacid, followed by the controlled dropwise addition of 2-methylfuran to maintain the exotherm within safe limits, ensuring that the reaction temperature does not spike and degrade the product. Once the intermediate ketone is isolated, it is subjected to the ammonolysis step in a sealed pressure vessel, where the ratio of ammonia water to substrate and the loading of the phase transfer resin are critical parameters that dictate the final yield and impurity profile. Detailed standardized operating procedures regarding the specific temperature ramps, pressure holding times, and workup sequences involving hydrazine hydrate for color control are essential for reproducibility. For a comprehensive guide on the exact experimental parameters and safety protocols required for commercial implementation, please refer to the step-by-step synthesis guide provided below.
- Prepare 2-propionyl-5-methylfuran by reacting 2-methylfuran with propionic anhydride using a solid superacid catalyst like SO4 2-/ZrO2 at 120°C.
- Conduct ring expansion by reacting the intermediate with ammonia water and ammonium chloride in the presence of a phase transfer catalyst resin at 170°C and 3.0 MPa.
- Purify the crude product via recrystallization using isopropanol and decolorization with activated carbon to achieve high purity standards.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented methodology offers a transformative opportunity to optimize the cost structure and reliability of the antioxidant intermediate supply chain. By eliminating the multi-step chlorination sequence, manufacturers can drastically reduce the consumption of hazardous reagents and the associated costs of waste disposal and environmental compliance, leading to a leaner and more sustainable operation. The shift to a two-step process inherently reduces the lead time for high-purity pharmaceutical intermediates, as fewer unit operations mean less equipment occupancy time and faster turnaround from raw material intake to finished goods. Furthermore, the use of readily available starting materials like 2-methylfuran and propionic anhydride insulates the supply chain from the volatility often seen with specialized halogenated precursors, ensuring a steady flow of materials even during market disruptions. These operational efficiencies compound to create a robust supply foundation that can support the growing global demand for cardiovascular therapeutics without the typical bottlenecks associated with older, less efficient technologies.
- Cost Reduction in Manufacturing: The replacement of homogeneous liquid acids with recyclable solid superacids eliminates the need for neutralization and extensive washing steps, significantly reducing water consumption and wastewater treatment costs. Additionally, the higher overall yield of the two-step route compared to the 10% yield of the picoline route means that less raw material is required to produce the same amount of active ingredient, directly lowering the variable cost per kilogram. The ability to recover and reuse the phase transfer catalyst resin further contributes to cost optimization by minimizing the consumption of expensive organic additives. These cumulative savings allow for a more competitive pricing structure while maintaining healthy margins, making the final API more accessible to healthcare systems worldwide.
- Enhanced Supply Chain Reliability: Simplifying the synthesis from four complex steps to two robust reactions reduces the number of potential failure points in the manufacturing process, thereby increasing the overall reliability of supply. The avoidance of chlorine gas and heavy metal reductants removes significant safety hazards and regulatory hurdles, facilitating smoother logistics and storage of raw materials. With a shorter production cycle and higher throughput capacity, suppliers can respond more agilely to fluctuations in market demand, ensuring that downstream pharmaceutical partners receive their orders on time. This reliability is crucial for maintaining continuous production lines for life-saving cardiovascular medications, where any interruption in the supply of key intermediates could have serious consequences.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing standard reactor types such as autoclaves and distillation columns that are common in fine chemical plants, allowing for seamless transition from pilot scale to commercial production of complex pharmaceutical intermediates. The green chemistry attributes, including the absence of chlorinated waste and the recyclability of catalysts, ensure that the manufacturing process meets increasingly stringent global environmental regulations. This compliance reduces the risk of shutdowns due to environmental violations and enhances the corporate social responsibility profile of the manufacturing entity. Consequently, partners can confidently scale up production volumes knowing that the technology is both economically viable and environmentally sustainable for the long term.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis route, providing clarity on how it resolves historical pain points in Mexidole production. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation, offering a factual basis for decision-making. Understanding these nuances is vital for technical teams evaluating the feasibility of technology transfer and for commercial teams negotiating supply agreements based on improved quality and cost metrics. We encourage stakeholders to review these answers to gain a deeper appreciation of the process advantages.
Q: Why is solid superacid preferred over liquid acid for 2-methylfuran acylation?
A: Liquid acids often degrade the acid-sensitive furan ring, leading to low yields around 50%. Solid superacids like SO4 2-/ZrO2 provide strong acidity without dissolving into the reaction medium, preserving the furan structure and allowing catalyst recycling for greener processing.
Q: How does the new process control product color and oxidation?
A: The patent specifies the addition of hydrazine hydrate during the post-treatment phase. This reducing agent effectively prevents the oxidation of the hydroxypyridine structure at high temperatures, ensuring the final product meets strict color specifications without requiring excessive purification steps.
Q: What are the yield improvements compared to traditional picoline routes?
A: Traditional 4-step routes starting from 2-picoline suffer from cumulative low yields totaling only about 10%. This novel 2-step furan route achieves a significant yield increase, with individual step yields reaching 83% and 74%, drastically improving overall material efficiency.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 6-Methyl-2-Ethyl-3-Hydroxypyridine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the development of next-generation cardiovascular therapies, and we possess the technical expertise to bring this advanced solid-acid catalyzed route to full commercial fruition. Our R&D team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory success to industrial reality is smooth and efficient. We operate state-of-the-art facilities equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of 6-methyl-2-ethyl-3-hydroxypyridine meets the highest international standards for pharmaceutical use. Our commitment to quality is matched only by our dedication to process safety and environmental stewardship, making us an ideal partner for companies seeking a sustainable and reliable source of this vital antioxidant intermediate.
We invite global pharmaceutical partners to engage with our technical procurement team to discuss how this innovative synthesis method can be tailored to your specific volume requirements and quality targets. By collaborating with us, you can leverage our process optimization capabilities to achieve a Customized Cost-Saving Analysis that quantifies the economic benefits of switching to this greener, more efficient route. We encourage you to contact us today to request specific COA data and route feasibility assessments, allowing you to make informed decisions that will strengthen your supply chain and enhance your product competitiveness in the global marketplace.
