Advanced Manufacturing Strategy for High-Purity Posaconazole Intermediates and API
Advanced Manufacturing Strategy for High-Purity Posaconazole Intermediates and API
The global demand for second-generation triazole antifungals continues to surge, driven by the critical need for effective treatments against invasive fungal infections in immunocompromised patients. Within this competitive landscape, Patent CN102863431A emerges as a pivotal technological advancement, offering a robust and scalable preparation method for posaconazole that addresses significant safety and efficiency bottlenecks found in legacy synthetic routes. This proprietary methodology replaces hazardous catalytic hydrogenation steps with a safer, more controllable acid-mediated deprotection sequence, fundamentally altering the risk profile of manufacturing this complex molecule. By leveraging standard aprotic solvents and inorganic bases, the process achieves yields exceeding 60% while maintaining exceptional purity profiles suitable for pharmaceutical grade applications. For procurement leaders and technical directors, understanding the nuances of this patent is essential for securing a reliable pharmaceutical intermediates supplier capable of delivering consistent quality. The following analysis dissects the chemical innovation and commercial implications of this breakthrough synthesis strategy.
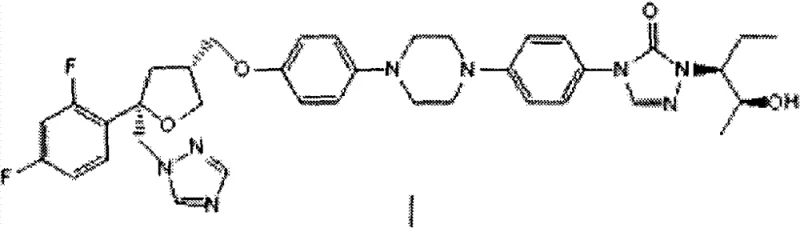
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of posaconazole has been plagued by operational complexities that hinder efficient cost reduction in API manufacturing. Prior art, such as the methodologies disclosed in patent WO2009141837, frequently relies on palladium-on-carbon (Pd/C) catalytic hydrogenation to remove protecting groups. This approach necessitates the use of high-pressure hydrogen gas and specialized reactor vessels, introducing severe safety hazards including explosion risks and the potential for catalyst leaching. Furthermore, many existing synthetic pathways depend heavily on column chromatography for the purification of intermediates, a technique that is notoriously difficult to scale beyond laboratory settings due to solvent consumption and throughput limitations. These traditional methods often result in fragmented production schedules, increased capital expenditure for safety infrastructure, and inconsistent batch-to-batch quality, creating significant vulnerabilities in the supply chain for high-purity posaconazole.
The Novel Approach
In stark contrast, the novel approach detailed in CN102863431A streamlines the synthetic pathway by utilizing a direct nucleophilic substitution followed by a mild acid-catalyzed deprotection. Instead of dangerous hydrogenation, the process employs hydrochloric acid or hydrogen bromide to cleave protecting groups under controlled thermal conditions, typically around 50°C. This shift eliminates the need for high-pressure equipment and precious metal catalysts, drastically simplifying the reactor requirements and reducing the overall environmental footprint of the production facility. The method also incorporates a highly efficient workup procedure involving aqueous extraction and pH adjustment, which allows for the isolation of intermediates through simple filtration rather than complex chromatographic separation. This transition from hazardous unit operations to standard chemical processing represents a paradigm shift in the commercial scale-up of complex antifungal agents, offering a safer and more economically viable route to market.
Mechanistic Insights into Nucleophilic Etherification and Acid Deprotection
The core of this synthetic strategy lies in the precise execution of a nucleophilic substitution reaction between a phenolic precursor and a halide intermediate within an aprotic solvent matrix. The process initiates by dissolving the phenolic compound (Formula II) in solvents such as dimethyl sulfoxide (DMSO) or dimethylformamide (DMF), which are chosen for their ability to stabilize the phenoxide anion generated upon the addition of an inorganic base like sodium hydroxide. This activation step is critical, as the resulting phenoxide ion acts as a potent nucleophile that attacks the electrophilic center of the tosylate or halide derivative (Formula III). The reaction is conducted at moderate temperatures ranging from 10°C to 35°C, a condition that balances reaction kinetics with the suppression of side reactions such as elimination or over-alkylation. The careful control of stoichiometry, with a molar ratio of reactants optimized between 1.0:1 and 1.2:1, ensures high conversion rates while minimizing the formation of difficult-to-remove impurities that could compromise the final drug substance quality.
Following the formation of the protected ether intermediate, the mechanism proceeds to a critical deprotection and cyclization phase mediated by strong mineral acids. The introduction of hydrochloric acid serves a dual purpose: it cleaves the acid-labile protecting groups, such as benzyl or methyl ethers, and facilitates the final structural rearrangement required to form the triazolone ring system characteristic of posaconazole. This acid-mediated step is performed at elevated temperatures, typically around 50°C, to overcome the activation energy barrier for bond cleavage without degrading the sensitive triazole and piperazine moieties present in the molecule. The subsequent neutralization with alkaline aqueous solutions allows for the selective precipitation or extraction of the free base form of the drug. Finally, the implementation of a binary solvent recrystallization system, specifically utilizing acetone and methanol, exploits the differential solubility of the target molecule versus its impurities, thereby achieving the high purity levels necessary for regulatory compliance and therapeutic efficacy.
