Advanced Manufacturing of 4-(4-Aminophenyl)-3-Morpholone for Global Antithrombotic Supply Chains
The pharmaceutical industry continuously seeks robust synthetic pathways for key antithrombotic intermediates, and the preparation of 4-(4-aminophenyl)-3-morpholone stands as a critical challenge in the value chain for Razaxaban production. Patent CN103804221A introduces a transformative methodology that redefines the efficiency and safety profile of this synthesis by strategically reordering reaction sequences to maximize selectivity and minimize hazardous waste. Unlike traditional approaches that struggle with regiocontrol and expensive catalyst requirements, this innovation leverages a unique acylation-first strategy followed by mild iron-catalyzed reduction, offering a compelling alternative for large-scale manufacturing. The structural integrity of the target molecule, essential for downstream biological activity, is preserved through gentle reaction conditions that avoid deep cooling or high-pressure environments. 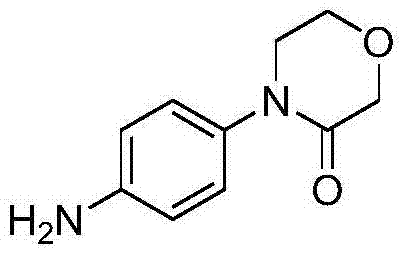 By addressing the fundamental limitations of prior art, this technology provides a scalable foundation for reliable pharmaceutical intermediate supplier networks aiming to optimize their production capabilities.
By addressing the fundamental limitations of prior art, this technology provides a scalable foundation for reliable pharmaceutical intermediate supplier networks aiming to optimize their production capabilities.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of morpholinone derivatives has been plagued by reliance on precious metal catalysts and harsh reaction conditions that impose significant burdens on both operational costs and environmental compliance. Conventional routes often necessitate the use of palladium-on-carbon for nitro group reduction, which not only escalates raw material expenses but also introduces complex purification steps to remove trace heavy metal residues from the final active pharmaceutical ingredient. Furthermore, traditional nitration strategies frequently suffer from poor regioselectivity, leading to the formation of difficult-to-separate ortho-isomers that drastically reduce overall process efficiency and yield. The requirement for high-pressure hydrogenation equipment adds another layer of capital expenditure and safety risk, making these methods less attractive for flexible, multi-purpose manufacturing facilities. 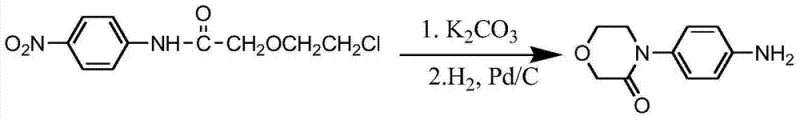 These cumulative inefficiencies create bottlenecks in the supply chain, resulting in longer lead times and higher volatility in pricing for downstream drug manufacturers who depend on consistent quality and availability.
These cumulative inefficiencies create bottlenecks in the supply chain, resulting in longer lead times and higher volatility in pricing for downstream drug manufacturers who depend on consistent quality and availability.
The Novel Approach
The innovative pathway disclosed in the patent data circumvents these historical obstacles by implementing a sequence that prioritizes mild conditions and earth-abundant catalysts to drive the transformation. By shifting the nitration step to occur after the formation of the morpholinone ring, the process inherently benefits from the directing effects of the existing substituents, thereby achieving superior para-selectivity without the need for additional protecting groups. The replacement of catalytic hydrogenation with a ferric chloride and hydrazine hydrate system eliminates the dependency on high-pressure infrastructure and precious metals, fundamentally altering the cost structure of the synthesis. This approach not only simplifies the operational workflow but also enhances the safety profile of the plant by removing high-pressure hydrogen from the process equation. The result is a streamlined manufacturing protocol that aligns perfectly with modern green chemistry principles while delivering the high purity standards required by global regulatory bodies.
