Revolutionizing Dye Intermediate Production: The One-Pot Synthesis of Sulfonated Para-Ester
The chemical manufacturing landscape for dye intermediates is undergoing a significant transformation driven by the urgent need for greener, more efficient synthesis routes. Patent CN102391163A, published in March 2012, introduces a groundbreaking preparation method for sulfonated para-ester, a critical precursor in the production of high-performance reactive dyes. This technology addresses the longstanding inefficiencies of conventional multi-step processes by integrating sulfonation, esterification, and hydrolysis reactions into a single, telescoped unit operation. By utilizing para-beta-hydroxyethyl sulfonyl acetanilide as the starting material and reacting it directly in 105% sulfuric acid under controlled thermal conditions, the process eliminates the need for intermediate isolation and drastically reduces the generation of hazardous waste acid. For R&D directors and procurement managers seeking reliable dye intermediate suppliers, this innovation represents a pivotal shift towards sustainable manufacturing that aligns with global environmental regulations while maintaining rigorous quality standards for complex organic synthesis.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the industrial production of sulfonated para-ester has relied on a fragmented and resource-intensive workflow that poses significant challenges for modern chemical plants. The conventional route typically begins with the chlorosulfonation of acetanilide, followed by a reduction reaction and condensation with ethylene oxide to generate para-beta-hydroxyethyl sulfonyl acetanilide. However, the critical bottleneck lies in the subsequent steps, where esterification, sulfonation, and hydrolysis are carried out as three entirely independent unit processes. This separation necessitates multiple transfer operations, extensive equipment usage, and, most detrimentally, the generation of substantial volumes of waste acid from at least two of these distinct reaction units. The environmental burden of treating this spent acid is immense, leading to high operational costs and severe contamination risks that fail to meet current energy-saving and emission-reduction mandates. Furthermore, the complexity of managing three separate reactors increases the potential for yield loss during material transfer and complicates the overall process control, making it difficult to achieve consistent purity levels required for high-end textile applications.
The Novel Approach
In stark contrast to the fragmented legacy methods, the novel approach detailed in the patent streamlines the entire downstream synthesis into a highly efficient one-pot reaction system. By charging para-beta-hydroxyethyl sulfonyl acetanilide directly into 105% sulfuric acid, the process leverages the dual functionality of the acid medium to catalyze both sulfonation and esterification simultaneously. The reaction mixture is carefully heated from an initial range of 20-40°C up to 120°C over a period of 3.5 hours, ensuring complete conversion before the temperature is lowered to 50°C for the controlled addition of water. This strategic introduction of water triggers the hydrolysis phase at 96±1°C over 10 hours, effectively cleaving the acetyl group to reveal the free amine without requiring a separate reactor vessel. This consolidation not only removes the waste acid production associated with two independent unit processes but also simplifies the operational workflow, reduces energy consumption for heating and cooling multiple vessels, and significantly enhances the overall atom economy of the synthesis. 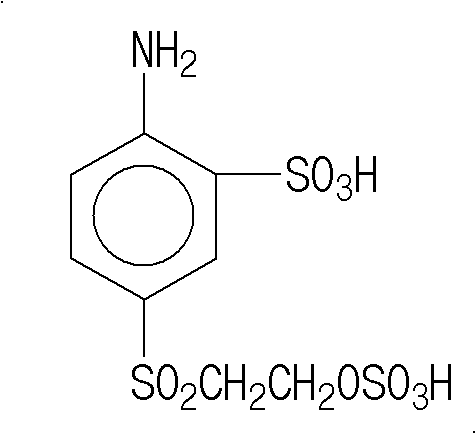
Mechanistic Insights into One-Pot Sulfonation and Hydrolysis
The core mechanistic advantage of this synthesis lies in the precise manipulation of reaction kinetics within a highly acidic medium to achieve sequential transformations without intermediate workups. When para-beta-hydroxyethyl sulfonyl acetanilide is introduced to 105% sulfuric acid, the oleum acts as a powerful dehydrating agent and sulfonating source, facilitating the formation of the sulfonic acid group on the aromatic ring while simultaneously promoting esterification dynamics. The gradual temperature ramp to 120°C provides the activation energy required for these electrophilic substitutions to proceed to completion, ensuring that the sulfone bridge remains intact while the aromatic system is functionalized. Following this, the controlled cooling to 50°C prior to water addition is critical; it prevents an exothermic runaway that could degrade the sensitive sulfate ester moiety. Once water is added, the medium transitions to favor hydrolysis, where the protonated acetyl group becomes susceptible to nucleophilic attack by water molecules, eventually yielding the primary amine functionality essential for the dye intermediate's reactivity. This delicate balance of acidity and temperature ensures that the final product retains its structural integrity, minimizing the formation of desulfonated byproducts or degraded sulfone species that often plague less optimized processes.
