Revolutionizing Vitamin E Production: Advanced Gold-Catalyzed Chroman Synthesis for Commercial Scale
The pharmaceutical and nutritional industries are constantly seeking more efficient and sustainable pathways for the synthesis of critical bioactive molecules. Patent CN107001209B introduces a groundbreaking methodology for the formation of chromans, specifically targeting the synthesis of tocopherols and tocotrienols, which are vital forms of vitamin E. This technology leverages the unique catalytic properties of gold (I) complexes to facilitate intermolecular reactions between alkynes and dimethylfurans. By shifting away from traditional harsh chemical environments, this innovation offers a robust platform for producing high-purity nutritional ingredients with improved ecological profiles. The strategic implementation of this patent data suggests a significant leap forward in the commercial scale-up of complex pharmaceutical intermediates, addressing long-standing challenges in yield and selectivity that have plagued conventional manufacturing processes.
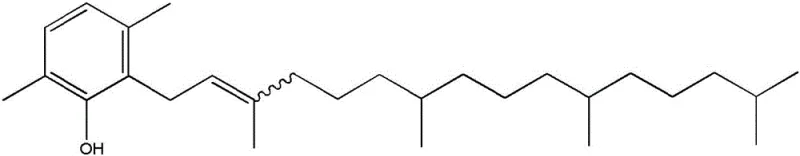
The demand for reliable nutritional ingredients supplier partners who can deliver consistent quality while adhering to green chemistry principles has never been higher. This patent outlines a process where 2,5-dimethylfuran serves as a key building block, providing an ecologically beneficial synthesis of beta-tocopherol. The ability to derive this furan from biomass sources such as cellulose underscores the sustainability of the route, aligning with global trends towards renewable feedstocks. For decision-makers in the supply chain, this represents a tangible opportunity to enhance supply chain reliability by diversifying raw material sources away from purely petrochemical dependencies. The technical depth of this invention ensures that the resulting chromans possess the necessary structural integrity for subsequent conversion into alpha-tocopherol and alpha-tocotrienol through methylation, thereby securing a versatile production pipeline.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of chroman derivatives like vitamin E has relied heavily on Friedel-Crafts alkylation processes that utilize substoichiometric Lewis acid salts. These traditional methods often necessitate corrosive reaction conditions that pose significant safety hazards and equipment maintenance challenges in a large-scale manufacturing setting. Furthermore, conventional routes frequently struggle with selectivity issues, leading to the formation of equimolar amounts of unwanted by-products such as alkenylfurans, which complicate downstream purification and reduce overall process efficiency. The reliance on harsh acids also generates substantial waste streams, increasing the environmental burden and disposal costs associated with production. Additionally, previous intermolecular gold-catalyzed cyclizations were often limited to aryl-substituted alkynes or very short-chain alkanyl acetylenes, restricting their applicability for synthesizing the long-chain hydrocarbyl substituents required for biologically active tocopherols.
The Novel Approach
The novel approach detailed in this patent overcomes these hurdles by employing gold (I) complexes to catalyze the reaction between 2,5-dimethylfuran and substituted alkynes containing long-chain unsaturated alkyl groups. This method eliminates the need for corrosive Lewis acid conditions, thereby creating a safer and more manageable operational environment for chemical plants. By enabling the use of biomass-derived 2,5-dimethylfuran, the process not only improves the ecological food print of the final product but also opens new avenues for cost reduction in vitamin E manufacturing through the utilization of renewable resources. The reaction proceeds with high yields and selectivity, ensuring that the desired phenol derivatives are formed with minimal by-product generation. This technological advancement allows for the synthesis of beta-tocopherol and beta-tocotrienol with superior efficiency, providing a very interesting route for the subsequent preparation of alpha-variants which are highly valued in the market.