Mechanistic Insights into FeCl3-Catalyzed Reduction and Regioselective Nitration
A deep understanding of the reaction mechanism reveals why this specific sequence offers such profound advantages over direct functionalization strategies. The initial acylation of aniline creates an amide bond that moderates the electron density of the aromatic ring, preventing the runaway oxidation and tar formation often seen during the direct nitration of free anilines. Subsequent cyclization forms the rigid morpholinone scaffold, which sterically and electronically directs the incoming nitro group almost exclusively to the para-position, ensuring a clean impurity profile that simplifies downstream crystallization. 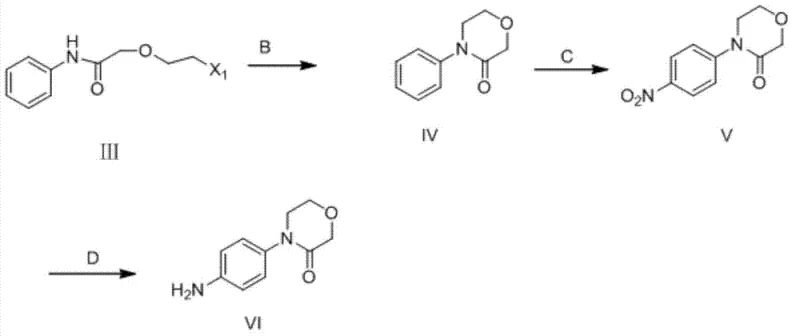 The final reduction step utilizes a single-electron transfer mechanism mediated by iron species, which selectively reduces the nitro group to the amine while leaving the sensitive lactam ring intact, a selectivity that is sometimes compromised by more aggressive reducing agents. This mechanistic precision ensures that the final product meets stringent specifications for isomeric purity, which is critical for the efficacy of the final anticoagulant drug.
The final reduction step utilizes a single-electron transfer mechanism mediated by iron species, which selectively reduces the nitro group to the amine while leaving the sensitive lactam ring intact, a selectivity that is sometimes compromised by more aggressive reducing agents. This mechanistic precision ensures that the final product meets stringent specifications for isomeric purity, which is critical for the efficacy of the final anticoagulant drug.
Controlling the impurity profile in this synthesis is achieved through the careful management of reaction stoichiometry and temperature gradients during the nitration phase. By maintaining the reaction temperature between 0°C and 5°C during the addition of nitric acid, the process minimizes the kinetic energy available for side reactions such as dinitration or oxidative degradation of the morpholine ring. The use of acetic anhydride as a solvent further buffers the acidity and scavenges water produced during the reaction, driving the equilibrium towards the desired mono-nitrated product. Post-reaction workup involves precise pH adjustments and recrystallization steps that leverage the solubility differences between the target para-isomer and any minor ortho-contaminants. This rigorous control strategy ensures that the chemical identity of the intermediate remains consistent across batches, providing the reliability that procurement teams demand for long-term supply agreements.
How to Synthesize 4-(4-Aminophenyl)-3-Morpholone Efficiently
Implementing this synthesis requires strict adherence to the sequential addition of reagents and temperature controls outlined in the patent examples to ensure optimal yield and safety. The process begins with the condensation of aniline and an acylating agent to form the key amide intermediate, followed by base-mediated cyclization to close the morpholine ring under heated conditions.
- Perform intramolecular cyclization of the amide intermediate using potassium carbonate in DMF at elevated temperatures to form the morpholinone core.
- Execute regioselective nitration using concentrated nitric acid in acetic anhydride at low temperatures (0-5°C) to ensure para-substitution.
- Conduct catalytic reduction using ferric chloride and hydrazine hydrate in alcohol solvents to convert the nitro group to amine without precious metals.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented methodology translates directly into enhanced margin protection and reduced supply risk through the elimination of critical bottleneck materials. By removing the requirement for palladium catalysts, manufacturers can insulate themselves from the extreme price volatility associated with precious metals markets, leading to more predictable and stable costing models for long-term contracts. The simplified equipment requirements, specifically the absence of high-pressure hydrogenation reactors, allow for production in a wider range of standard chemical facilities, thereby increasing the available capacity and reducing the likelihood of supply disruptions due to specialized maintenance needs. Furthermore, the use of inexpensive and readily available starting materials like aniline ensures that raw material sourcing remains robust even during periods of global chemical shortage. These factors combine to create a supply chain that is not only cost-efficient but also resilient against external market shocks.
