Advanced Grignard-Based Synthesis of Deuterated Phenylboronic Acid for Industrial Scale-Up
The pharmaceutical and advanced materials industries are constantly seeking robust methodologies for synthesizing isotopically labeled compounds, particularly deuterated intermediates which offer significant pharmacokinetic advantages. Patent CN115925730A introduces a groundbreaking preparation method for deuterated phenylboronic acid that addresses critical safety and efficiency bottlenecks found in traditional synthetic routes. This innovation leverages 2-methyltetrahydrofuran (2-MeTHF) as a superior reaction medium, replacing hazardous solvents like diethyl ether while achieving remarkably high conversion rates. The structural integrity and isotopic purity of the final product are paramount for its application in Suzuki coupling reactions and OLED material synthesis, making this patented approach a vital development for industrial chemists. By optimizing reaction temperatures and selecting specific esterifying agents, the process ensures that the valuable deuterium label is preserved throughout the synthesis, avoiding the common pitfall of hydrogen-deuterium exchange.
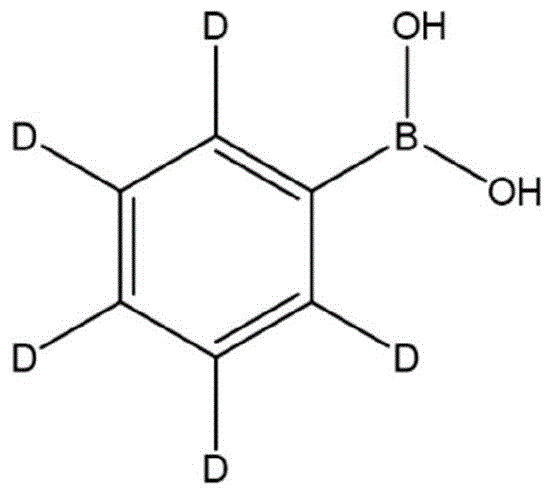
Furthermore, the transition to this novel solvent system represents a significant leap forward in green chemistry principles within fine chemical manufacturing. The use of 2-MeTHF not only mitigates the severe safety hazards associated with peroxide formation in ethers but also enhances the solubility and reactivity profiles of the organometallic intermediates involved. For R&D directors and process engineers, understanding the nuances of this solvent switch is essential for scaling production from laboratory benchtop to multi-ton commercial facilities. The patent details a comprehensive workflow that balances rigorous temperature control with efficient workup procedures, ensuring that the final deuterated phenylboronic acid meets the stringent purity specifications required for high-value applications in drug discovery and electronic materials.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of phenylboronic acids and their deuterated analogues has relied heavily on diethyl ether or standard tetrahydrofuran (THF) as the primary reaction solvents, both of which present distinct disadvantages for large-scale operations. Diethyl ether, while effective for Grignard reagent formation, possesses a dangerously low boiling point and high volatility, creating substantial risks of peroxide accumulation and subsequent explosion during storage and handling. These safety concerns necessitate expensive containment systems and rigorous monitoring protocols, driving up operational costs and complicating regulatory compliance for manufacturing sites. Additionally, when standard THF is substituted to mitigate some volatility issues, the conversion rate of the precious deuterated bromobenzene starting material often drops precipitously. Given the exorbitant cost of deuterated raw materials compared to their non-labeled counterparts, even a marginal decrease in yield translates to unacceptable economic losses and resource wastage in a commercial setting.
The Novel Approach
The methodology outlined in patent CN115925730A fundamentally re-engineers the solvent system by employing 2-methyltetrahydrofuran, a bio-based solvent derived from renewable resources that offers an optimal balance of safety and performance. This novel approach eliminates the explosive hazards of diethyl ether while providing a reaction environment that supports significantly higher conversion rates of the deuterated bromobenzene substrate. The process utilizes a carefully controlled temperature profile during the Grignard formation step, typically ranging between 50°C and 85°C, which accelerates the reaction kinetics without compromising the stability of the organometallic species. Furthermore, the selection of triisopropyl borate as the esterifying agent, coupled with precise low-temperature control during the subsequent quenching and acidification steps, ensures that the isotopic label remains intact. This holistic optimization results in a streamlined process that is not only safer for operators but also economically superior due to the maximized utilization of high-value inputs.
