Advanced Ferriporphyrin-Catalyzed Synthesis of Cyclopropylboronic Acid for Commercial Scale-Up
The pharmaceutical industry's relentless pursuit of efficient synthetic routes for critical building blocks has found a significant breakthrough in the technology disclosed in patent CN109305983B. This patent details a sophisticated yet operationally simple method for the synthesis of cyclopropylboronic acid, a vital intermediate widely utilized in Suzuki-Miyaura cross-coupling reactions for drug discovery and development. Unlike conventional methodologies that rely on hazardous and expensive starting materials, this innovative approach leverages the reactivity of aldehyde boronic acid and strongly electron-withdrawing sulfonyl hydrazides. By introducing ethylene gas under the catalytic influence of ferriporphyrin complexes, the process achieves a direct cyclopropanation that bypasses the notorious instability associated with traditional organometallic reagents. This technological advancement represents a paradigm shift in how high-value boronic acids are manufactured, offering a pathway that is not only chemically elegant but also commercially robust for global supply chains.
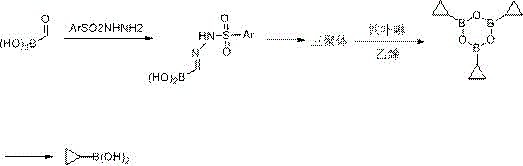
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of cyclopropylboronic acid has been plagued by significant chemical and logistical challenges that hinder efficient commercial production. The most prevalent traditional method involves the use of cyclopropyl bromide as the primary raw material, which is converted into a Grignard reagent or an organolithium species before reacting with borate esters. This approach suffers from inherent safety risks due to the pyrophoric nature of the intermediates and the high cost of cyclopropyl bromide itself. Furthermore, the preparation of cyclopropyl Grignard reagents is often inefficient, with yields compromised by the self-coupling of the bromide and precipitation issues in tetrahydrofuran solvents when concentrations exceed 1M. Similarly, cyclopropyl lithium reagents exhibit problematic half-lives in various solvents, leading to poor reproducibility and making the amplification of these processes from the laboratory to the plant floor extremely difficult and unpredictable for manufacturing teams.
The Novel Approach
In stark contrast to these legacy methods, the novel approach outlined in the patent utilizes a metal-catalyzed cyclopropanation strategy that fundamentally alters the risk profile and cost structure of the synthesis. By starting from aldehyde boronic acid and reacting it with strongly electron-withdrawing sulfonyl hydrazides, the process forms a stable hydrazone intermediate that can be dehydrated to a reactive trimer. This trimer then serves as a precursor for carbene generation in the presence of ethylene gas and a ferriporphyrin catalyst. This methodology completely avoids the use of cyclopropyl bromide and the subsequent generation of unstable organometallic species. The operational simplicity is remarkable, involving standard reflux conditions and gas introduction rather than cryogenic temperatures and strictly anhydrous environments required for lithiation, thereby providing a new, safer, and more reliable synthesis path for the global production of cyclopropyl boronic acid.
Mechanistic Insights into Ferriporphyrin-Catalyzed Cyclopropanation
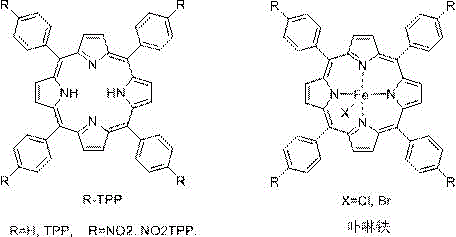
The core of this technological innovation lies in the precise mechanistic action of the ferriporphyrin catalyst, which facilitates the decomposition of the hydrazone-derived trimer to generate a reactive metal-carbene species. As illustrated in the structural diagrams provided in the patent, the catalyst typically consists of an iron center coordinated within a tetraphenylporphyrin (TPP) ligand framework, potentially substituted with electron-withdrawing groups to modulate reactivity. When the trimer interacts with the iron porphyrin complex, nitrogen gas is extruded, generating an iron-carbenoid intermediate that is electrophilic in nature. This highly reactive species then undergoes a concerted [2+1] cycloaddition with the introduced ethylene gas. The efficiency of this step is critical, as it determines the overall yield of the cyclopropyl ring formation without the side reactions typical of radical pathways. The use of iron, an abundant and non-toxic transition metal, further enhances the environmental profile of the reaction compared to noble metal catalysts often used in similar transformations.
From an impurity control perspective, this catalytic cycle offers distinct advantages over the traditional Grignard route. In conventional synthesis, the formation of Wurtz-type coupling byproducts from cyclopropyl bromide is a persistent issue that complicates purification and lowers the purity of the final API intermediate. The ferriporphyrin-catalyzed route minimizes these homocoupling impurities because the reaction mechanism does not involve free radical intermediates that typically lead to dimerization. Instead, the carbene transfer is a controlled, metal-mediated process. Additionally, the starting material, aldehyde boronic acid, can be prepared with high purity, and the subsequent hydrazone formation acts as a protective strategy for the boronic acid moiety during the harsh cyclopropanation conditions. This ensures that the final hydrolysis step yields cyclopropylboronic acid with a clean impurity profile, meeting the stringent specifications required for pharmaceutical applications without the need for extensive chromatographic purification.