- Cost Reduction in Manufacturing: The elimination of expensive palladium-on-carbon catalysts and high-pressure hydrogenation equipment results in substantial capital and operational expenditure savings for manufacturing partners. Without the need for specialized heavy metal removal steps to meet pharmaceutical purity standards, the downstream processing costs are drastically simplified, allowing for a leaner production budget. The use of ferric chloride as a catalyst represents a shift to low-cost, earth-abundant materials that do not fluctuate wildly in price, ensuring consistent unit economics over the lifecycle of the product. Additionally, the high selectivity of the reaction reduces the loss of valuable intermediates to byproduct formation, maximizing the output from every kilogram of raw material input.
- Enhanced Supply Chain Reliability: Sourcing aniline and basic acylating agents is significantly more reliable than securing specialized catalysts or high-pressure services, as these commodities are produced by a vast global network of chemical suppliers. The mild reaction conditions reduce the risk of unplanned shutdowns caused by equipment failure or safety incidents, ensuring a steady flow of material to downstream customers. This stability is crucial for pharmaceutical companies that require just-in-time delivery to maintain their own production schedules without the burden of holding excessive safety stock. The robustness of the process allows for flexible scaling, meaning supply can be ramped up quickly to meet unexpected surges in demand without compromising quality.
- Scalability and Environmental Compliance: The process generates significantly less hazardous waste compared to traditional methods, particularly by avoiding heavy metal contamination that requires complex and costly disposal procedures. The mild conditions and ambient pressure operations lower the energy consumption of the facility, contributing to a reduced carbon footprint and easier compliance with increasingly strict environmental regulations. Scalability is inherent in the design, as the reaction does not rely on mass-transfer limited gas-liquid hydrogenation steps that often fail when moving from pilot to commercial scale. This makes the technology ideal for commercial scale-up of complex pharmaceutical intermediates where consistency and regulatory approval are paramount.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this specific synthetic route for 4-(4-aminophenyl)-3-morpholone. These answers are derived directly from the technical disclosures and experimental data provided in the patent documentation to ensure accuracy and relevance for industry stakeholders. Understanding these details is essential for evaluating the feasibility of integrating this technology into existing manufacturing portfolios.
Q: Why is the nitration step performed after cyclization in this patent?
A: Performing nitration after acylation and cyclization avoids the need for protecting groups and significantly improves regioselectivity, minimizing ortho-substituted impurities common in direct nitration of aniline derivatives.
Q: Does this process require expensive palladium catalysts?
A: No, the patented method utilizes ferric chloride (FeCl3) and hydrazine hydrate for the reduction step, completely eliminating the need for costly palladium-on-carbon catalysts and high-pressure hydrogenation equipment.
Q: What is the overall yield potential of this synthetic route?
A: According to the experimental data in patent CN103804221A, the comprehensive yield ranges between 27% and 58%, with individual step yields reaching as high as 96% for acylation and 91% for reduction under optimized conditions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-(4-Aminophenyl)-3-Morpholone Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patent theory to commercial reality requires a partner with deep technical expertise and proven scale-up capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising yields observed in the lab are faithfully reproduced in our manufacturing plants. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 4-(4-aminophenyl)-3-morpholone meets the exacting standards required for antithrombotic drug synthesis. Our commitment to quality assurance means that you can rely on us for consistent supply without the risk of batch-to-batch variability that often plagues smaller producers.
We invite you to engage with our technical procurement team to discuss how this optimized synthetic route can benefit your specific supply chain requirements. By requesting a Customized Cost-Saving Analysis, you can gain clear visibility into the potential economic advantages of switching to this palladium-free methodology. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your project timelines. Let us demonstrate how our engineering excellence can drive efficiency and reliability in your production of high-purity pharmaceutical intermediates.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
