Advanced Manufacturing of Ramelteon Key Intermediate via Novel Cyanoacetic Acid Condensation
Introduction to the Novel Synthetic Route
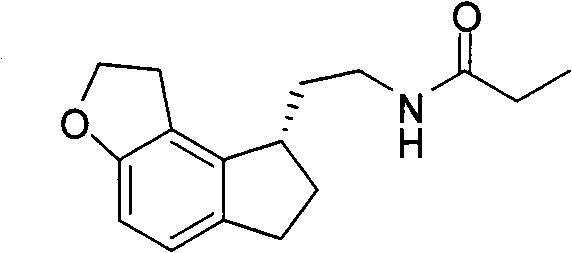
The pharmaceutical landscape for sleep disorder treatments has been significantly shaped by the development of melatonin receptor agonists, most notably Ramelteon. As detailed in patent CN102432571A, a groundbreaking preparation method has been established for the critical precursor, 2-(1,2,6,7-tetrahydro-8H-indeno[5,4-b]furan-8-yl)ethylamine hydrochloride. This intermediate serves as the foundational scaffold for the final active pharmaceutical ingredient (API), necessitating a synthesis pathway that balances high stereochemical integrity with economic viability. The traditional reliance on complex organophosphorus chemistry has long presented bottlenecks in terms of cost and safety, prompting the industry to seek more robust alternatives. This new methodology leverages a dehydration condensation strategy using cyanoacetic acid, a commodity chemical, to construct the carbon framework efficiently. By integrating a seamless decarboxylation and hydrogenation sequence, the process eliminates unnecessary isolation steps that often plague multi-step syntheses. For R&D teams evaluating supply chain resilience, this patent represents a pivotal shift towards greener, more cost-effective manufacturing paradigms.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of the ethylamine side chain on the indeno-furan core has relied heavily on Wittig-type or Horner-Wadsworth-Emmons reactions utilizing phosphorus-based reagents. Prior art, such as EP 0885210 and CN101654445, describes the condensation of the ketone precursor with diethyl cyanomethylphosphonate or cyanomethylene triphenyl phosphonium salts. These methodologies suffer from inherent drawbacks that escalate operational expenditures and environmental liabilities. The phosphine reagents employed are not only toxic and irritating to handle but also generate stoichiometric amounts of phosphine oxide waste, which is notoriously difficult to remove from the final product stream. Furthermore, these routes often exhibit mediocre yields and require rigorous purification protocols to meet the stringent impurity profiles demanded by regulatory bodies. The reliance on such hazardous materials complicates the supply chain, introducing risks associated with the procurement and storage of reactive phosphorus species. Consequently, manufacturers face inflated production costs and extended lead times due to the complexity of waste treatment and downstream processing.
The Novel Approach
In stark contrast, the innovative route disclosed in the patent data replaces these problematic phosphine reagents with cyanoacetic acid, a stable, inexpensive, and widely available organic acid. This transformation fundamentally alters the reaction mechanism to a Knoevenagel-type condensation followed by thermal decarboxylation, streamlining the construction of the unsaturated nitrile intermediate. The process operates under mild conditions, typically utilizing refluxing toluene to facilitate the azeotropic removal of water, driving the equilibrium towards product formation without the need for exotic dehydrating agents. Crucially, the subsequent hydrogenation and salification are performed in a telescoped manner, meaning the intermediate nitrile is reduced directly to the amine salt without isolation. This "one-pot" philosophy drastically reduces solvent consumption and labor hours while minimizing the exposure of sensitive intermediates to potential degradation. For a reliable pharmaceutical intermediate supplier, adopting this route translates to a more robust and predictable manufacturing cycle that aligns perfectly with modern principles of process intensification.
![Structure of the key intermediate 2-(1,2,6,7-tetrahydro-8H-indeno[5,4-b]furan-8-yl)ethylamine hydrochloride](/insights/img/ramelteon-intermediate-synthesis-pharma-supplier-20260307222222-02.png)
Mechanistic Insights into Cyanoacetic Acid Condensation and Hydrogenation
The core of this synthetic advancement lies in the efficient coupling of the indeno-furan ketone with cyanoacetic acid, catalyzed by weak organic bases such as piperidine or amines. The mechanism initiates with the deprotonation of the alpha-carbon of cyanoacetic acid, generating a nucleophilic enolate that attacks the carbonyl carbon of the ketone. Following the elimination of water, an alpha,beta-unsaturated nitrile is formed, which subsequently undergoes thermal decarboxylation to yield the desired chain-extended nitrile intermediate. The choice of catalyst is critical; the patent specifies a molar ratio of catalyst to ketone ranging from 0.05% to 2%, with piperidine proving highly effective in promoting the reaction while minimizing side products. The use of toluene as a solvent is particularly advantageous due to its ability to form an azeotrope with water, continuously removing the byproduct of condensation and pushing the reaction to completion. This mechanistic elegance ensures high conversion rates, with experimental data indicating yields exceeding 90% for the condensation step, a significant improvement over phosphine-mediated alternatives.
Following the formation of the nitrile, the process employs a dual-catalyst hydrogenation strategy to ensure complete reduction of both the cyano group and any olefinic bonds. The initial reduction utilizes Raney Nickel in an ammonia-saturated ethanol medium, which effectively reduces the nitrile to the primary amine while suppressing secondary amine formation. This is followed by a polishing step using Palladium on Carbon (Pd/C) to ensure full saturation and removal of trace impurities. The final step involves the direct introduction of hydrogen chloride gas into the reaction mixture, precipitating the amine as its hydrochloride salt. This direct salification is a masterstroke in impurity control, as it bypasses the isolation of the free base, which can be unstable or prone to oxidation. The resulting hydrochloride salt exhibits high purity and a sharp melting point, confirming the efficacy of this integrated approach in delivering a specification-ready intermediate for downstream chiral resolution and amidation.
