Scalable Synthesis of 3',5'-Dichloro-2,2,2-trifluoroacetophenone via Mild Grignard Chemistry
The chemical industry is constantly seeking more efficient pathways for synthesizing critical intermediates, and the recent disclosure in patent CN112028752B offers a transformative approach to producing 3',5'-dichloro-2,2,2-trifluoroacetophenone. This compound serves as a pivotal building block in the synthesis of advanced agrochemicals and veterinary pharmaceuticals, where purity and cost-efficiency are paramount. The patented methodology replaces hazardous, energy-intensive cryogenic processes with a robust Grignard-based protocol that operates under significantly milder conditions. By utilizing readily available starting materials such as 1,3,5-trichlorobenzene or 3,5-dichlorobromobenzene, this innovation addresses long-standing supply chain bottlenecks. For R&D directors and procurement managers alike, this represents a shift towards sustainable manufacturing that does not compromise on the stringent quality standards required for downstream API synthesis.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of fluorinated acetophenones has relied heavily on organolithium chemistry, a method fraught with operational challenges and safety hazards. As detailed in prior art such as CN107353189A, traditional routes necessitate the use of strong alkalis like n-butyllithium and require deep cooling to temperatures as low as -78°C to control reactivity and prevent decomposition. These cryogenic conditions impose a massive energy burden on manufacturing facilities, requiring specialized refrigeration equipment that drives up capital expenditure and operational costs. Furthermore, the handling of pyrophoric organolithium reagents introduces significant safety risks, demanding rigorous containment protocols and increasing the complexity of waste management. The reliance on such extreme conditions often limits the scalability of the process, making it difficult to transition from laboratory benchtop synthesis to multi-ton commercial production without encountering yield fluctuations and impurity profile issues.
The Novel Approach
In stark contrast, the novel methodology disclosed in CN112028752B leverages the stability and versatility of Grignard reagents to achieve the same transformation under ambient or mildly heated conditions. By reacting magnesium with dichloro-haloarenes, the process generates the necessary nucleophile without the need for cryogenic stabilization. The subsequent reaction with trifluoroacetylating agents proceeds efficiently at temperatures ranging from 0°C to 100°C, with optimal results observed between 20°C and 60°C. This dramatic relaxation of thermal requirements eliminates the need for expensive cooling infrastructure and allows for the use of standard stainless-steel reactors. Moreover, the selection of trifluoroacetamides as acylating agents reduces corrosion risks compared to acidic halides, further extending equipment lifespan and reducing maintenance downtime. This approach not only simplifies the operational workflow but also enhances the overall economic viability of producing high-purity agrochemical intermediates.
Mechanistic Insights into Grignard-Mediated Trifluoroacetylation
The core of this synthetic breakthrough lies in the controlled formation and reaction of the Grignard intermediate. The process initiates with the oxidative insertion of magnesium into the carbon-halogen bond of the starting aryl halide, facilitated by solvents like tetrahydrofuran which stabilize the organomagnesium species. 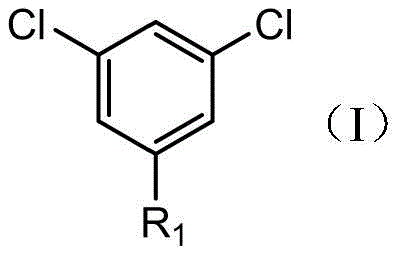 As illustrated in the structural representation of Compound I, the presence of electron-withdrawing chlorine atoms at the 3 and 5 positions influences the reactivity of the halogen at the 1-position, allowing for selective Grignard formation. The use of initiators such as 1,2-dibromoethane ensures reliable initiation of the magnesium surface, preventing induction periods that could lead to runaway reactions upon heating. This step is critical for maintaining a consistent concentration of the active nucleophile, which directly impacts the final yield and impurity profile of the product.
As illustrated in the structural representation of Compound I, the presence of electron-withdrawing chlorine atoms at the 3 and 5 positions influences the reactivity of the halogen at the 1-position, allowing for selective Grignard formation. The use of initiators such as 1,2-dibromoethane ensures reliable initiation of the magnesium surface, preventing induction periods that could lead to runaway reactions upon heating. This step is critical for maintaining a consistent concentration of the active nucleophile, which directly impacts the final yield and impurity profile of the product.
Following the generation of the Grignard reagent, the system undergoes a nucleophilic acyl substitution with Compound II. 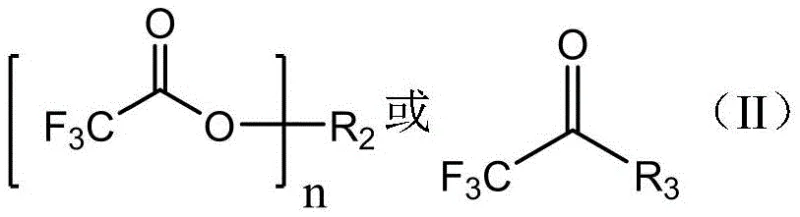 The patent highlights various forms of Compound II, including trifluoroacetamides and salts, which react with the Grignard species to form a tetrahedral intermediate. Upon acidic workup, typically using dilute hydrochloric acid, this intermediate collapses to release the target ketone and soluble amine or salt byproducts. A key mechanistic advantage is the solubility profile of these byproducts; the generated inorganic salts and organic amines exhibit high water solubility, allowing for their facile removal during the aqueous extraction phase. This inherent separation efficiency minimizes the need for complex chromatographic purification, ensuring that the final product meets high-purity specifications (>99%) suitable for sensitive pharmaceutical applications.
