Advanced Liquid-Phase Convergent Synthesis of Pasireotide Intermediates for Commercial Scale-Up
Introduction to Next-Generation Pasireotide Manufacturing
The pharmaceutical landscape for treating Cushing's disease and hypercortisolism has been significantly advanced by the development of pasireotide, a multi-receptor targeted somatostatin analogue. As detailed in patent CN110627874B, the demand for high-purity pasireotide necessitates a departure from traditional manufacturing bottlenecks towards more robust, scalable methodologies. This patent introduces a groundbreaking preparation method that utilizes a liquid-phase convergent synthesis strategy, fundamentally altering the production economics and quality profile of this critical orphan drug. By segmenting the complex cyclic peptide into three distinct, stable polypeptide fragments—designated as Compounds I, II, and III—the process achieves superior control over stereochemistry and impurity profiles. This technical insight report analyzes the transformative potential of this liquid-phase approach for global supply chains.
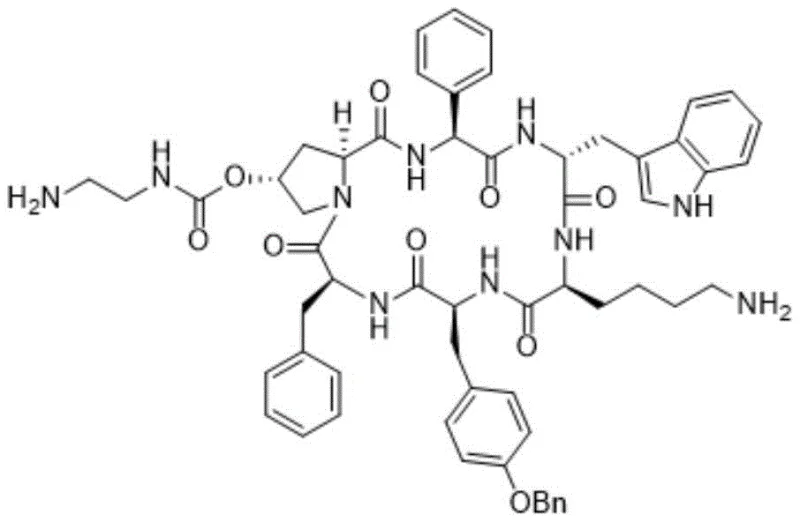
The structural complexity of pasireotide, featuring multiple chiral centers and a macrocyclic ring, presents formidable challenges for synthetic chemists. Traditional methods often struggle to maintain optical purity while achieving commercially viable yields. The methodology outlined in CN110627874B addresses these challenges head-on by employing a rational design of synthetic routes that prioritizes the stability of intermediates. This approach not only enhances the total yield but also simplifies the purification workflow, making it an ideal candidate for reliable pasireotide intermediate supplier partnerships aiming for industrial excellence.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the mainstream synthesis of pasireotide has relied heavily on solid-phase peptide synthesis (SPPS) strategies, such as the classic Fmoc/tBu protocol using SASRIN or Wang resins. While SPPS is effective for small-scale laboratory discovery, it suffers from inherent limitations when translated to commercial manufacturing. The primary drawback is the difficulty in purifying intermediates while they are attached to the solid support, leading to a cumulative buildup of deletion sequences and truncated byproducts that are hard to remove in the final cleavage step. Furthermore, solid-phase synthesis requires specialized, often proprietary equipment for resin handling and washing, which increases capital expenditure and limits flexibility. The low purity of the crude peptide obtained from SPPS necessitates extensive and costly downstream purification, often involving preparative HPLC, which drastically reduces overall throughput and increases the cost of goods sold (COGS).