Mechanistic Insights into Gold (I)-Catalyzed Cyclization
The core of this innovation lies in the precise mechanistic action of the gold (I) complex, which activates the alkyne moiety towards nucleophilic attack by the dimethylfuran. The gold catalyst, typically formulated as [Au(I)OL]AN where OL is an organic ligand and AN is a singly charged anion, facilitates the formation of the carbon-carbon bond under mild conditions, often at temperatures between 15 and 30°C. This mildness is crucial for preserving the integrity of sensitive functional groups within the long hydrocarbyl chains, which might otherwise degrade under the high temperatures required by traditional methods. The selection of specific ligands, such as phosphorus-containing ligands or imidazol-2-ylidene ligands, allows for fine-tuning of the catalyst's electronic and steric properties, optimizing the reaction for specific substrate variations. This level of control is essential for R&D directors focused on purity and杂质谱 (impurity profiles), as it minimizes the formation of structural isomers that are difficult to separate later in the process.
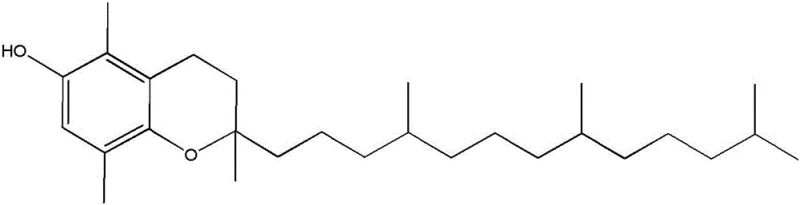
Furthermore, the process includes a sophisticated sequence of oxidation, reduction, and acid ring closure steps that ensure the final chroman structure is achieved with high fidelity. The oxidation step converts the initial phenol intermediate into a quinone, which is then reduced to a hydroquinone before undergoing acid-catalyzed ring closure. A critical advantage of this pathway is that various isomers of the hydroquinone precursors, which may differ in the position of double bonds within the side chain, all converge to yield the same final chroman product. This convergence means that rigorous separation of isomers at the intermediate stage is not necessary, resulting in a very high overall yield of the complete synthesis. This mechanistic robustness significantly reduces processing time and solvent consumption, directly contributing to cost reduction in nutritional ingredients manufacturing and enhancing the economic viability of the entire production line.
How to Synthesize Beta-Tocopherol Intermediates Efficiently
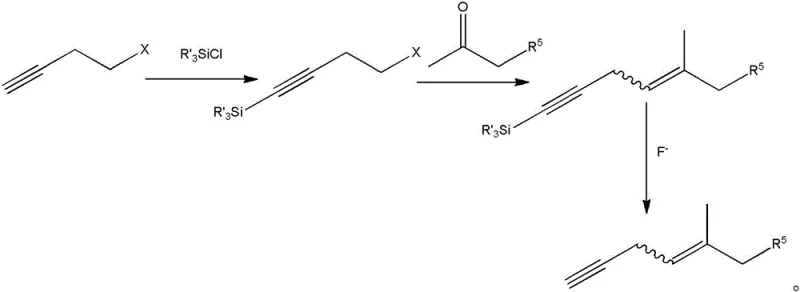
The synthesis of these high-value intermediates requires a systematic approach that integrates the preparation of the alkyne component with the gold-catalyzed coupling reaction. The process begins with the generation of the substituted alkyne, which can be achieved through several pathways including the reaction of halo-alkynes with ketones or the elimination of water from propargylic alcohols. Once the alkyne is prepared, it is reacted with commercially available 2,5-dimethylfuran in the presence of the gold catalyst in an inert solvent such as dichloromethane. The detailed standardized synthesis steps see the guide below.
- Prepare the substituted alkyne intermediate via silylation and coupling reactions.
- React 2,5-dimethylfuran with the alkyne using a gold (I) complex catalyst in an inert solvent.
