Advanced Glyburide Manufacturing: Enhancing Purity and Scalability for Global Pharma Supply Chains
Advanced Glyburide Manufacturing: Enhancing Purity and Scalability for Global Pharma Supply Chains
The global demand for effective antidiabetic medications continues to drive innovation in the synthesis of key active pharmaceutical ingredients (APIs) like Glyburide, also known as Glibenclamide. As a second-generation sulfonylurea, Glyburide plays a critical role in managing Type 2 diabetes by stimulating insulin release through the inhibition of ATP-sensitive potassium channels in pancreatic beta cells. However, the manufacturing of this complex molecule has historically faced challenges related to solubility limitations and impurity profiles. The technical breakthrough detailed in patent CN107879955B presents a transformative approach to Glyburide synthesis, addressing these long-standing issues through a refined five-step protocol. This method not only achieves exceptional purity levels exceeding 99.8% but also optimizes the reaction conditions to minimize waste and maximize yield. For pharmaceutical manufacturers and procurement specialists, understanding the nuances of this patented route is essential for securing a reliable glyburide supplier capable of meeting stringent regulatory standards.
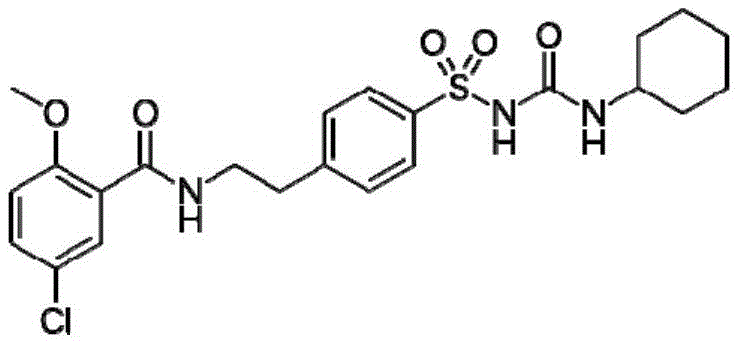
The structural complexity of Glyburide, characterized by its chloromethoxybenzamide and cyclohexylcarbamoyl groups linked via a sulfonamide bridge, necessitates precise control over each synthetic transformation. Conventional methods often struggle with the poor solubility of intermediates in aqueous and alcoholic media, leading to incomplete reactions and difficult purification processes. The methodology outlined in CN107879955B overcomes these barriers by introducing a strategic amino protection group and utilizing advanced phase transfer catalysis in the final coupling step. This ensures that the nucleophilic attack of the sulfonamide on the isocyanate is both efficient and selective. By adopting this advanced synthetic pathway, production facilities can significantly reduce the burden on downstream purification units, thereby lowering the overall cost of goods sold (COGS) while enhancing the consistency of the final drug product.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis routes for Glyburide have frequently been plagued by inefficiencies stemming from the physicochemical properties of the intermediates. Specifically, the limited solubility of Glyburide and its precursors in common solvents like water and ethanol creates significant bottlenecks during reaction workups and crystallization steps. In many legacy processes, the direct reaction of sulfonamides with isocyanates suffers from poor compatibility between the reactants, often requiring harsh conditions or excessive reagent loading to drive the reaction to completion. This not only increases raw material consumption but also generates a complex mixture of by-products and unreacted starting materials that are difficult to separate. Furthermore, the absence of a robust protecting group strategy in older methods leaves the amino functionality vulnerable to oxidation and other side reactions, resulting in a broader impurity spectrum that complicates regulatory approval and quality control.
The Novel Approach
In stark contrast, the novel approach described in patent CN107879955B introduces a sophisticated protection-deprotection strategy that fundamentally alters the reaction landscape. By initially protecting the amino group of 2-phenethylamine with a trifluoroacetyl group, the synthesis effectively shields the reactive center from unwanted oxidation during the subsequent sulfonation and sulfamidation steps. 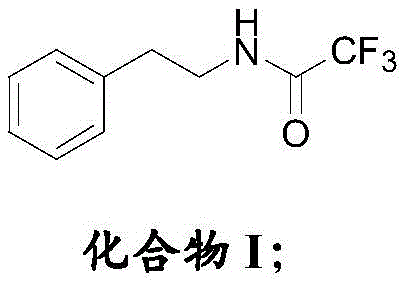 This protective measure ensures that the substrate remains stable throughout the harsh acidic conditions required for introducing the sulfonyl chloride moiety. Moreover, the process incorporates a meticulous recrystallization regimen using acid and base solutions, which systematically strips away impurities at each stage. The culmination of this strategy is a highly pure intermediate that is perfectly poised for the final coupling reaction, thereby eliminating the carryover of contaminants that typically degrade the quality of the final API.
