Revolutionizing OLED Material Synthesis: Direct Oxidative Coupling for 4-(2-Nitrophenyl) Dibenzo[b,d]Furan
The landscape of organic photoelectric material synthesis is undergoing a significant transformation driven by the urgent need for cost-effective and scalable manufacturing processes. Patent CN113999196A introduces a groundbreaking preparation method for 4-(2-nitrophenyl) dibenzo[b,d]furan, a critical intermediate widely utilized in the fabrication of Organic Light Emitting Diodes (OLEDs), Organic Solar Cells (OSCs), and Organic Field Effect Transistors (OFETs). Traditionally, the synthesis of this high-value compound has been plagued by multi-step procedures involving hazardous reagents and expensive precursors. This new technology disrupts the status quo by enabling a direct, one-step oxidative coupling between inexpensive dibenzofuran and nitrobenzene. By leveraging a sophisticated Nickel/Silver dual-catalytic system coordinated with specialized beta-diketoimine ligands, this method achieves exceptional yields exceeding 95% under mild thermal conditions. For R&D directors and procurement strategists alike, this represents a paradigm shift from complex, low-efficiency pathways to a streamlined, economically viable production model that promises to redefine supply chain dynamics for advanced electronic chemicals.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of 4-(2-nitrophenyl) dibenzo[b,d]furan has relied heavily on cross-coupling reactions, specifically utilizing dibenzofuran-4-boric acid and o-nitrohalobenzene as starting materials. As illustrated in the reaction scheme below, this conventional pathway (Path A) necessitates a preliminary and highly demanding synthesis of the boronic acid precursor (Path B). This upstream step typically involves lithiation using n-butyllithium at cryogenic temperatures followed by boronation, a process that is not only operationally hazardous due to the pyrophoric nature of the reagents but also incurs substantial energy costs for maintaining low-temperature environments. Furthermore, the reliance on halogenated nitrobenzenes and palladium-catalyzed coupling introduces challenges related to heavy metal residue removal, which is critical for electronic-grade purity. The cumulative effect of these multi-step requirements results in a fragmented production workflow with lower overall atom economy and significantly higher manufacturing overheads.
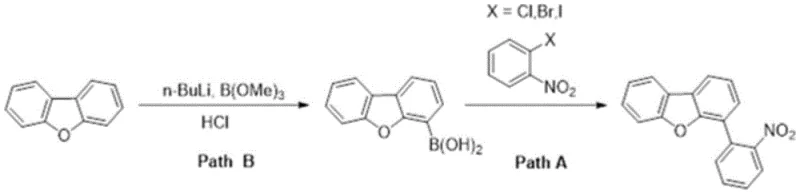
The Novel Approach
In stark contrast to the cumbersome traditional routes, the novel methodology disclosed in the patent employs a direct C-H/C-H oxidative coupling strategy. This approach bypasses the need for pre-functionalized boronic acid intermediates entirely, utilizing raw dibenzofuran and nitrobenzene directly. As depicted in the following reaction diagram, the process merges the reactants in a single vessel under the influence of a nickel catalyst, a silver co-catalyst, and a specific oxidant. This consolidation of steps drastically reduces the physical footprint of the reaction and eliminates the logistical complexities associated with handling unstable organolithium species. The use of nitrobenzene in large excess serves a dual purpose as both a reactant and a high-boiling solvent, which simplifies the reaction medium and facilitates easy recovery through distillation post-reaction. This elegant simplification of the synthetic route translates directly into enhanced process robustness and a dramatic reduction in the cost of goods sold (COGS) for high-purity OLED intermediates.
