Scalable Synthesis of Azvudine Key Intermediate via D-Ribose Route
Scalable Synthesis of Azvudine Key Intermediate via D-Ribose Route
The global demand for antiviral therapeutics has intensified the search for robust, scalable synthetic routes for critical nucleoside analogs. Patent CN116143848A introduces a transformative preparation method for 1-acetoxy-2,3,5-tribenzoyloxy-1-beta-D-ribofuranose, a pivotal intermediate in the manufacturing of Azvudine. This technology addresses the longstanding bottlenecks in nucleoside chemistry by shifting the starting material from expensive adenosine derivatives to readily available D-ribose. For R&D directors and supply chain leaders, this represents a significant opportunity to optimize the cost structure and reliability of their API supply chains. The disclosed methodology not only enhances reaction yields but also simplifies the downstream processing requirements, thereby reducing the operational complexity typically associated with carbohydrate chemistry.
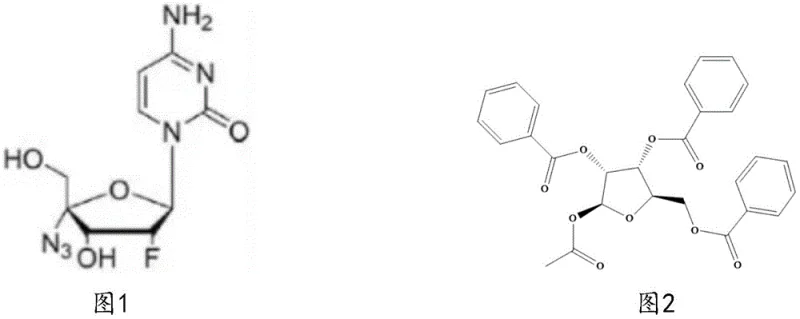
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of protected ribofuranose intermediates has relied heavily on adenosine as the foundational building block. As noted in prior art, such as the method disclosed by Li Jun et al., utilizing adenosine necessitates a multi-step sequence involving benzoylation and subsequent acylation. A critical drawback of this conventional approach is the relatively modest yield, often hovering around 70%, which creates substantial material loss at a stage where input costs are already high due to the price of adenosine. Furthermore, traditional pathways frequently require rigorous purification steps between reactions to remove unreacted starting materials and side products, leading to increased solvent consumption and extended production cycles. These inefficiencies compound the cost of goods sold (COGS) and introduce variability that can jeopardize supply continuity for high-volume API manufacturing.
The Novel Approach
The innovative strategy outlined in the patent data fundamentally reengineers the synthetic logic by employing D-ribose as the起始原料 (starting material). This shift leverages the abundant availability and lower cost profile of D-ribose compared to nucleoside starting materials. The new process integrates a streamlined three-step sequence: esterification, benzoylation, and acylation. Crucially, the protocol is designed to operate as a telescoped or semi-telescoped process where intermediate concentrates are carried forward without isolation or chromatographic purification. This "pot-to-pot" philosophy drastically reduces the time and resources spent on workup procedures. By optimizing reaction conditions, specifically temperature control and reagent stoichiometry, the novel approach achieves superior purity levels exceeding 99.0% directly from the crystallization or washing step, bypassing the need for resource-intensive purification technologies.
Mechanistic Insights into the Three-Step Cascade Synthesis
The chemical elegance of this process lies in its sequential functionalization of the ribose scaffold. The first stage involves the acid-catalyzed Fischer glycosidation of D-ribose in methanol. Under the influence of an acidic catalyst such as sulfuric or hydrochloric acid, the anomeric hydroxyl group of D-ribose reacts with methanol to form a methyl glycoside mixture, which serves as the substrate for subsequent protection. This step is critical for locking the ring structure and preparing the molecule for selective acylation. The reaction is conducted at ambient temperatures for 8 to 12 hours, ensuring complete conversion while minimizing degradation of the sensitive sugar backbone. The resulting concentrate contains the methyl riboside ready for the next transformation without the need for distillation or recrystallization.
