Advanced Synthesis of 3-(Alpha-Methoxy)Methylenebenzofuran-2(3H)-One for Commercial Agrochemical Production
Advanced Synthesis of 3-(Alpha-Methoxy)Methylenebenzofuran-2(3H)-One for Commercial Agrochemical Production
The global demand for high-efficiency strobilurin fungicides, particularly azoxystrobin, has necessitated the development of robust and cost-effective synthetic routes for key intermediates such as 3-(α-methoxy)-methylenebenzofuran-2(3H)-one. Patent CN102190640B introduces a transformative methodology that leverages hydrocyanic acid, potentially sourced from acrylonitrile waste gas, to achieve this transformation with superior efficiency. This technical insight report analyzes the proprietary two-step sequence involving hydrocyanation followed by methoxylation, which stands in stark contrast to legacy processes reliant on orthoformates. For R&D directors and procurement strategists, understanding this shift is critical, as it represents a move towards atom-economical processes that minimize hazardous waste while maximizing yield consistency in the production of high-purity agrochemical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of 3-(α-methoxy)-methylenebenzofuran-2(3H)-one has relied heavily on the condensation of benzofuran-2(3H)-one with trimethyl orthoformate in the presence of acetic anhydride. As illustrated in the reaction scheme below, this traditional pathway suffers from significant thermodynamic and economic inefficiencies. The acetic anhydride serves a dual role as both a catalyst and a dehydrating agent to scavenge the methanol byproduct, forming methyl acetate. However, this results in the generation of substantial volumes of methyl acetate waste, which complicates solvent recovery systems and increases the environmental burden on manufacturing facilities. Furthermore, the reliance on trimethyl orthoformate introduces a high raw material cost factor, and the reaction kinetics often dictate prolonged processing times to drive equilibrium towards the product, ultimately leading to lower space-time yields and inflated operational expenditures.
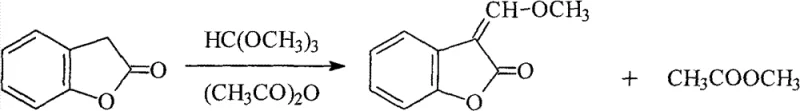
The Novel Approach
In a decisive break from convention, the methodology disclosed in CN102190640B utilizes a direct formylation strategy employing hydrocyanic acid (HCN) and a mineral acid catalyst, followed by a methoxylation step. This novel approach fundamentally alters the reaction landscape by bypassing the equilibrium limitations inherent in orthoformate chemistry. By introducing HCN, the process facilitates a direct addition to the carbonyl system under mild conditions, typically ranging from -10°C to 25°C, which preserves the integrity of the sensitive benzofuran ring system. The subsequent methoxylation using dimethyl sulfate proceeds with high selectivity, eliminating the formation of bulky ester byproducts. This streamlined pathway not only simplifies the workup procedure but also drastically reduces the volume of organic waste streams, positioning it as a preferred route for manufacturers seeking to optimize cost reduction in strobilurin fungicide manufacturing while adhering to green chemistry principles.
Mechanistic Insights into Hydrocyanation and Methoxylation
The core of this innovative synthesis lies in the electrophilic activation of the benzofuran-2(3H)-one substrate by a strong acid catalyst, such as hydrogen chloride or hydrogen bromide. In the first stage, the catalyst protonates the carbonyl oxygen, increasing the electrophilicity of the C3 position and facilitating the nucleophilic attack by the cyanide ion derived from hydrocyanic acid. This results in the formation of a transient imine or nitrile intermediate which, upon acidic hydrolysis during the workup phase, yields the crucial 3-formylbenzofuran-2(3H)-one. The precision of this mechanism allows for tight control over impurity profiles, as the mild temperature window (-10°C to 25°C) suppresses side reactions such as polymerization or ring-opening that are common in harsher acidic environments. The stoichiometry is carefully balanced, with a molar ratio of benzofuran-2(3H)-one to catalyst to HCN maintained at approximately 1.0:1.0~1.5:1.0~1.5, ensuring complete conversion without excessive reagent waste.

Following the isolation of the aldehyde intermediate, the second mechanistic phase involves an O-methylation reaction utilizing dimethyl sulfate as the methylating agent in the presence of a base like potassium carbonate. This step converts the enolizable aldehyde functionality into the stable α-methoxy methylene group. The reaction mechanism proceeds via the formation of an enolate anion, which attacks the methyl group of the dimethyl sulfate in an SN2 fashion. The choice of dimethyl sulfate is strategic; it is a potent methylating agent that reacts rapidly at temperatures between 0°C and 60°C, completing the transformation within 1 to 4 hours. This rapid kinetics minimizes the exposure of the product to basic conditions, thereby preventing potential degradation or racemization issues, and ensures that the final crystalline product achieves a melting point of 100~101°C with high purity, suitable for direct use in subsequent coupling reactions for azoxystrobin synthesis.