Impurity control in this one-pot system is inherently superior due to the elimination of physical transfer steps where product loss and contamination typically occur. In multi-step processes, each isolation and filtration stage introduces opportunities for mechanical loss and the entrapment of inorganic salts or residual solvents. By maintaining the reaction mass in a single vessel from the initial sulfonation through to the final hydrolysis, the process minimizes exposure to external contaminants and reduces the variance in reaction conditions that can lead to side reactions. The specific protocol of salting out the product after dilution with trash ice further enhances purity by leveraging solubility differences to precipitate the target sulfonated para-ester while leaving soluble inorganic impurities in the mother liquor. For R&D teams focused on impurity profiles, this method offers a robust pathway to achieving high-purity specifications consistently, as the reaction environment is tightly regulated by the stoichiometry of the sulfuric acid and the precise thermal profile, leaving little room for the stochastic variations that generate hard-to-remove trace impurities in batch-to-batch production.
How to Synthesize Sulfonated Para-Ester Efficiently
The synthesis of sulfonated para-ester via this patented method requires strict adherence to thermal profiles and reagent grades to ensure safety and yield. The process begins with the preparation of the precursor, para-beta-hydroxyethyl sulfonyl acetanilide, which is then subjected to the telescoped sulfonation-hydrolysis sequence in concentrated sulfuric acid. Operators must monitor the temperature closely during the exothermic addition of the precursor and the subsequent heating phases to prevent localized overheating. The detailed standardized synthetic steps, including specific agitation rates and quenching protocols, are outlined in the structured guide below to assist technical teams in replicating this high-efficiency route.
- Prepare para-beta-hydroxyethyl sulfonyl acetanilide via chlorosulfonation of acetanilide, reduction, and condensation with ethylene oxide.
- Add the precursor to 105% sulfuric acid at 20-40°C, ramp temperature to 120°C for 3.5 hours to effect sulfonation and esterification.
- Cool to 50°C, add water, heat to 96°C for 10 hours to hydrolyze the acetyl group, then salt out the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this one-pot synthesis technology translates into tangible operational improvements that extend far beyond simple chemical yield. By collapsing three distinct unit operations into a single reactor run, the process drastically reduces the capital expenditure required for equipment, as fewer vessels, pumps, and filtration units are needed to achieve the same output volume. This simplification of the plant footprint allows for greater flexibility in production scheduling and reduces the maintenance overhead associated with complex multi-reactor setups. Furthermore, the significant reduction in waste acid generation directly lowers the costs associated with environmental compliance and waste treatment, which are increasingly becoming major line items in the chemical manufacturing budget. The ability to produce high-purity dye intermediates with a smaller environmental footprint positions suppliers as preferred partners for multinational corporations striving to meet their own sustainability goals, thereby enhancing the long-term security of the supply relationship.
- Cost Reduction in Manufacturing: The elimination of two independent unit processes fundamentally alters the cost structure of producing sulfonated para-ester by removing the energy and labor costs associated with intermediate isolation and transfer. Without the need to filter, dry, and recharge materials between sulfonation, esterification, and hydrolysis steps, the consumption of utilities such as steam for heating and electricity for agitation is significantly decreased. Additionally, the reduction in waste acid volume means lower expenditures on neutralization agents and disposal fees, contributing to a leaner and more cost-effective manufacturing model. This qualitative improvement in process efficiency allows for competitive pricing strategies without compromising on the quality of the final dye intermediate, offering a distinct economic advantage in the global market.