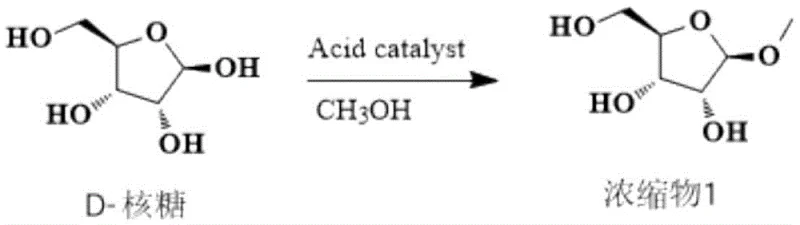
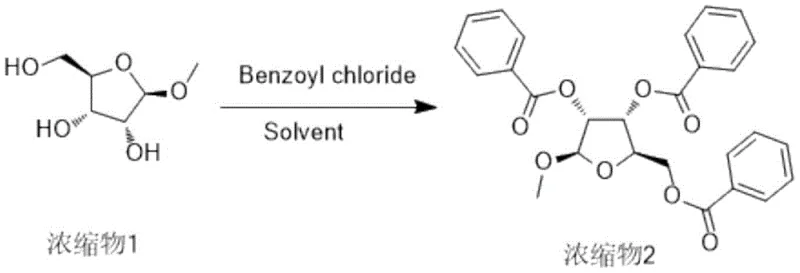
Following the initial glycosidation, the process moves to a comprehensive benzoylation phase. In this step, the free hydroxyl groups at the 2, 3, and 5 positions of the ribose ring are protected using benzoyl chloride. The reaction is facilitated by an acid-binding agent, such as sodium carbonate or triethylamine, and a nucleophilic catalyst like DMAP (4-dimethylaminopyridine). Maintaining the temperature between 10°C and 15°C during the addition of benzoyl chloride is paramount to controlling exothermicity and preventing side reactions. This careful thermal management ensures high regioselectivity and prevents the formation of over-acylated byproducts. The result is a fully protected tribenzoylated intermediate (Concentrate 2), which is isolated simply by concentration of the organic phase after aqueous workup, demonstrating the robustness of the protection strategy.
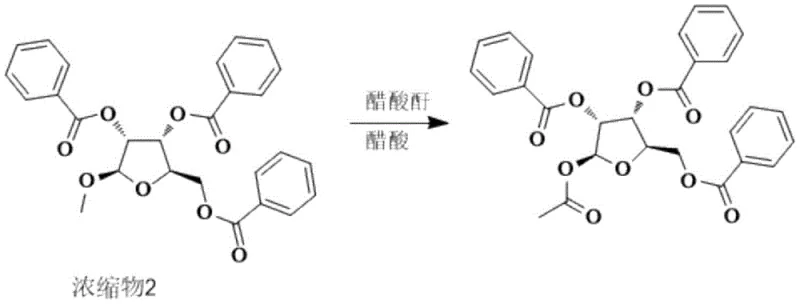
The final transformation involves the conversion of the methyl glycoside into the 1-acetoxy species, which is the defining feature of the target intermediate. This is achieved through an acylation reaction using acetic anhydride in glacial acetic acid. The reaction conditions are tightly controlled, with the addition of acetic anhydride occurring at low temperatures (5-10°C) to manage the reaction kinetics. Upon completion, the addition of water quenches the excess anhydride and induces precipitation of the product. The specific washing protocol, involving water washes to pH 6 followed by a solvent rinse, is mechanistically designed to remove acetic acid and benzoic acid byproducts while retaining the crystalline product. This step yields the final 1-acetoxy-2,3,5-tribenzoyloxy-1-beta-D-ribofuranose with exceptional purity, ready for the subsequent coupling reactions required to synthesize Azvudine.
How to Synthesize 1-Acetoxy-2,3,5-tribenzoyloxy-1-beta-D-ribofuranose Efficiently
Implementing this synthesis requires strict adherence to the thermal profiles and stoichiometric ratios defined in the patent to ensure reproducibility at scale. The process is designed to minimize unit operations, allowing for a continuous flow of material from one reactor to the next with minimal hold times. Operators must focus on the precise control of pH during the quenching phases and the efficiency of the washing steps to guarantee the removal of acidic impurities. The detailed standardized synthesis steps, including specific reagent grades and mixing speeds, are essential for maintaining the high-quality standards required for pharmaceutical intermediates.