How to Synthesize 3-(Alpha-Methoxy)Methylenebenzofuran-2(3H)-one Efficiently
The operational protocol for this synthesis is designed for scalability and safety, beginning with the careful handling of hydrocyanic acid in an anhydrous toluene medium. The process initiates by cooling the reaction mixture to sub-zero temperatures before the introduction of dry hydrogen chloride gas, a critical control point to manage the exotherm and ensure the formation of the reactive iminium species. Following the formylation and hydrolysis, the organic layer is separated and dried, yielding the intermediate oil which is then subjected to methoxylation in a polar aprotic solvent like DMF. The detailed standardized operating procedures, including specific quenching methods and crystallization protocols to maximize recovery, are outlined in the technical guide below.
- React benzofuran-2(3H)-one with hydrocyanic acid and a catalyst (HCl or HBr) at -10 to 25°C to form the 3-formyl intermediate.
- Hydrolyze the reaction mixture, separate the organic layer, and isolate the 3-formylbenzofuran-2(3H)-one intermediate.
- React the isolated intermediate with dimethyl sulfate in the presence of a base (e.g., potassium carbonate) at 0 to 60°C to yield the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of the HCN-based route described in CN102190640B offers compelling strategic advantages that extend beyond simple yield metrics. The primary value driver is the substitution of high-cost specialty reagents like trimethyl orthoformate with commodity chemicals such as hydrocyanic acid and dimethyl sulfate. Since HCN can be sourced as a byproduct from acrylonitrile production, the raw material cost basis is significantly lowered, providing a buffer against market volatility in fine chemical pricing. Moreover, the elimination of acetic anhydride and the consequent reduction in methyl acetate byproduct generation simplifies the distillation and solvent recovery infrastructure required on-site. This reduction in downstream processing complexity translates directly into lower utility consumption and reduced turnaround time between batches, enhancing overall plant throughput without the need for capital-intensive equipment upgrades.
- Cost Reduction in Manufacturing: The economic model of this process is fundamentally superior due to the avoidance of expensive dehydrating agents and the utilization of lower-cost C1 building blocks. By removing the need for acetic anhydride, manufacturers eliminate a significant line item from their bill of materials, while the high atom economy of the hydrocyanation step ensures that a greater proportion of input mass is converted into valuable product rather than waste. This structural cost advantage allows for more competitive pricing in the global agrochemical intermediate market, enabling suppliers to maintain healthy margins even during periods of raw material inflation.
- Enhanced Supply Chain Reliability: The reliance on widely available industrial gases and bulk liquids enhances supply security compared to routes dependent on specialized esters or orthoesters which may have limited supplier bases. The robustness of the reaction conditions, which tolerate a broad temperature range and utilize common solvents like toluene and DMF, reduces the risk of batch failures due to minor parameter deviations. This operational stability ensures consistent delivery schedules for downstream API manufacturers, mitigating the risk of production stoppages caused by intermediate shortages.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, the drastic reduction in liquid waste volume simplifies effluent treatment protocols. The process generates fewer organic residues, lowering the cost and complexity of hazardous waste disposal. Furthermore, the ability to utilize waste HCN streams aligns with circular economy initiatives, potentially qualifying the manufacturing site for green incentives. The simplicity of the workup, involving standard extraction and crystallization, facilitates seamless scale-up from pilot kilogram quantities to multi-ton commercial production campaigns.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These answers are derived directly from the experimental data and claims presented in the patent literature, providing a factual basis for technology transfer discussions. Understanding these nuances is essential for engineering teams evaluating the feasibility of retrofitting existing production lines or designing new facilities dedicated to strobilurin intermediate manufacture.
Q: What are the primary advantages of the HCN-based route over the traditional orthoformate method?
A: The HCN-based route described in patent CN102190640B eliminates the need for expensive trimethyl orthoformate and acetic anhydride, significantly reducing raw material costs. Furthermore, it avoids the generation of large quantities of methyl acetate byproduct, simplifying downstream purification and waste treatment compared to conventional methods.
Q: What are the critical reaction conditions for the formylation step?
A: The formylation reaction requires precise temperature control between -10°C and 25°C and a reaction time of 2 to 4 hours. The molar ratio of benzofuran-2(3H)-one to catalyst (HCl or HBr) and hydrocyanic acid is maintained between 1.0:1.0~1.5:1.0~1.5 to ensure optimal conversion to the 3-formyl intermediate.
Q: How does this synthesis method impact environmental compliance?
A: By utilizing hydrocyanic acid potentially sourced from acrylonitrile waste gas and avoiding excessive organic byproducts like methyl acetate, this method generates significantly less 'three wastes' (waste water, gas, and residue). This aligns with stricter environmental regulations for agrochemical manufacturing facilities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-(Alpha-Methoxy)Methylenebenzofuran-2(3H)-one Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to more efficient synthetic routes requires a partner with deep technical expertise and proven manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patent CN102190640B are fully realized in a GMP-compliant environment. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced analytical instrumentation to guarantee that every batch of 3-(α-methoxy)-methylenebenzofuran-2(3H)-one meets the exacting standards required for pharmaceutical and agrochemical applications. Our commitment to quality assurance ensures that impurities are kept well below threshold limits, safeguarding the efficacy and safety of the final fungicide products.
We invite global partners to engage with our technical procurement team to discuss how this optimized synthesis route can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can quantify the potential economic benefits specific to your volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that drive value and sustainability in your agrochemical manufacturing operations.