The Novel Approach
In stark contrast, the novel liquid-phase convergent synthesis described in the patent offers a paradigm shift by decoupling the synthesis into manageable, high-purity fragments. This method involves the independent preparation of three key building blocks: Compound I (a tripeptide derivative), Compound II (a proline-ethylenediamine derivative), and Compound III (a dipeptide derivative). 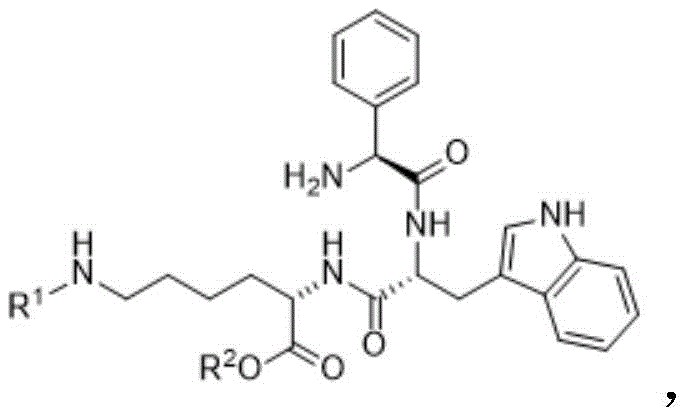
By synthesizing these fragments in solution, chemists can apply rigorous purification techniques such as crystallization and extraction at every stage, ensuring that only high-quality materials enter the final coupling steps. This modular approach significantly mitigates the risk of racemization, a common pitfall in peptide synthesis, by allowing for optimized conditions for each specific bond formation. The convergence of these fragments in the liquid phase facilitates easier scale-up in standard stainless steel reactors, eliminating the need for specialized solid-phase hardware and enabling a more flexible, cost-effective manufacturing process suitable for a reliable pasireotide intermediate supplier.
Mechanistic Insights into Liquid-Phase Convergent Cyclization
The core innovation of this synthesis lies in the strategic selection of orthogonal protecting groups and the sequence of condensation reactions. The process utilizes a combination of acid-labile (Boc), hydrogenation-labile (Cbz), and base-labile (Fmoc) protecting groups to manage reactivity. For instance, the synthesis of Compound I involves the condensation of protected D-tryptophan and L-lysine derivatives, where the careful arrangement of protecting groups allows for selective deprotection without affecting other sensitive moieties. This orthogonality is crucial for preventing side reactions and ensuring that the reactive amino and carboxyl groups are exposed only when needed for the next coupling step. The use of hydrogenation for removing Cbz groups is particularly advantageous as it proceeds with high conversion rates and generates benign byproducts, simplifying post-reaction workup.
The most critical step in the entire sequence is the intramolecular cyclization of the linear Intermediate II to form the macrocyclic Intermediate III. 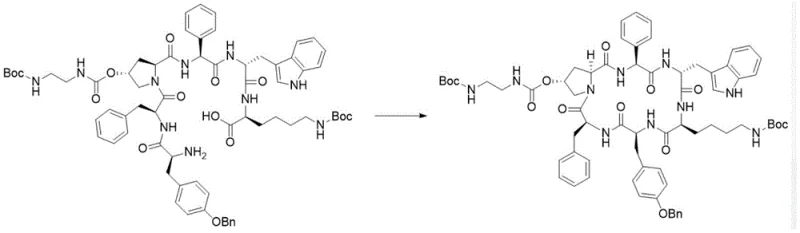
This cyclization is performed under activation by HATU, HOBt, and DIPEA, a combination known for its efficiency in forming peptide bonds with minimal epimerization. The patent specifies precise molar ratios (e.g., Intermediate II to HATU at 1:2.5 to 3.0) to drive the reaction to completion while suppressing intermolecular polymerization, which is a common competitor in macrocyclization reactions. The successful formation of the cyclic backbone with high yield and low racemization degree is the linchpin of this technology, directly translating to higher purity in the final API. This mechanistic precision ensures that the high-purity pasireotide produced meets the stringent regulatory requirements for orphan drugs.
How to Synthesize Pasireotide Efficiently
The synthesis of pasireotide via this liquid-phase route is a multi-step process that requires precise control over reaction conditions and stoichiometry. The general workflow involves the parallel preparation of the three key fragments, followed by their sequential assembly into a linear precursor and subsequent cyclization. Each step is designed to maximize yield and minimize impurity generation, leveraging the stability of the intermediates to allow for robust purification. For R&D teams looking to implement this technology, understanding the specific activation reagents and protecting group manipulations is essential for successful technology transfer. The detailed standardized synthesis steps for implementing this convergent strategy are provided in the guide below.
- Prepare Compound I by condensing protected D-tryptophan and L-lysine derivatives, followed by hydrogenation deprotection.
- Synthesize Compound II from protected hydroxyproline and ethylenediamine, ensuring orthogonal protection for subsequent coupling.
- Condense Compound I and II, then couple with Compound III (protected tyrosine-phenylalanine dipeptide) to form the linear precursor.
- Perform intramolecular cyclization of the linear intermediate using HATU/HOBt activation to form the cyclic peptide backbone.
