Advanced Manufacturing of High-Purity DIDNTB for Diagnostic Applications
Advanced Manufacturing of High-Purity DIDNTB for Diagnostic Applications
The landscape of diagnostic reagent manufacturing is undergoing a significant transformation driven by the demand for higher purity intermediates that ensure clinical accuracy. Patent CN116375680A introduces a groundbreaking preparation method for DIDNTB (5', 5'-dinitro-3', 3'-diiodo-3,4,5, 6-tetrabromophenol sulfophthalein), a critical protein error indicator used extensively in early kidney disease diagnosis. This technology addresses the longstanding industry challenge of low product purity, which historically hovered around 85 percent, directly impacting the stability and sensitivity of diagnostic kits. By implementing a refined sequence of iodination, nitration, and a unique dual-stage purification involving salification and acid conversion, this process elevates the final purity to an exceptional 97.0 percent range. For R&D directors and procurement specialists, this represents a pivotal shift towards more reliable supply chains for high-purity pharmaceutical intermediates.
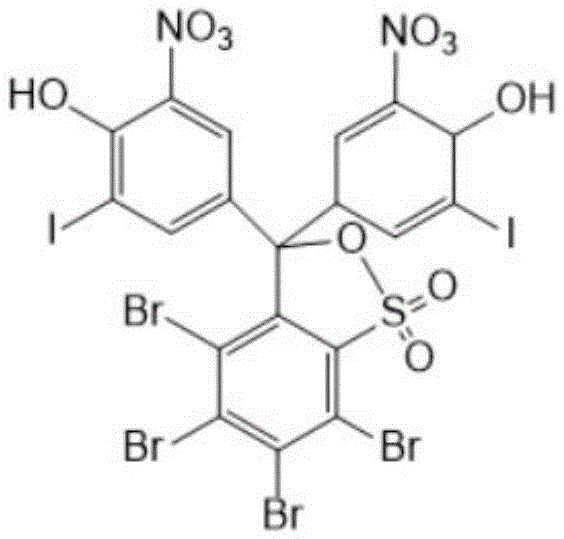
The strategic value of this patent lies not just in the chemical outcome but in its operational robustness. Traditional synthesis routes often struggled with difficult purification profiles and low yields, creating bottlenecks in the commercial scale-up of complex diagnostic intermediates. The new methodology leverages specific reagent combinations, such as iodine monochloride paired with acetic anhydride, to drive reactions to completion under mild conditions. This approach minimizes side reactions that typically generate hard-to-remove impurities. Furthermore, the integration of a salification step using alkali metal salts followed by a trans-acid purification creates a powerful impurity rejection mechanism. This ensures that the final DIDNTB pure product meets the stringent quality specifications required for sensitive clinical applications, thereby reducing the risk of batch failures and reagent instability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art synthesis technologies for DIDNTB were plagued by inherent inefficiencies that compromised both economic viability and product performance. The most critical defect was the inability to achieve high purity, with conventional methods typically yielding products with purity levels less than or equal to 85 percent. In the context of diagnostic reagents, such impurity profiles are unacceptable as they directly influence the stability, sensitivity, and accuracy of the final test kits. Additionally, the purification processes in older methods were notoriously difficult, often requiring complex chromatographic separations or multiple recrystallizations that drastically reduced overall yield. These inefficiencies made mass production challenging, limiting the availability of high-quality DIDNTB and constraining the further application of this vital protein error indicator in advanced medical diagnostics.
The Novel Approach
The innovative strategy outlined in the patent fundamentally restructures the synthesis pathway to prioritize both yield and purity through controlled reaction kinetics and selective crystallization. Instead of a direct one-pot synthesis, the process separates the iodination and nitration steps, allowing for precise control over substitution patterns on the phenol sulfophthalein backbone. Crucially, the introduction of a salification purification step transforms the crude product into an alkali metal salt, which exhibits different solubility characteristics than the impurities, allowing them to remain in the mother liquor. This is followed by a trans-acid purification where the salt is converted back to the acid form using organic carboxylic acids. This dual-purification logic effectively strips away macromolecular impurities and unreacted starting materials, resulting in a total yield reaching 80 percent with purity exceeding 97.0 percent.