Mechanistic Insights into Grignard-Mediated Boronation
The core of this synthesis lies in the precise generation and stabilization of the deuterated phenylmagnesium bromide intermediate, which serves as the nucleophilic driver for the boronation reaction. In the initial stage, magnesium strips react with deuterated bromobenzene in the presence of iodine initiators within the 2-MeTHF solvent matrix. The choice of 2-MeTHF is mechanistically significant because its oxygen atom coordinates effectively with the magnesium center, stabilizing the Grignard reagent and preventing premature decomposition or Wurtz-type coupling side reactions. The reaction temperature is critically maintained between 50°C and 85°C, a range that is sufficiently high to overcome the activation energy barrier for magnesium insertion but low enough to prevent thermal degradation of the sensitive deuterated aromatic ring. This thermal window allows for a rapid and complete consumption of the magnesium metal, ensuring that the resulting Grignard solution is highly concentrated and reactive for the subsequent transformation.
Following the formation of the Grignard reagent, the mechanism proceeds through a nucleophilic attack on the boron center of the triisopropyl borate ester. This step is conducted at cryogenic temperatures, specifically between -40°C and -80°C, to suppress competing side reactions such as proton abstraction or homocoupling that could erode the yield. The low temperature kinetically favors the formation of the tetra-coordinate borate complex, which is then subjected to acidic hydrolysis using dilute hydrochloric acid. A crucial mechanistic detail highlighted in the patent is the prevention of hydrogen-deuterium (H/D) exchange during the workup and purification phases. The protocol mandates that any thermal treatment, particularly vacuum distillation for solvent removal, must be conducted below 60°C. Exceeding this threshold in the presence of acidic residues can catalyze the exchange of the deuterium atoms on the aromatic ring with protons from the environment, leading to isotopic dilution. By strictly adhering to these thermal limits, the process guarantees that the final deuterated phenylboronic acid retains the high isotopic purity of the starting material, a critical quality attribute for its intended applications.
How to Synthesize Deuterated Phenylboronic Acid Efficiently
Implementing this synthesis route requires strict adherence to the sequential addition of reagents and precise thermal management to ensure reproducibility and safety. The process begins with the activation of magnesium metal and the controlled addition of the deuterated halide, followed by the low-temperature boronation and careful acidic workup. Operators must be vigilant regarding the temperature constraints during the distillation phase to preserve the deuterium label. The detailed standardized synthesis steps, including specific molar ratios, dropwise addition rates, and purification protocols, are outlined in the technical guide below for immediate reference by process development teams.
- Prepare the Grignard reagent by reacting deuterated bromobenzene with magnesium strips in 2-methyltetrahydrofuran solvent at 50-85°C, initiated by iodine.
- React the Grignard solution with triisopropyl borate at low temperatures (-40°C to -80°C) to form the borate ester intermediate.
- Acidify the mixture with dilute hydrochloric acid and purify via vacuum distillation below 60°C to prevent deuterium loss.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented synthesis method offers tangible strategic benefits that extend beyond simple chemical yield improvements. The shift away from hazardous solvents like diethyl ether significantly reduces the regulatory burden and insurance costs associated with storing and handling volatile, peroxide-forming chemicals. This simplification of the safety profile allows for more flexible manufacturing scheduling and reduces the likelihood of production stoppages due to safety audits or incident investigations. Moreover, the enhanced conversion rate of the deuterated bromobenzene starting material directly impacts the cost of goods sold, as it minimizes the waste of one of the most expensive components in the supply chain. By maximizing the output per unit of input, manufacturers can achieve a more predictable and stable cost structure, which is essential for long-term contracting with pharmaceutical clients who demand consistent pricing.