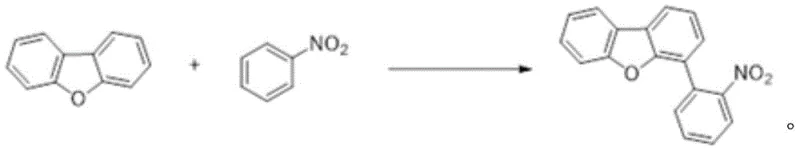
Mechanistic Insights into Ni/Ag Dual-Catalyzed Oxidative Coupling
The success of this direct functionalization hinges on the precise orchestration of a bimetallic catalytic cycle involving nickel and silver salts. The mechanism likely proceeds through a high-valent nickel species generated in situ, which activates the inert C-H bond of the dibenzofuran core. The presence of the silver salt acts as a crucial oxidant or Lewis acid promoter, facilitating the regeneration of the active catalytic species and stabilizing the transition states involved in the C-C bond formation. Crucially, the reaction efficiency is profoundly dependent on the electronic and steric environment provided by the ligand system. The patent highlights that conventional nitrogen-based ligands fail to support this transformation, underscoring the unique capability of the beta-diketoimine framework to stabilize the metal center while allowing sufficient access for the bulky aromatic substrates. This synergistic interaction between the metal centers and the tailored ligand architecture enables the activation of strong C-H bonds under relatively mild thermal conditions, avoiding the extreme temperatures often required for non-catalytic oxidative couplings.
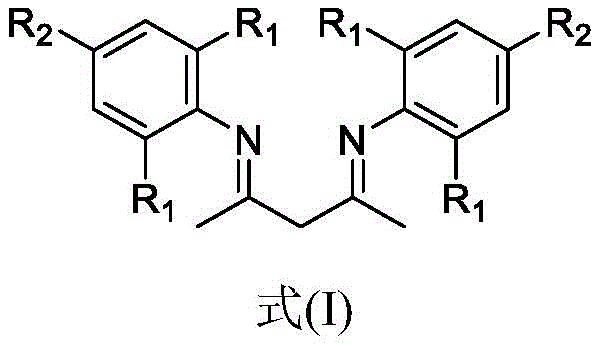
Central to the impurity control and yield optimization is the specific structural design of the beta-diketoimine ligand, as shown in Formula (I). The patent data reveals that subtle modifications to the substituents on the phenyl rings of the ligand (R1 and R2 groups) exert a profound influence on catalytic turnover. For instance, the introduction of electron-donating methyl groups enhances the electron density at the metal center, thereby accelerating the oxidative addition step. Conversely, the steric bulk provided by these substituents prevents the formation of inactive catalyst aggregates, ensuring a sustained catalytic cycle. Comparative examples in the patent demonstrate that omitting the ligand or substituting it with standard alternatives results in negligible product formation, confirming that the ligand is not merely an additive but an integral component of the active catalytic complex. This level of mechanistic control allows for the suppression of side reactions, such as over-oxidation or polymerization, ensuring that the final crude product possesses a purity profile suitable for direct crystallization without extensive chromatographic purification.
How to Synthesize 4-(2-Nitrophenyl) Dibenzo[b,d]Furan Efficiently
The implementation of this synthesis protocol is designed for seamless integration into existing chemical manufacturing infrastructure, requiring standard reactor setups capable of handling elevated temperatures and inert atmospheres. The procedure begins with the charging of dibenzofuran, a significant molar excess of nitrobenzene, the nickel and silver catalyst salts, the specific beta-diketoimine ligand, and an organic peroxide oxidant into a reaction vessel. The mixture is then heated to a temperature range of 60-150°C, with optimal results observed around 90°C, and maintained under vigorous stirring for a period of 6 to 24 hours. Following the completion of the reaction, the workup is remarkably straightforward: unreacted nitrobenzene is recovered via reduced pressure distillation, and the residue is subjected to a simple aqueous-organic extraction. The final high-purity product is isolated through crystallization from ethanol, yielding material with purity levels exceeding 99%. For detailed operational parameters and specific stoichiometric ratios optimized for different scales, please refer to the standardized synthesis guide below.
- Charge dibenzofuran, excess nitrobenzene (acting as both reactant and solvent), nickel salt, silver salt, beta-diketoimine ligand, and oxidant into a reactor under nitrogen protection.
- Heat the reaction mixture to 60-150°C and maintain stirring for 6-24 hours to facilitate direct C-H/C-H oxidative coupling.