The patent highlights various forms of Compound II, including trifluoroacetamides and salts, which react with the Grignard species to form a tetrahedral intermediate. Upon acidic workup, typically using dilute hydrochloric acid, this intermediate collapses to release the target ketone and soluble amine or salt byproducts. A key mechanistic advantage is the solubility profile of these byproducts; the generated inorganic salts and organic amines exhibit high water solubility, allowing for their facile removal during the aqueous extraction phase. This inherent separation efficiency minimizes the need for complex chromatographic purification, ensuring that the final product meets high-purity specifications (>99%) suitable for sensitive pharmaceutical applications.
How to Synthesize 3',5'-Dichloro-2,2,2-trifluoroacetophenone Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for replicating these high-yield results in a production environment. The process is designed to be robust against minor variations in temperature and stoichiometry, making it ideal for technology transfer. Detailed below is the standardized procedure derived from the preferred embodiments, which balances reaction kinetics with safety considerations to maximize throughput.
- Disperse magnesium metal in a solvent such as tetrahydrofuran and react with 1,3,5-trichlorobenzene or 3,5-dichlorobromobenzene at 20-60°C to form the Grignard reagent.
- Add a trifluoroacetylating agent (e.g., trifluoroacetyldimethylamine) to the Grignard mixture at 10-30°C to effect nucleophilic addition.
- Quench the reaction with dilute hydrochloric acid, separate the organic phase, and purify via distillation to obtain the target ketone.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this Grignard-based synthesis offers tangible strategic benefits beyond mere technical feasibility. The shift away from cryogenic conditions and pyrophoric reagents translates directly into reduced operational expenditures and enhanced supply security. By utilizing commodity chemicals like magnesium and trichlorobenzene, manufacturers can decouple their production from the volatile pricing and availability issues often associated with specialized organolithium reagents. This stability is crucial for maintaining consistent delivery schedules to downstream clients in the agrochemical sector.
- Cost Reduction in Manufacturing: The elimination of deep cooling requirements (-78°C) removes a significant energy cost center from the production budget. Traditional lithiation processes consume vast amounts of electricity for refrigeration, whereas this new method operates near room temperature, drastically lowering utility bills. Additionally, the replacement of expensive n-butyllithium with inexpensive magnesium metal reduces raw material costs substantially. The use of less corrosive trifluoroacetamides instead of acid halides also lowers equipment maintenance and replacement costs, contributing to a leaner manufacturing overhead.
- Enhanced Supply Chain Reliability: The raw materials for this process, specifically 1,3,5-trichlorobenzene and magnesium, are widely produced commodity chemicals with stable global supply chains. This contrasts sharply with the supply constraints often faced for high-purity organolithium solutions. By diversifying the input material base to include common industrial feedstocks, manufacturers can mitigate the risk of production stoppages due to raw material shortages. Furthermore, the simplified workup procedure reduces the dependency on specialized purification resins or solvents, streamlining the logistics of consumable procurement.
- Scalability and Environmental Compliance: The process has been validated for scale-up, as evidenced by the successful amplification experiments in the patent data, ensuring that lab-scale efficiencies translate to commercial tonnage. The generation of water-soluble byproducts simplifies wastewater treatment, as inorganic salts can be easily managed compared to complex organic waste streams. This aligns with increasingly stringent environmental regulations, reducing the compliance burden and potential fines associated with hazardous waste disposal. The mild reaction conditions also improve workplace safety, lowering insurance premiums and liability risks.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on process optimization and quality control.
Q: What are the advantages of this Grignard method over traditional lithiation?
A: Unlike traditional methods requiring n-butyllithium and cryogenic temperatures (-78°C), this Grignard-based process operates at mild temperatures (20-60°C), significantly reducing energy costs and safety risks associated with pyrophoric reagents.
Q: Which trifluoroacetylating agents provide the best yield?
A: Experimental data indicates that trifluoroacetamides, specifically trifluoroacetyldimethylamine, provide superior yields (over 86%) and purity compared to trifluoroacetyl halides or salts, while being less corrosive to equipment.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the patent demonstrates successful scale-up in Examples 44-47, showing consistent yield and purity when increasing reactant quantities, confirming its viability for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3',5'-Dichloro-2,2,2-trifluoroacetophenone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of next-generation agrochemicals and pharmaceuticals. Our technical team has extensively analyzed the Grignard-based pathway described in CN112028752B and possesses the expertise to implement this technology at scale. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this mild synthesis are fully realized in our manufacturing facilities. Our rigorous QC labs enforce stringent purity specifications, guaranteeing that every batch of 3',5'-dichloro-2,2,2-trifluoroacetophenone meets the exacting standards required for downstream coupling reactions.
We invite potential partners to engage with our technical procurement team to discuss how this optimized synthesis can benefit your specific supply chain. By leveraging our capabilities, you can secure a Customized Cost-Saving Analysis tailored to your volume requirements. We encourage you to request specific COA data and route feasibility assessments to verify how our implementation of this patent technology can enhance your product portfolio's competitiveness and reliability.