- Perform oxidation, reduction, and acid ring closure to finalize the chroman structure.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented technology offers substantial strategic benefits that extend beyond mere technical feasibility. The elimination of corrosive Lewis acids and the ability to operate at near-ambient temperatures significantly reduce the wear and tear on reactor vessels and associated piping, leading to lower capital expenditure on maintenance and equipment replacement over time. The use of biomass-derived starting materials mitigates the risk associated with volatile petrochemical prices, providing a more stable cost structure for long-term production planning. Additionally, the high selectivity of the gold-catalyzed reaction minimizes the generation of hazardous waste, simplifying compliance with increasingly stringent environmental regulations and reducing the costs associated with waste treatment and disposal.
- Cost Reduction in Manufacturing: The process eliminates the need for expensive and hazardous Lewis acid catalysts, which traditionally require complex quenching and neutralization steps that generate large volumes of salt waste. By replacing these with catalytic amounts of gold complexes that can be optimized for recovery, the overall material costs are significantly reduced. Furthermore, the convergence of isomer pathways means that less material is lost during purification stages, maximizing the yield of the final active ingredient and lowering the cost per kilogram of production. The mild reaction conditions also translate to lower energy consumption for heating and cooling, contributing to substantial cost savings in utility expenses.
- Enhanced Supply Chain Reliability: Sourcing 2,5-dimethylfuran from biomass provides a diversified supply base that is less susceptible to the geopolitical and market fluctuations affecting fossil fuel-derived chemicals. This diversification enhances supply chain reliability by ensuring a consistent flow of raw materials even during periods of petrochemical scarcity. The robustness of the synthesis route, which tolerates various isomeric forms of the alkyne precursor, reduces the dependency on ultra-high purity starting materials that may have long lead times or limited availability. This flexibility allows procurement teams to negotiate better terms with suppliers and maintain inventory levels more effectively without compromising on the quality of the final product.
- Scalability and Environmental Compliance: The transition from batch to continuous flow processing is facilitated by the homogeneous nature of the gold catalysis and the absence of solid corrosive by-products, making the technology highly scalable for commercial production. The reduced environmental footprint, characterized by lower waste generation and the use of renewable feedstocks, aligns with corporate sustainability goals and facilitates easier permitting for new manufacturing facilities. This environmental compliance is increasingly becoming a prerequisite for doing business with major multinational corporations, thereby opening up new market opportunities for suppliers who can demonstrate green manufacturing capabilities.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this gold-catalyzed synthesis route. These answers are derived directly from the patent specifications and are intended to clarify the operational benefits and chemical principles underlying the technology. Understanding these details is crucial for stakeholders evaluating the feasibility of integrating this process into their existing manufacturing portfolios.
Q: What are the advantages of using gold (I) complexes over traditional Lewis acids?
A: Gold (I) complexes allow for milder reaction conditions, avoiding corrosive environments and reducing the need for substoichiometric Lewis acid salts, which simplifies downstream purification.
Q: How does this process impact the ecological footprint of vitamin E production?
A: The use of 2,5-dimethylfuran, which can be derived from biomass such as cellulose, provides an ecologically beneficial synthesis route compared to petrochemical-dependent methods.
Q: Is isomer separation required for the intermediates in this pathway?
A: No, the process is designed such that various isomers of the precursors yield the same final chroman product after acid ring closure, eliminating the need for complex isomer separation steps.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Beta-Tocopherol Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this gold-catalyzed synthesis route for the production of high-purity beta-tocopherol and related tocotrienols. As a leading CDMO expert, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that laboratory innovations are successfully translated into industrial reality. Our facilities are equipped with stringent purity specifications and rigorous QC labs capable of handling the precise analytical requirements of gold-catalyzed reactions, including trace metal analysis to ensure catalyst removal meets pharmaceutical standards. We are committed to delivering consistent quality and supply continuity for our global partners in the nutritional and pharmaceutical sectors.
We invite you to collaborate with us to optimize your supply chain and reduce manufacturing costs through the adoption of this advanced technology. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific production volumes and quality requirements. Please contact us to request specific COA data and route feasibility assessments that will demonstrate how we can support your goals for reducing lead time for high-purity tocopherols and enhancing your market competitiveness.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