Mechanistic Insights into Iodination-Nitration and Salification Purification
The core of this synthesis relies on a highly controlled electrophilic aromatic substitution sequence. The iodination step utilizes iodine monochloride in the presence of acetic anhydride, which acts as both a solvent and a dehydrating agent to activate the iodinating species. This specific reagent system is superior to elemental iodine alone because it generates a more reactive electrophile that can efficiently substitute onto the sterically hindered tetrabromophenol ring. Following this, the nitration is performed using fuming nitric acid and acetic anhydride. The acetic anhydride reacts with nitric acid to form acetyl nitrate, a potent nitrating agent that operates effectively at lower temperatures, thereby preventing the oxidative degradation of the sensitive bromine and iodine substituents. This careful management of reaction energetics is what allows the process to achieve a crude purity of roughly 90 percent before any purification even begins.
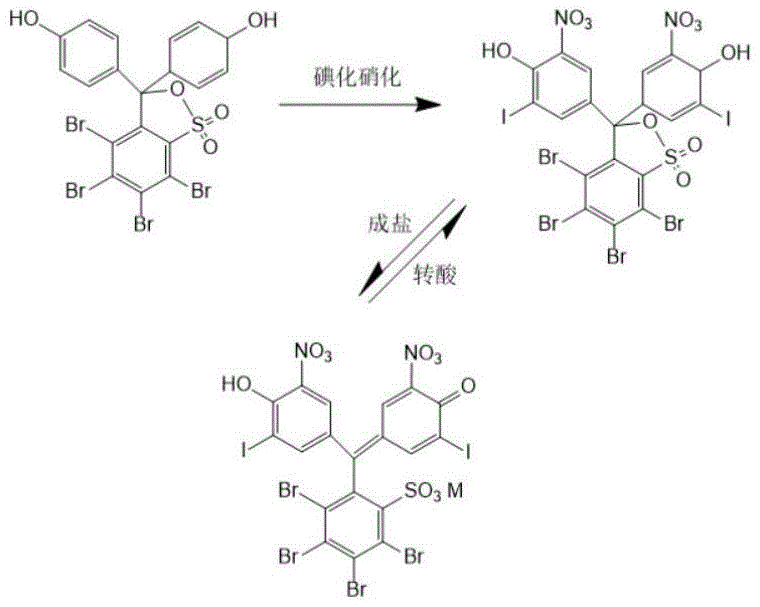
The purification mechanism is equally sophisticated, relying on the physicochemical properties of the sulfonphthalein structure. During the salification step, the addition of an alkali metal bicarbonate converts the phenolic hydroxyl groups into their corresponding salts. This ionization dramatically increases the polarity of the target molecule, causing it to precipitate out of the aprotic solvent while non-ionic or differently charged impurities remain dissolved. The subsequent trans-acid purification exploits the differential solubility of the free acid form in an aqueous organic acid solution. By recrystallizing the DIDNTB from this specific medium, macromolecular impurities are kept in solution while the target molecule forms a highly ordered crystal lattice. This recrystallization from an organic acid aqueous solution is the key determinant in pushing the final purity from the 90 percent range to the critical 97.0 percent threshold required for premium diagnostic applications.
How to Synthesize DIDNTB Efficiently
The synthesis of DIDNTB described in this patent offers a streamlined route that balances reaction efficiency with ease of isolation. The process begins with the iodination of 3,4,5,6-tetrabromophenol sulfophthalein, followed immediately by nitration to generate the crude scaffold. The true innovation lies in the downstream processing, where the crude material is not simply washed but chemically transformed into a salt and then back to an acid to purge impurities. This sequence ensures that the final material is free from the colored byproducts and residual halogens that typically plague sulfonphthalein synthesis. For process chemists looking to implement this, the detailed standardized synthesis steps are provided in the guide below, which outlines the specific molar ratios and temperature controls necessary to replicate the high-yield results observed in the patent examples.
- Perform iodination of 3,4,5,6-tetrabromophenol sulfophthalein using iodine monochloride and acetic anhydride in a solvent like dichloromethane.