How to Synthesize 2-(1,2,6,7-tetrahydro-8H-indeno[5,4-b]furan-8-yl)ethylamine Hydrochloride Efficiently
The execution of this synthesis requires precise control over reaction parameters to maximize yield and minimize impurity generation. The process begins with the charging of the ketone starting material and cyanoacetic acid into a reactor equipped with a Dean-Stark trap for water removal. The reaction is heated to reflux in toluene with a catalytic amount of piperidine until TLC analysis confirms the complete consumption of the starting ketone. Upon completion, the solvent is removed under reduced pressure, and the crude nitrile is subjected to hydrogenation without further purification. This telescoping of steps is the key to the process efficiency, saving significant time and resources. The hydrogenation is conducted in two stages: first with Raney Nickel at moderate temperatures (around 40°C) and pressure, followed by a second stage with Pd/C to ensure completeness. Finally, dry HCl gas is bubbled through the solution to precipitate the product, which is then filtered and dried to obtain the off-white solid intermediate. For detailed operational parameters and safety guidelines, please refer to the standardized protocol below.
- Condense 1,2,6,7-tetrahydro-8H-indeno[5,4-b]furan-8-one with cyanoacetic acid using an organic base catalyst in refluxing toluene.
- Perform thermal decarboxylation to obtain the nitrile intermediate without isolation.
- Execute sequential hydrogenation using Raney Nickel and Palladium Carbon, followed by salification with hydrogen chloride gas.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement perspective, the transition to this cyanoacetic acid-based route offers substantial benefits that extend far beyond simple reaction chemistry. The elimination of expensive and hazardous phosphine reagents results in a drastic simplification of the raw material supply chain, reducing dependency on specialized chemical vendors who often impose long lead times and volatile pricing structures. By switching to commodity-grade chemicals like cyanoacetic acid and toluene, manufacturers can achieve significant cost reduction in API manufacturing, insulating their production budgets from the fluctuations typical of the fine chemical market. Furthermore, the reduction in hazardous waste generation lowers the environmental compliance burden, translating into lower disposal costs and a smaller carbon footprint for the overall production facility. This alignment with green chemistry principles not only enhances corporate sustainability profiles but also mitigates regulatory risks associated with the handling of toxic substances.
- Cost Reduction in Manufacturing: The substitution of complex organophosphorus reagents with inexpensive cyanoacetic acid fundamentally alters the cost structure of the synthesis. Since the new route avoids the use of costly catalysts and eliminates the need for extensive purification steps to remove phosphine oxides, the overall cost of goods sold is significantly optimized. Additionally, the high yield of the condensation step means that less starting material is wasted, further enhancing the economic efficiency of the process. The ability to telescope the hydrogenation and salification steps also reduces solvent usage and energy consumption, contributing to a leaner and more profitable manufacturing operation.
- Enhanced Supply Chain Reliability: Sourcing strategies are greatly improved by the use of widely available bulk chemicals. Unlike specialized phosphine reagents which may have limited suppliers and potential supply disruptions, cyanoacetic acid and standard hydrogenation catalysts are produced by multiple global vendors. This diversification of the supply base ensures continuity of supply and provides procurement managers with greater leverage in negotiations. The robustness of the process also means that production schedules are less likely to be impacted by quality issues with raw materials, ensuring a steady flow of intermediates to support downstream API production.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, utilizing standard unit operations such as reflux, filtration, and hydrogenation that are common in multipurpose pharmaceutical plants. The absence of highly toxic reagents simplifies the engineering controls required for operator safety and environmental protection. Waste streams are easier to treat, and the overall E-factor of the process is improved, making it easier to obtain necessary environmental permits for expansion. This scalability ensures that the supply chain can respond flexibly to market demand surges without requiring massive capital investment in new specialized infrastructure.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this new preparation method is essential for stakeholders evaluating its adoption for commercial production. The following questions address common concerns regarding process safety, scalability, and product quality, drawing directly from the technical disclosures in the patent literature. These insights are intended to provide clarity on how this novel route compares to established methods and what specific advantages it offers for large-scale manufacturing. By addressing these key areas, we aim to facilitate informed decision-making for technical and commercial teams alike.
Q: What are the advantages of using cyanoacetic acid over phosphine reagents?
A: Cyanoacetic acid is significantly cheaper, safer, and easier to source than toxic organophosphorus reagents like diethyl cyanomethylphosphonate, reducing both raw material costs and hazardous waste disposal burdens.
Q: How does this process improve yield and purity?
A: The novel route achieves high yields (over 90% in the condensation step) and utilizes a one-pot hydrogenation strategy that minimizes intermediate handling, thereby reducing degradation and impurity formation.
Q: Is this method suitable for large-scale industrial production?
A: Yes, the process uses common solvents like toluene and standard hydrogenation equipment, avoiding exotic catalysts or extreme conditions, making it highly scalable for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ramelteon Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of complex pharmaceuticals like Ramelteon depends on a partner who can deliver both technical excellence and supply chain stability. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can transition smoothly from pilot scale to full industrial output. We are committed to maintaining stringent purity specifications and operating rigorous QC labs to guarantee that every batch of intermediate meets the highest international standards. Our facility is equipped to handle the specific requirements of this novel synthesis, including safe hydrogenation processes and efficient solvent recovery systems, positioning us as a strategic partner for your long-term growth.
We invite you to engage with our technical procurement team to discuss how this advanced manufacturing route can benefit your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic advantages of switching to this cyanoacetic acid-based method. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your volume requirements. Let us collaborate to optimize your supply chain and accelerate the delivery of high-quality sleep disorder medications to the market.