- Execute final acidic hydrolysis to remove remaining protecting groups and yield high-purity pasireotide.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the shift from solid-phase to liquid-phase convergent synthesis represents a significant opportunity for cost reduction in peptide manufacturing. The elimination of expensive solid supports (resins) and the associated solvents required for resin swelling and washing drastically reduces raw material costs. Furthermore, the ability to purify intermediates via crystallization rather than solely relying on preparative chromatography lowers processing costs and increases batch throughput. The use of commercially available starting materials, such as protected amino acids and simple diamines, ensures a stable and diversified supply base, reducing the risk of raw material shortages. This route transforms the production of complex cyclic peptides from a boutique laboratory operation into a scalable industrial process.
- Cost Reduction in Manufacturing: The liquid-phase approach eliminates the capital expenditure associated with specialized solid-phase synthesizers and reduces the consumption of high-volume solvents typically used for resin washing. By enabling the use of standard chemical reactors, the process leverages existing infrastructure, thereby lowering depreciation costs per kilogram. Additionally, the higher purity of intermediates reduces the burden on final purification steps, leading to substantial savings in chromatography media and processing time. The overall efficiency gains result in a more competitive cost structure for the final active pharmaceutical ingredient.
- Enhanced Supply Chain Reliability: The convergent nature of this synthesis allows for the parallel production of Fragments I, II, and III. This modularity means that if one supply line faces a disruption, the others can continue, and inventory can be buffered at the fragment level rather than just the final product. The reliance on widely available, commodity-grade protecting group reagents (like Boc anhydride and Cbz chloride) further insulates the supply chain from volatility. This robustness ensures consistent delivery schedules and reduces lead times for high-purity peptide intermediates, a critical factor for maintaining continuous drug supply.
- Scalability and Environmental Compliance: Scaling liquid-phase reactions is a well-understood engineering challenge, unlike the complex fluid dynamics of scaling solid-phase reactors. This route facilitates a smoother transition from pilot plant to commercial scale (100 kgs to 100 MT), ensuring that process parameters remain consistent. Moreover, the reduction in solvent waste and the avoidance of hazardous resin disposal contribute to a greener manufacturing footprint. The process aligns with modern environmental, social, and governance (ESG) goals by minimizing waste generation and energy consumption associated with extensive purification cycles.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the liquid-phase synthesis of pasireotide. These answers are derived from the specific technical disclosures and beneficial effects outlined in the patent data, providing clarity on the feasibility and advantages of this manufacturing route. Understanding these details is vital for stakeholders evaluating the technology for potential licensing or procurement partnerships. The responses highlight the balance between chemical complexity and operational simplicity achieved by this method.
Q: Why is liquid-phase synthesis preferred over solid-phase for pasireotide production?
A: Liquid-phase synthesis allows for rigorous purification of intermediates at each step, resulting in significantly higher final purity compared to solid-phase methods which often trap impurities within the resin matrix. It also eliminates the need for expensive specialized solid-phase synthesizers.
Q: How does the convergent strategy reduce racemization risks?
A: By synthesizing shorter, stable fragments (Compounds I, II, and III) separately under optimized conditions, the exposure of sensitive chiral centers to harsh coupling conditions is minimized. The final cyclization is performed on a pre-assembled linear chain with carefully selected activating agents like HATU to preserve stereochemistry.
Q: What are the scalability advantages of this specific route?
A: The route utilizes standard liquid-phase reactors and common reagents (e.g., Boc, Cbz, Fmoc protecting groups) that are easily sourced at industrial scales. The convergent nature allows parallel production of fragments, drastically reducing overall lead time for large-scale batches.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pasireotide Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of robust synthetic routes for complex molecules like pasireotide. Our team of expert chemists has extensively evaluated the liquid-phase convergent synthesis strategy and is fully equipped to translate this patented technology into commercial reality. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this route are realized in actual manufacturing. Our state-of-the-art facilities are designed to handle the specific requirements of peptide synthesis, including dedicated zones for hydrogenation and strict temperature control for sensitive coupling steps. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of pasireotide intermediate meets the highest global standards.
We invite pharmaceutical partners to collaborate with us to optimize this synthesis for your specific supply chain needs. By leveraging our process development capabilities, we can provide a Customized Cost-Saving Analysis that quantifies the economic benefits of switching to this liquid-phase route. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments. Let us help you secure a sustainable, high-quality supply of pasireotide intermediates that drives your drug development forward.
