Advanced Synthesis of Bis(fluorosulfonyl)imide Salts for Next-Generation Battery Electrolytes
Advanced Synthesis of Bis(fluorosulfonyl)imide Salts for Next-Generation Battery Electrolytes
The rapid expansion of the new energy sector, driven by global carbon neutrality goals, has placed immense pressure on the supply chain for high-performance battery materials. Central to this challenge is the development of advanced electrolyte salts that offer superior conductivity and thermal stability compared to traditional lithium hexafluorophosphate. Patent CN114031053B introduces a groundbreaking preparation method for bis(fluorosulfonyl)imide salts, specifically targeting lithium, sodium, and potassium variants essential for both power and energy storage batteries. This technology addresses critical bottlenecks in purity and yield that have historically plagued the mass production of these vital components. By leveraging a unique dual-solvent precipitation strategy, the invention promises to deliver electrolyte salts with exceptional quality metrics, including ultra-low acid values and minimal halogen residues. For industry leaders seeking a reliable electrolyte salt supplier, understanding the mechanistic advantages of this patent is crucial for securing a competitive edge in the evolving battery market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of bis(fluorosulfonyl)imide salts has been fraught with significant technical and environmental challenges that hinder efficient commercial scale-up of complex electrolyte additives. Traditional routes often involve reacting dichlorosulfimide acid with fluorinated alkali metal salts, a process that generates massive quantities of corrosive hydrogen chloride and chloralkali metal salt byproducts, leading to severe three-waste accumulation. Alternative double decomposition reactions using organic or inorganic alkali salts frequently suffer from incomplete conversion, leaving behind difficult-to-separate organic amine residues that cause discoloration and fail to meet strict chromaticity standards. Furthermore, direct neutralization with inorganic bases introduces water into the system, triggering hydrolysis of the sensitive imide acid and generating sulfate and ammonium impurities that drastically elevate acid values. Even methods utilizing lithium salts often result in persistent halogen residues or high acid values due to the inability to separate byproduct acids during solvent evaporation. These legacy processes not only compromise the electrochemical performance of the final battery but also impose heavy burdens on waste treatment and purification costs.
The Novel Approach
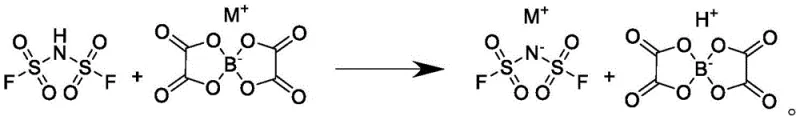
In stark contrast to these cumbersome legacy techniques, the novel approach disclosed in the patent utilizes a sophisticated solubility differential to achieve near-perfect separation in a single step. The core innovation lies in dissolving the bisoxalato borate in a benign solvent and adding it dropwise to a solution of difluorosulfimide acid in an inert solvent. This specific combination ensures that the inert solvent remains in excess throughout the reaction, creating an environment where the target bis(fluorosulfonyl)imide salt precipitates out of the solution immediately upon formation. Simultaneously, the byproduct, bisoxalato boric acid, remains highly soluble in the mixed solvent system, preventing it from co-precipitating with the product. This elegant design eliminates the need for energy-intensive evaporation, recrystallization, or complex washing procedures that are typical in older methods. Consequently, the final product can be obtained simply by filtration, resulting in a material with significantly reduced processing time and superior structural integrity.
Mechanistic Insights into Dual-Solvent Precipitation
The chemical efficacy of this process is rooted in the precise manipulation of solvation energies and intermolecular interactions within the reaction medium. When the bisoxalato borate solution is introduced to the acidic inert solvent, the immediate proton transfer facilitates the formation of the imide salt anion, which possesses low solubility in the specific inert solvent mixture employed, such as 1,2-tetrachloroethane or petroleum ether derivatives. The presence of the benign solvent, which acts as a carrier for the borate reactant, is carefully calibrated to maintain the solubility of the oxalic acid byproduct while simultaneously depressing the solubility of the target salt. This thermodynamic driving force ensures that nucleation occurs rapidly and selectively for the desired product, effectively excluding impurities from the crystal lattice as it forms. The reaction conditions, typically maintained between -15°C and -5°C, further suppress side reactions and hydrolysis, preserving the integrity of the fluorosulfonyl groups which are sensitive to thermal degradation. This controlled environment is essential for achieving the high purity levels required for high-voltage battery applications.
From an impurity control perspective, the mechanism effectively isolates the product from the most problematic contaminants found in conventional syntheses. Because the byproduct bisoxalato boric acid stays in the supernatant, the filtered cake is inherently free from the organic residues and metal ions that typically necessitate multiple recrystallization cycles. The absence of water generation during the reaction prevents the hydrolytic cleavage of the S-F bonds, thereby keeping fluoride ion release and acid value formation to trace levels. Analytical data from the patent examples demonstrates that this method consistently yields products with acid values as low as 20ppm and chloride ion content below 3ppm, metrics that are difficult to achieve with standard neutralization routes. This inherent purity reduces the burden on downstream quality control and ensures that the electrolyte salt performs optimally in reducing internal resistance and enhancing cycle life within the battery cell.