How to Synthesize Posaconazole Efficiently
The practical implementation of this synthesis route requires strict adherence to the specified reaction parameters to ensure reproducibility and optimal yield. The process begins with the preparation of the reaction mixture in a non-protonic solvent, followed by the controlled addition of base and the electrophilic partner to generate the key intermediate. Once the intermediate is isolated and washed, it undergoes the acid treatment step which is the defining feature of this patent's novelty over hydrogenation-based routes. The final purification via recrystallization is not merely a cleanup step but a critical quality control point that defines the physical properties of the final API. For technical teams looking to adopt this methodology, the detailed standardized synthesis steps see the guide below provide the necessary framework for technology transfer and process validation.
- Dissolve the phenolic precursor (Compound II) in an aprotic solvent like DMSO or DMF, add aqueous inorganic base, and react with the halide intermediate (Compound III) at 10-35°C to form the protected ether intermediate.
- Treat the isolated intermediate with hydrochloric acid or hydrogen bromide at elevated temperatures (approx. 50°C) to effect deprotection and cyclization, yielding the crude posaconazole.
- Purify the crude product through a specialized recrystallization process using an acetone and methanol mixed solvent system to achieve high pharmaceutical purity.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, the adoption of this patent-covered methodology offers profound benefits that extend far beyond simple chemical yield improvements. By eliminating the requirement for high-pressure hydrogenation infrastructure, manufacturers can significantly reduce capital expenditure (CAPEX) associated with plant construction and maintenance, passing these savings down the value chain. The reliance on commodity chemicals such as sodium hydroxide, hydrochloric acid, and common organic solvents ensures that raw material availability remains stable even during global supply disruptions, thereby enhancing supply chain reliability for critical antifungal medications. Furthermore, the removal of column chromatography from the purification workflow dramatically increases throughput capacity, allowing facilities to produce larger batch sizes with shorter cycle times. This operational efficiency translates directly into improved lead times for high-purity posaconazole derivatives, enabling pharmaceutical partners to respond more agilely to market demands without compromising on quality or safety standards.
- Cost Reduction in Manufacturing: The elimination of expensive palladium catalysts and the associated recovery processes removes a significant variable cost component from the production budget. Additionally, the avoidance of specialized high-pressure reactors reduces energy consumption and maintenance costs, leading to substantial cost savings in the overall manufacturing economics. The simplified workup procedure also reduces solvent usage and waste disposal costs, contributing to a leaner and more profitable production model that aligns with modern green chemistry principles.
- Enhanced Supply Chain Reliability: By utilizing widely available reagents and avoiding rare earth metals or specialized gases, the risk of supply bottlenecks is minimized. The robustness of the reaction conditions, which tolerate slight variations in temperature and mixing without catastrophic failure, ensures consistent batch output. This stability is crucial for maintaining continuous supply agreements with major pharmaceutical companies who require guaranteed delivery schedules for their finished dosage forms.
- Scalability and Environmental Compliance: The process is inherently designed for scale, utilizing unit operations like filtration and crystallization that are standard in multi-ton chemical plants. The reduction in hazardous waste generation, particularly the absence of spent metal catalysts, simplifies environmental compliance and waste treatment protocols. This makes the technology highly attractive for manufacturing in regions with strict environmental regulations, ensuring long-term operational sustainability and license to operate.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for decision-making. Understanding these details helps stakeholders evaluate the feasibility of integrating this route into their existing manufacturing portfolios. The answers reflect the specific advantages of avoiding hydrogenation and utilizing acid-mediated deprotection strategies.
Q: How does this synthesis method improve safety compared to traditional hydrogenation routes?
A: Unlike prior art methods such as WO2009141837 which rely on hazardous Pd/C catalytic hydrogenation requiring specialized high-pressure equipment, this patent utilizes standard acid-mediated deprotection. This eliminates explosion risks associated with hydrogen gas and significantly lowers operational hazards in large-scale manufacturing environments.
Q: What purity levels can be achieved using the described recrystallization technique?
A: The patent data demonstrates that utilizing a specific acetone-to-methanol volume ratio (optimized around 1:4) during the final recrystallization step consistently yields posaconazole with HPLC purity exceeding 99.0%, effectively removing synthetic byproducts and meeting stringent regulatory standards for antifungal APIs.
Q: Is this process suitable for large-scale industrial production without column chromatography?
A: Yes, a key advantage of this methodology is the complete avoidance of column chromatography for purification. The process relies on precipitation and filtration followed by recrystallization, which are unit operations easily scalable from pilot plants to multi-ton commercial reactors, ensuring robust supply chain continuity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Posaconazole Supplier
As the pharmaceutical industry increasingly prioritizes supply chain resilience and process safety, the expertise to execute complex synthetic routes like the one described in CN102863431A becomes a vital asset. NINGBO INNO PHARMCHEM stands at the forefront of this evolution, leveraging extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production to deliver world-class antifungal intermediates. Our state-of-the-art facilities are equipped to handle the specific solvent systems and reaction conditions required for this synthesis, ensuring that every batch meets stringent purity specifications through our rigorous QC labs. We understand that consistency is key in API manufacturing, and our dedicated technical team works closely with clients to optimize every step of the process from raw material selection to final crystallization.
We invite global partners to engage with us for a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. By collaborating with our technical procurement team, you can access specific COA data and route feasibility assessments that demonstrate the tangible benefits of switching to this safer, more efficient manufacturing platform. Let us help you secure a stable supply of high-quality posaconazole intermediates that drive your drug development programs forward with confidence and economic efficiency.