- Perform acid-catalyzed esterification of D-ribose in methanol to form methyl riboside concentrate.
- Conduct benzoylation of the methyl riboside using benzoyl chloride and a base catalyst to protect hydroxyl groups.
- Execute final acylation with acetic anhydride in glacial acetic acid to yield the target 1-acetoxy intermediate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this D-ribose-based methodology offers profound strategic benefits beyond mere technical feasibility. The primary value driver is the substantial reduction in raw material costs. By replacing adenosine with D-ribose, manufacturers can access a commodity chemical market characterized by stable pricing and high availability, insulating the supply chain from the volatility often seen in specialized nucleoside markets. Furthermore, the elimination of intermediate purification steps translates directly into reduced solvent usage and lower waste disposal costs, contributing to a greener and more cost-effective manufacturing footprint. The simplified workflow also means shorter batch cycles, allowing facilities to increase throughput without requiring additional capital investment in new reactor trains.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the substitution of high-cost starting materials with inexpensive commodities and the removal of chromatographic purification. Traditional methods often incur significant expenses related to silica gel, eluents, and the labor associated with column chromatography. By achieving high purity through crystallization and washing alone, this method eliminates those variable costs entirely. Additionally, the high yield reported in the patent examples indicates a more efficient utilization of atoms, meaning less raw material is wasted as byproduct, further driving down the effective cost per kilogram of the active intermediate.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of ubiquitous reagents such as methanol, benzoyl chloride, and acetic anhydride. These chemicals are produced globally at massive scales, ensuring that procurement teams can source them from multiple vendors without risk of shortage. The robustness of the reaction conditions, which tolerate standard industrial equipment and do not require exotic catalysts or cryogenic temperatures, further reduces the risk of production delays. This reliability is crucial for maintaining the continuous supply of Azvudine, especially in a market environment where demand can fluctuate rapidly.
- Scalability and Environmental Compliance: From an environmental and scaling perspective, the process is inherently safer and cleaner. The avoidance of heavy metal catalysts or hazardous reagents simplifies the regulatory compliance burden regarding residual impurities in the final API. The wastewater generated is primarily acidic or saline, which is easier to treat compared to waste streams containing complex organic residues from chromatography. This ease of waste management facilitates smoother environmental permitting for scale-up, enabling manufacturers to ramp up production capacity from pilot plants to multi-ton commercial facilities with greater speed and confidence.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a clear picture of the technology's capabilities and limitations for potential adopters.
Q: What is the primary advantage of using D-ribose over adenosine for this synthesis?
A: Using D-ribose as the starting material significantly reduces raw material costs compared to adenosine. Additionally, the D-ribose route described in patent CN116143848A eliminates the need for intermediate purification steps, streamlining the workflow and improving overall throughput.
Q: How does this process ensure high purity without chromatography?
A: The process achieves high purity (99.0-99.5%) through precise temperature control during the acylation step and a specialized washing protocol. The crude product is washed with water to neutral pH and rinsed with solvents like methanol, removing impurities effectively without the need for expensive column chromatography.
Q: Is this synthesis method suitable for large-scale industrial production?
A: Yes, the method is specifically designed for industrial scalability. It utilizes common reagents like methanol, benzoyl chloride, and acetic anhydride, and avoids complex purification techniques, making it highly adaptable for commercial scale-up of complex pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1-Acetoxy-2,3,5-tribenzoyloxy-1-beta-D-ribofuranose Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of antiviral drugs depends on the reliability of the underlying intermediate supply. Our technical team has extensively analyzed the pathway described in CN116143848A and possesses the expertise to execute this D-ribose route with precision. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can move seamlessly from development to full-scale manufacturing. Our facilities are equipped with stringent purity specifications and rigorous QC labs capable of verifying the 99.0%+ purity benchmarks required for this critical intermediate, guaranteeing that every batch meets the exacting standards of the global pharmaceutical industry.
We invite you to collaborate with us to leverage this cost-effective technology for your Azvudine projects. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that optimize both your supply chain security and your bottom line.