How to Synthesize Bis(fluorosulfonyl)imide Salt Efficiently
Implementing this synthesis route requires careful attention to solvent selection and temperature control to maximize the precipitation efficiency described in the patent documentation. The process begins with the preparation of two distinct solutions: one containing the difluorosulfimide acid dissolved in a chlorinated inert solvent, and the other containing the appropriate bisoxalato borate salt dissolved in an ester or ether-based benign solvent. Operators must ensure that the reaction vessel is cooled to the specified sub-zero range before initiating the dropwise addition, as thermal management is critical for controlling particle size and preventing oiling out. Once the addition is complete, the mixture is stirred for a defined period to allow the precipitation to reach completion before undergoing simple filtration. For a detailed breakdown of the specific molar ratios, solvent volumes, and operational parameters, please refer to the standardized synthesis guide provided below.
- Prepare a solution of difluorosulfimide acid in an inert solvent such as 1,2-tetrachloroethane within a reaction vessel.
- Dissolve bisoxalato borate (lithium, sodium, or potassium salt) in a benign solvent like ethyl acetate or tetrahydrofuran.
- Dropwise add the borate solution to the acid solution at low temperatures (-5°C to -15°C), stir for 3-7 hours, and filter the precipitated salt directly.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this precipitation technology represents a strategic opportunity to optimize the cost structure and reliability of battery material sourcing. By eliminating the need for multiple purification steps such as recrystallization and solvent evaporation, the process drastically simplifies the manufacturing workflow, leading to substantial reductions in energy consumption and equipment utilization time. The ability to obtain high-purity product directly via filtration means that production throughput can be significantly increased without compromising on quality specifications, addressing the growing demand for electrolyte salts in the electric vehicle sector. Furthermore, the reduction in hazardous byproducts and waste streams aligns with increasingly stringent environmental regulations, mitigating the risk of compliance-related disruptions. This streamlined approach not only enhances the economic viability of producing high-purity LiFSI and NaFSI but also strengthens the resilience of the supply chain against raw material volatility.
- Cost Reduction in Manufacturing: The elimination of energy-intensive evaporation and repeated crystallization steps directly translates to lower operational expenditures per kilogram of finished product. By avoiding the use of expensive fluorinating agents and minimizing the generation of corrosive waste that requires specialized disposal, the overall cost of goods sold is significantly optimized. The high yield reported in the patent examples indicates that raw material utilization is maximized, reducing the effective cost of the input acids and borate salts. Additionally, the simplified workflow reduces the labor and maintenance costs associated with complex purification trains, allowing for a more lean and efficient production model that supports cost reduction in battery material manufacturing.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route ensures a consistent supply of critical electrolyte components, which is vital for maintaining uninterrupted battery production lines. Since the process relies on readily available solvents and avoids the bottlenecks associated with difficult-to-source fluorinating reagents, the risk of supply disruption is minimized. The high purity achieved without extensive post-processing means that lead times for delivering ready-to-use material can be drastically shortened compared to traditional methods. This reliability is crucial for partners seeking a reliable electrolyte salt supplier who can guarantee steady volumes to meet the aggressive scaling targets of the new energy industry.
- Scalability and Environmental Compliance: The simplicity of the filtration-based isolation method makes this technology highly scalable from pilot batches to multi-ton commercial production facilities. The reduction in liquid waste volume and the absence of heavy metal or halogen-rich effluents simplify the wastewater treatment requirements, ensuring easier compliance with local environmental standards. This environmental advantage not only reduces regulatory risk but also enhances the sustainability profile of the final battery product, a key factor for end-users in the automotive and energy storage sectors. The process design inherently supports green chemistry principles, making it a future-proof solution for the sustainable commercial scale-up of complex electrolyte additives.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this novel synthesis method. These insights are derived directly from the experimental data and technical disclosures within the patent to provide clarity on its practical application. Understanding these details is essential for evaluating the feasibility of integrating this technology into existing production frameworks or sourcing strategies.
Q: What are the primary impurities removed by this new precipitation method?
A: The method effectively minimizes halogen ion residues (chloride and fluoride), reduces acid value to trace levels (e.g., 20ppm), and significantly lowers turbidity compared to traditional double decomposition or neutralization routes.
Q: Why is the dual-solvent system critical for product quality?
A: The use of an inert solvent for the acid and a benign solvent for the borate ensures that the byproduct, bisoxalato boric acid, remains soluble in the mixture while the target imide salt precipitates, eliminating the need for complex purification steps.
Q: Can this process be scaled for industrial battery material production?
A: Yes, the process avoids energy-intensive evaporation and crystallization steps, relying instead on simple filtration, which makes it highly suitable for large-scale commercial manufacturing of electrolyte salts.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Bis(fluorosulfonyl)imide Salt Supplier
As the demand for high-performance battery materials continues to surge, partnering with an experienced CDMO capable of executing complex synthetic routes is essential for maintaining market leadership. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. Our facility is equipped with rigorous QC labs and adheres to stringent purity specifications, guaranteeing that every batch of electrolyte salt meets the exacting standards required for next-generation power and energy storage batteries. We understand the critical nature of impurity profiles in electrolyte formulations and are committed to delivering products that enhance battery safety and longevity.
We invite you to engage with our technical team to explore how this advanced precipitation technology can be tailored to your specific production requirements. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits this method offers for your specific application. We encourage potential partners to contact our technical procurement team to索取 specific COA data and route feasibility assessments, ensuring that your transition to high-purity electrolyte salts is seamless and commercially viable.