- Conduct nitration on the iodinated product using fuming nitric acid and acetic anhydride to obtain the crude DIDNTB.
- Purify via salification with alkali metal bicarbonate followed by acid conversion with organic carboxylic acid and recrystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel DIDNTB preparation method translates into tangible operational improvements and risk mitigation. The primary advantage is the drastic simplification of the purification workflow. By achieving high purity through crystallization rather than complex chromatography or extensive washing, the manufacturing cycle time is significantly reduced. This efficiency gain lowers the overall cost of goods sold (COGS) by minimizing solvent consumption, energy usage for extended heating or cooling, and labor hours associated with difficult purification tasks. Furthermore, the robustness of the reaction conditions means that batch-to-batch variability is minimized, ensuring a consistent supply of material that meets strict quality control standards without the need for expensive re-processing or blending.
- Cost Reduction in Manufacturing: The elimination of harsh purification conditions and the improvement in total yield directly contribute to substantial cost savings. By utilizing reagents like iodine monochloride and acetic anhydride which drive reactions to near completion, the amount of wasted raw material is significantly decreased. The high yield of the salification and acid conversion steps means that less starting material is required to produce the same amount of final API intermediate. Additionally, the ability to use common aprotic solvents like acetone or acetonitrile, which are easier to recover and recycle compared to exotic solvents, further enhances the economic profile of the process, leading to a more competitive pricing structure for the final diagnostic reagent.
- Enhanced Supply Chain Reliability: The use of readily available and stable reagents ensures that the supply chain is resilient against market fluctuations. Unlike processes that rely on scarce catalysts or unstable intermediates, this method uses commodity chemicals that can be sourced from multiple suppliers globally. The mild reaction temperatures, ranging from -10 degrees Celsius to 50 degrees Celsius, reduce the dependency on specialized high-energy infrastructure, making the process easier to transfer between manufacturing sites. This flexibility guarantees continuity of supply, a critical factor for diagnostic companies that cannot afford interruptions in the production of life-saving kidney disease testing kits.
- Scalability and Environmental Compliance: The process is inherently designed for mass production, with parameters that scale linearly from laboratory to industrial reactors. The avoidance of heavy metal catalysts and the use of aqueous workups in the final acid conversion step simplify waste treatment protocols. This aligns with increasingly stringent environmental regulations, reducing the burden of hazardous waste disposal. The high purity of the final product also means less chemical waste is generated downstream by customers who do not need to perform additional purification, creating a greener value chain from the intermediate supplier to the final diagnostic device manufacturer.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this DIDNTB synthesis technology. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on how this method outperforms traditional routes. Understanding these details is crucial for technical teams evaluating the feasibility of adopting this new standard for producing high-purity diagnostic intermediates.
Q: What is the achieved purity of DIDNTB using this novel method?
A: The patented process achieves a final product purity of up to 97.0 percent to 98.75 percent, significantly overcoming the prior art limitation of less than 85 percent purity.
Q: Why is alkali metal bicarbonate preferred for the salification step?
A: Alkali metal bicarbonates provide optimal alkalinity that avoids the product discoloration associated with stronger bases like hydroxides while ensuring complete reaction compared to weaker bases like acetates.
Q: Can this synthesis method be scaled for industrial production?
A: Yes, the method utilizes mild reaction conditions, common solvents, and robust purification steps specifically designed to facilitate mass production and commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable DIDNTB Supplier
As the global demand for accurate early kidney disease diagnosis grows, the need for ultra-high purity indicators like DIDNTB has never been more critical. NINGBO INNO PHARMCHEM stands at the forefront of this evolution, leveraging advanced synthetic methodologies to deliver intermediates that exceed industry standards. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the volume requirements of major diagnostic manufacturers without compromising on quality. We operate with stringent purity specifications and utilize rigorous QC labs to verify that every batch of DIDNTB meets the >97 percent purity benchmark essential for sensitive protein error assays.
We invite procurement leaders and R&D directors to collaborate with us to optimize their supply chains. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume needs and formulation requirements. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us demonstrate how our mastery of complex halogenated sulfonphthalein synthesis can enhance the performance of your diagnostic products while driving down your overall manufacturing costs.