This protective measure ensures that the substrate remains stable throughout the harsh acidic conditions required for introducing the sulfonyl chloride moiety. Moreover, the process incorporates a meticulous recrystallization regimen using acid and base solutions, which systematically strips away impurities at each stage. The culmination of this strategy is a highly pure intermediate that is perfectly poised for the final coupling reaction, thereby eliminating the carryover of contaminants that typically degrade the quality of the final API.
Mechanistic Insights into Phase Transfer Catalyzed Amidation
The cornerstone of this innovative synthesis lies in the final addition step, where the synergy between a strong base and a phase transfer catalyst (PTC) drives the formation of the urea linkage. In this critical transformation, Compound IV, which contains the sulfonamide nitrogen, must act as a nucleophile to attack cyclohexyl isocyanate. However, under standard conditions, the sulfonamide anion generated by deprotonation can be tightly ion-paired with the metal cation (such as potassium), reducing its reactivity and solubility in organic media. The introduction of 18-crown-6 ether resolves this issue by selectively complexing with the potassium ion. This sequestration creates a "naked" sulfonamide anion with significantly enhanced nucleophilicity, allowing it to attack the electrophilic carbon of the isocyanate with greater efficiency. 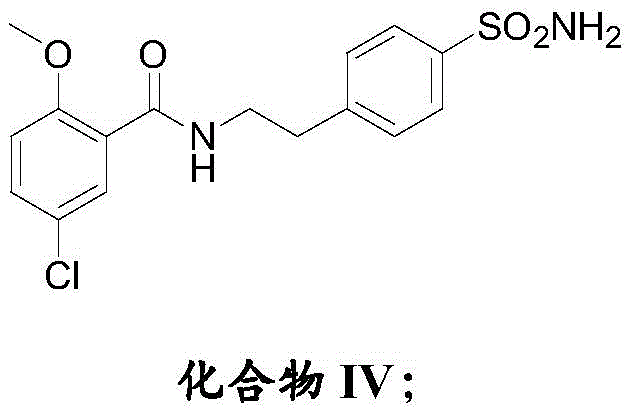 This mechanistic advantage ensures that the reaction proceeds to near-completion even at moderate temperatures, minimizing the formation of side products and maximizing the atom economy of the process.
This mechanistic advantage ensures that the reaction proceeds to near-completion even at moderate temperatures, minimizing the formation of side products and maximizing the atom economy of the process.
Furthermore, the choice of reagents in the amidation step highlights a commitment to impurity control. The activation of 5-chloro-2-methoxybenzoic acid using N,N'-carbonyldiimidazole (CDI) generates a highly reactive acyl imidazole intermediate in situ. This activated species reacts rapidly and cleanly with the amine of Compound III, avoiding the formation of stable salts or oligomers that are common with other coupling agents. The subsequent purification of Compound IV via ethanol recrystallization further refines the material, ensuring that only the desired isomer and connectivity proceed to the final step. This rigorous attention to mechanistic detail at the molecular level translates directly into macroscopic benefits for the manufacturing process, including reduced solvent usage, shorter cycle times, and a final product that consistently meets high-purity pharmaceutical intermediates specifications.
How to Synthesize Glyburide Efficiently
The implementation of this synthesis route requires precise adherence to the reaction parameters defined in the patent to ensure reproducibility and safety. The process begins with the careful control of temperature during the exothermic protection and sulfonation steps, followed by controlled pH adjustments during the recrystallization phases. The detailed standardized synthesis steps below outline the specific molar ratios, solvent choices, and thermal profiles necessary to achieve the reported yields of over 98% in early stages and high purity in the final product. Operators must pay particular attention to the stoichiometry of the crown ether and base in the final step, as this is the critical control point for maximizing conversion.
- Protect 2-phenethylamine with trifluoroacetic anhydride to form Compound I, preventing oxidation side reactions.