How to Synthesize Cyclopropylboronic Acid Efficiently
The synthesis protocol described in the patent provides a clear roadmap for executing this transformation with high fidelity and reproducibility. The process begins with the condensation of aldehyde boronic acid and a sulfonyl hydrazide in an alcoholic solvent, followed by a dehydration step in a non-polar solvent like toluene to generate the key trimer intermediate. The critical cyclopropanation step requires the careful introduction of ethylene gas into a solution of the trimer and the iron catalyst, maintained under slight pressure to ensure saturation.
- React aldehyde boronic acid with strongly electron-withdrawing sulfonyl hydrazide in an alcohol solvent under reflux to form the corresponding hydrazone intermediate.
- Dehydrate the hydrazone in a solvent like toluene or dioxane to generate the reactive trimer species, ensuring strict exclusion of moisture.
- Introduce ethylene gas into the reaction mixture containing the trimer and a ferriporphyrin catalyst (e.g., FeCl(TPP)) to effect cyclopropanation, followed by hydrolysis to isolate the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthesis route offers compelling strategic benefits that extend beyond mere chemical curiosity. The shift away from cyclopropyl bromide represents a significant mitigation of supply risk, as bromide derivatives are often subject to volatile pricing and regulatory scrutiny regarding halogenated waste. By utilizing ethylene, a commodity chemical available in vast quantities globally, the dependency on specialized, high-cost precursors is drastically reduced. This transition not only stabilizes the cost of goods sold but also simplifies the logistics of raw material sourcing, allowing for more predictable budgeting and long-term supply contracts. Furthermore, the elimination of pyrophoric reagents reduces the need for specialized storage and handling infrastructure, lowering the overall operational expenditure associated with safety compliance and insurance.
- Cost Reduction in Manufacturing: The economic implications of replacing cyclopropyl bromide with ethylene and aldehyde boronic acid are profound. Ethylene is one of the most inexpensive organic feedstocks available in the petrochemical industry, whereas cyclopropyl bromide is a specialized reagent with a much higher price point and limited supplier base. By removing the expensive halide and the complex steps associated with Grignard reagent preparation, the overall material cost per kilogram of the final product is significantly lowered. Additionally, the use of iron porphyrin catalysts, which can be synthesized from readily available porphyrins and iron salts, avoids the high costs associated with palladium or rhodium catalysts often used in fine chemical synthesis, leading to substantial savings in catalyst expenditure.
- Enhanced Supply Chain Reliability: Supply chain resilience is greatly improved by the robustness of the reagents involved in this new method. Traditional organolithium routes require strict temperature control and immediate usage, creating bottlenecks in production scheduling. In contrast, the hydrazone and trimer intermediates in this process are relatively stable and can be isolated or stored if necessary, providing greater flexibility in manufacturing planning. The reliance on gaseous ethylene, which can be sourced from multiple industrial suppliers, ensures that production is not held hostage by the availability of a single niche chemical vendor. This diversification of the supply base is crucial for maintaining continuous production lines and meeting the just-in-time delivery requirements of major pharmaceutical clients.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial production is facilitated by the simplicity of the unit operations involved. The reaction proceeds under reflux conditions in common solvents like toluene or dioxane, which are well-understood in large-scale reactor systems. The avoidance of cryogenic conditions (-78°C) required for lithiation removes a major energy burden and equipment constraint, allowing for faster batch turnover times. From an environmental standpoint, the process generates less hazardous waste compared to the magnesium or lithium salt byproducts of traditional methods. The use of iron, a benign metal, aligns with green chemistry principles, simplifying wastewater treatment and reducing the environmental footprint of the manufacturing facility, which is increasingly important for regulatory compliance and corporate sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These answers are derived directly from the experimental data and technical disclosures within the patent documentation to provide clarity for potential partners and licensees. Understanding these nuances is essential for evaluating the feasibility of integrating this technology into existing manufacturing portfolios.
Q: What are the primary advantages of this synthesis method over traditional Grignard routes?
A: This method eliminates the need for hazardous cyclopropyl bromide and avoids the formation of unstable Grignard or organolithium reagents, which often suffer from low yields and reproducibility issues during scale-up. It utilizes stable aldehyde boronic acid and inexpensive ethylene gas.
Q: What type of catalyst is required for the cyclopropanation step?
A: The process utilizes iron porphyrin complexes, such as FeCl(TPP) or FeBr(TPP), which effectively catalyze the decomposition of the hydrazone derivative to generate the reactive carbene species for ethylene insertion.
Q: Is this process suitable for large-scale manufacturing of pharmaceutical intermediates?
A: Yes, the patent highlights simple operational conditions, such as refluxing in common solvents and using gaseous ethylene, which are highly amenable to industrial scale-up compared to cryogenic organolithium processes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cyclopropylboronic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the ferriporphyrin-catalyzed synthesis route for producing high-quality cyclopropylboronic acid. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are fully realized in a GMP-compliant environment. Our rigorous QC labs and stringent purity specifications guarantee that every batch of cyclopropylboronic acid meets the exacting standards required for modern drug development, minimizing the risk of downstream failures in Suzuki coupling reactions. We are committed to leveraging this advanced chemistry to provide our clients with a superior product that combines cost-efficiency with unmatched reliability.
We invite forward-thinking pharmaceutical companies and research institutions to collaborate with us to explore the full capabilities of this synthesis platform. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements and project timelines. We encourage you to reach out today to obtain specific COA data and comprehensive route feasibility assessments, allowing you to make informed decisions that will optimize your supply chain and accelerate your time to market.