- Recover unreacted nitrobenzene via reduced pressure distillation, extract the residue with ethyl acetate and water, and crystallize the organic layer to obtain high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, this patented technology offers compelling advantages that address the core pain points of cost volatility and supply continuity in the electronic chemicals sector. By eliminating the dependency on specialized boronic acid derivatives, which are often subject to supply bottlenecks and premium pricing, manufacturers can secure a more stable and predictable raw material base. The ability to use commodity chemicals like dibenzofuran and nitrobenzene as feedstocks decouples production costs from the fluctuations of the fine chemical intermediate market. Moreover, the simplification of the process flow—from multiple discrete steps to a single pot operation—drastically reduces labor hours, utility consumption, and equipment occupancy time. These operational efficiencies compound to deliver substantial cost savings in OLED material manufacturing, allowing suppliers to offer more competitive pricing structures without compromising on the stringent quality standards required by the display industry.
- Cost Reduction in Manufacturing: The economic impact of this process is driven primarily by the elimination of the costly and hazardous lithiation-boronation sequence. By removing the need for cryogenic cooling and pyrophoric reagents like n-butyllithium, the process significantly lowers energy consumption and safety compliance costs. Additionally, the recovery and reuse of excess nitrobenzene as a solvent further minimizes raw material waste, contributing to a leaner and more cost-effective production model that enhances overall profit margins for high-volume manufacturers.
- Enhanced Supply Chain Reliability: Utilizing widely available bulk chemicals as starting materials mitigates the risk of supply chain disruptions often associated with custom-synthesized intermediates. The robustness of the catalytic system ensures consistent batch-to-batch reproducibility, which is critical for maintaining long-term supply contracts with major panel manufacturers. This reliability allows procurement managers to reduce safety stock levels and optimize inventory turnover, knowing that the production lead time is governed by a simple, scalable reaction rather than a complex multi-vendor synthesis chain.
- Scalability and Environmental Compliance: The mild reaction conditions and absence of heavy palladium residues simplify the scale-up process from pilot to commercial production. The reduced generation of hazardous waste, particularly the avoidance of lithium salts and halogenated byproducts typical of Suzuki couplings, aligns with increasingly stringent environmental regulations. This green chemistry profile not only reduces waste disposal costs but also future-proofs the manufacturing site against evolving regulatory pressures, ensuring long-term operational sustainability.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis route. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation, providing a clear understanding of the technology's capabilities and limitations. Understanding these nuances is essential for technical teams evaluating the feasibility of adopting this method for large-scale production of optoelectronic intermediates.
Q: What are the primary advantages of this new synthesis method over traditional Suzuki coupling?
A: The new method eliminates the need for pre-functionalizing dibenzofuran into expensive boronic acids via hazardous lithiation. It utilizes cheap, commercially available dibenzofuran and nitrobenzene directly in a one-step process, significantly reducing raw material costs and operational complexity.
Q: Why is the beta-diketoimine ligand critical for this reaction?
A: The specific beta-diketoimine ligand structure enhances the electron-donating properties and steric hindrance around the nickel center. Experimental data shows that without this specific ligand class, or when using conventional ligands like 1,10-phenanthroline, the reaction fails to produce the target molecule effectively.
Q: How does this process impact scalability and environmental compliance?
A: By operating at moderate temperatures (60-150°C) and avoiding cryogenic conditions required for lithiation, the process is inherently safer and easier to scale. Furthermore, the excess nitrobenzene serves as a recyclable solvent, minimizing waste generation and simplifying downstream purification.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-(2-Nitrophenyl) Dibenzo[b,d]Furan Supplier
As the demand for high-performance organic semiconductors continues to surge, the ability to deliver key intermediates with unmatched purity and consistency becomes a decisive competitive advantage. NINGBO INNO PHARMCHEM stands at the forefront of this evolution, leveraging our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production to bring this cutting-edge technology to market. Our state-of-the-art facilities are equipped with rigorous QC labs and advanced process control systems designed to meet the stringent purity specifications demanded by the OLED and OPV industries. We are committed to translating complex academic innovations into robust industrial realities, ensuring that our partners receive materials that drive the next generation of display and energy technologies.
We invite forward-thinking organizations to collaborate with us to unlock the full potential of this efficient synthesis route. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out today to obtain specific COA data and comprehensive route feasibility assessments, ensuring that your supply chain is optimized for both performance and profitability in the rapidly evolving landscape of electronic materials.