- Cost Reduction in Manufacturing: The primary driver for cost reduction in this process is the dramatic improvement in the utilization rate of deuterated bromobenzene, a high-value raw material that often dictates the overall economics of the synthesis. Traditional methods using THF or ether frequently suffer from incomplete conversions or side reactions that consume the expensive starting material without generating the desired product. By optimizing the solvent system and reaction temperatures, this new method ensures that a significantly larger proportion of the input material is converted into the final deuterated phenylboronic acid. This efficiency gain means that less raw material is required to produce the same amount of product, leading to substantial savings on material costs without the need for complex recycling loops or additional purification steps that would otherwise add expense.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of 2-methyltetrahydrofuran, a solvent that is increasingly available from renewable feedstocks and is less subject to the petrochemical supply fluctuations that affect traditional ethers. The reduced hazard profile of the solvent also simplifies logistics, as it does not require the same level of specialized containment and transportation protocols as highly volatile diethyl ether. This ease of handling translates to fewer delays in raw material receipt and a smoother flow of materials through the production facility. Additionally, the robustness of the reaction conditions means that the process is less sensitive to minor variations in ambient conditions, ensuring consistent batch-to-batch quality and reliable delivery schedules for downstream customers.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, the process aligns perfectly with modern green chemistry initiatives, facilitating easier permitting and expansion of production capacity. The elimination of peroxide risks and the use of a bio-based solvent reduce the generation of hazardous waste streams, lowering disposal costs and minimizing the environmental footprint of the manufacturing site. The reaction conditions are amenable to scale-up, as the exothermic nature of the Grignard formation can be effectively managed within standard industrial reactors equipped with appropriate cooling systems. This scalability ensures that the supply of deuterated phenylboronic acid can be ramped up to meet surging demand from the OLED and pharmaceutical sectors without requiring prohibitive capital investment in new safety infrastructure.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this deuterated phenylboronic acid synthesis method. These answers are derived directly from the experimental data and process descriptions provided in the patent documentation, offering clarity on critical process parameters and quality control measures. Understanding these details is essential for technical teams evaluating the feasibility of adopting this route for their own production lines or for procurement specialists assessing the quality of the final product.
Q: Why is 2-Methyltetrahydrofuran preferred over diethyl ether for this synthesis?
A: 2-Methyltetrahydrofuran offers superior safety profiles by eliminating the high volatility and peroxide explosion risks associated with diethyl ether, while simultaneously providing higher conversion rates for expensive deuterated starting materials.
Q: How does the process prevent the loss of deuterium labels?
A: The process strictly controls purification temperatures, specifically keeping vacuum distillation below 60°C, to prevent hydrogen-deuterium exchange that typically occurs at higher thermal thresholds.
Q: What is the primary advantage of using triisopropyl borate in this reaction?
A: Triisopropyl borate is selected as the esterifying agent to minimize side reactions and maximize the utilization rate of the high-value deuterated bromobenzene raw material.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Deuterated Phenylboronic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-purity deuterated intermediates play in the development of next-generation therapeutics and advanced electronic materials. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the intricate temperature controls and solvent handling requirements of this Grignard-based synthesis are executed with precision. We maintain stringent purity specifications and operate rigorous QC labs to verify that every batch of deuterated phenylboronic acid meets the exact isotopic enrichment and chemical purity standards demanded by global innovators. Our commitment to quality assurance means that clients can rely on us for consistent supply without compromising on the critical deuterium content that defines the value of the product.
We invite potential partners to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how our efficient manufacturing processes can reduce your overall material costs. We encourage you to contact us directly to obtain specific COA data and route feasibility assessments tailored to your volume needs, ensuring a seamless integration of our high-quality intermediates into your supply chain.