- Perform sulfonation with chlorosulfonic acid followed by sulfamidation with ammonia to generate the sulfonamide core (Compound III).
- Activate 5-chloro-2-methoxybenzoic acid with CDI and couple with Compound III to form the amide linkage in Compound IV.
- Execute the final addition of cyclohexyl isocyanate to Compound IV using potassium tert-butoxide and 18-crown-6 ether to maximize yield and purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented synthesis method offers compelling economic and operational advantages that extend beyond simple yield improvements. The primary value proposition lies in the drastic simplification of the purification workflow. By effectively managing impurities at the intermediate stage through the protection strategy and acid-base recrystallization, the need for extensive and costly chromatographic purification of the final API is eliminated. This reduction in downstream processing complexity translates directly into substantial cost savings in manufacturing, as it lowers solvent consumption, reduces waste disposal volumes, and shortens the overall production lead time. Additionally, the robustness of the reaction conditions means that the process is less sensitive to minor variations in raw material quality, enhancing supply chain reliability.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts and the use of readily available reagents like chlorosulfonic acid and CDI contribute to a leaner cost structure. The high efficiency of the phase transfer catalyzed step ensures that expensive reagents like cyclohexyl isocyanate are utilized with maximum atom economy, reducing raw material waste. Furthermore, the ability to achieve high purity through simple recrystallization rather than complex chromatography significantly lowers the operational expenditure associated with solvent recovery and waste treatment, driving down the total cost of ownership for the API.
- Enhanced Supply Chain Reliability: The synthetic route relies on commodity chemicals that are widely available in the global market, mitigating the risk of supply disruptions associated with exotic or proprietary reagents. The high yields reported in each step, particularly the 98.2% yield in the initial protection step, ensure that the throughput of the manufacturing plant is maximized. This efficiency allows for more predictable production scheduling and shorter lead times for high-purity pharmaceutical intermediates, enabling buyers to maintain lower safety stock levels while ensuring continuity of supply for their finished dosage forms.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated to work effectively with standard laboratory and pilot-scale equipment. The use of aqueous workups and recyclable solvents like THF and acetone aligns with modern green chemistry principles, reducing the environmental footprint of the manufacturing process. The minimization of hazardous by-products and the efficient use of reagents facilitate easier compliance with increasingly stringent environmental regulations, future-proofing the supply chain against regulatory changes and potential fines.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis of Glyburide using the methodology described in patent CN107879955B. These answers are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity on the process capabilities and quality outcomes. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this route into their existing manufacturing infrastructure.
Q: How does the novel synthesis method improve glyburide purity compared to conventional routes?
A: The patented method utilizes a trifluoroacetyl protecting group strategy combined with rigorous acid-base recrystallization steps. This effectively removes unreacted substrates and oxidation by-products, achieving HPLC purity levels exceeding 99.8%, which is superior to many commercially available grades.
Q: What is the role of 18-crown-6 ether in the final synthesis step?
A: In the final addition step, 18-crown-6 ether acts as a phase transfer catalyst that complexes with potassium ions from the base. This enhances the nucleophilicity of the sulfonamide group towards cyclohexyl isocyanate, ensuring the reaction proceeds to completion and significantly reducing residual impurities.
Q: Is this synthesis route scalable for industrial production?
A: Yes, the process is designed for scalability. It employs standard reagents like chlorosulfonic acid and CDI, and the workup procedures involve simple filtration and recrystallization. The high yields reported in each step (e.g., 98.2% in the protection step) indicate a robust process suitable for commercial scale-up from kilograms to metric tons.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Glyburide Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from a patented laboratory method to a commercial reality requires deep technical expertise and robust infrastructure. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the intricate details of the crown ether-catalyzed synthesis are perfectly translated to an industrial setting. We are committed to delivering high-purity Glyburide that adheres to stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our capability to manage complex multi-step syntheses involving sensitive intermediates positions us as a strategic partner for pharmaceutical companies seeking to secure their supply of critical antidiabetic APIs.
We invite you to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific supply chain needs. By requesting a Customized Cost-Saving Analysis, you can gain insights into the potential economic impact of switching to this high-efficiency manufacturing process. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your volume requirements, ensuring that your project moves forward with the highest degree of confidence and technical support.