- Enhanced Supply Chain Reliability: Simplifying the production route inherently increases the robustness of the supply chain by reducing the number of potential failure points. In a traditional multi-step process, a bottleneck or equipment failure in any one of the three unit operations can halt the entire production line; however, the consolidated one-pot approach minimizes these dependencies. The use of readily available raw materials like acetanilide and sulfuric acid, combined with a streamlined workflow, ensures that production can be scaled up or adjusted rapidly in response to market demand fluctuations. This agility is crucial for maintaining continuous supply to downstream dye manufacturers, preventing costly delays and ensuring that inventory levels remain stable even during periods of high volatility in the fine chemicals sector.
- Scalability and Environmental Compliance: The design of this process is intrinsically scalable, moving seamlessly from pilot batches to commercial tonnage production without the need for complex re-engineering of the reaction sequence. The single-vessel nature of the reaction simplifies scale-up calculations regarding heat transfer and mixing, reducing the risk of performance deviations when increasing batch sizes. From an environmental perspective, the drastic cut in waste acid generation aligns perfectly with tightening global regulations on industrial effluents, future-proofing the manufacturing site against stricter compliance standards. This proactive approach to environmental stewardship not only mitigates regulatory risk but also enhances the brand reputation of the supplier as a responsible and sustainable partner in the textile chemical value chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of sulfonated para-ester, derived directly from the specific innovations and data presented in the patent literature. These insights are intended to clarify the operational benefits and chemical mechanisms for stakeholders evaluating this technology for potential integration into their supply chains. Understanding these details is essential for making informed decisions about sourcing strategies and process optimization.
Q: How does the new one-pot process reduce environmental impact compared to traditional methods?
A: The traditional method requires three independent unit processes for esterification, sulfonation, and hydrolysis, generating significant amounts of waste acid from two separate units. The novel approach described in patent CN102391163A consolidates these reactions into a single unit operation, effectively eliminating the waste acid production associated with the separate units, thereby meeting stricter energy-saving and emission-reduction requirements.
Q: What are the critical temperature controls required for the hydrolysis step?
A: Precise thermal management is essential for high yield and purity. After the initial sulfonation and esterification at 120°C, the reaction mixture must be cooled to 50°C before the dropwise addition of water. Subsequently, the temperature is carefully raised to 96±1°C and maintained for 10 hours to ensure complete hydrolysis of the acetyl group without degrading the sensitive sulfonic acid groups.
Q: Why is 105% sulfuric acid used in this synthesis?
A: The use of 105% sulfuric acid (oleum) serves a dual purpose as both a solvent and a reagent. It provides the necessary dehydrating environment to drive the esterification and sulfonation reactions forward efficiently within the same vessel. Its high acidity also facilitates the subsequent hydrolysis step when water is introduced, allowing for a seamless transition between reaction phases without isolating intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Sulfonated Para-Ester Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthesis technologies to meet the evolving demands of the dye and fine chemical industries. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patents like CN102391163A are fully realized in practical, large-scale manufacturing. We are committed to delivering high-purity sulfonated para-ester that meets stringent purity specifications, supported by our rigorous QC labs which employ state-of-the-art analytical techniques to verify every batch. Our capability to implement complex one-pot reactions allows us to offer a product that is not only chemically superior but also produced with a significantly reduced environmental impact, aligning with the sustainability targets of our global clientele.
We invite procurement leaders and technical directors to engage with us for a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality standards. By partnering with our technical procurement team, you can access specific COA data and comprehensive route feasibility assessments that demonstrate how our optimized manufacturing processes can enhance your supply chain efficiency. Contact us today to discuss how our advanced production capabilities for sulfonated para-ester can support your next project, ensuring a reliable supply of high-quality intermediates that drive innovation in your final products.
