Advanced Synthesis of N,N-Dimethyl Trifluoromethyl Sulfonamide for High-Performance Battery Electrolytes
The rapid evolution of lithium-ion battery technology demands electrolyte additives that can simultaneously enhance safety profiles and electrochemical stability across extreme temperature ranges. Patent CN115703722A introduces a groundbreaking preparation method for N,N-dimethyl trifluoromethyl sulfonamide, a critical functional additive known for its ability to improve flame retardancy and stabilize active systems like lithium-sulfur and lithium-oxygen batteries. Unlike conventional approaches that struggle with harsh reaction conditions and complex purification, this invention leverages a sophisticated alcohol solvent system combined with a strategic alkoxide treatment step. This dual-phase approach not only resolves the persistent issue of byproduct decomposition but also facilitates the efficient recovery of valuable raw materials, marking a significant leap forward in the manufacturing of high-performance electronic chemicals. For industry leaders seeking reliable battery electrolyte additive suppliers, understanding this technological shift is paramount for securing supply chains that prioritize both purity and economic efficiency.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of N,N-dimethyl trifluoromethyl sulfonamide has been plagued by severe operational constraints that hinder scalable production. Traditional methodologies, such as those disclosed in earlier literature, typically rely on reacting dimethylamine gas with trifluoromethanesulfonyl fluoride in ether solvents at cryogenic temperatures as low as -78°C. These extreme conditions necessitate specialized refrigeration equipment and result in exorbitant energy costs, making the process economically unviable for mass manufacturing. Furthermore, the post-reaction workup in these legacy methods is notoriously cumbersome, involving multiple extraction steps, repeated washing, and complex distillation sequences to separate the product from unreacted gases and solvents. A critical failure point in these older schemes is the handling of the byproduct, dimethylamine hydrogen halide, which tends to decompose during the final distillation stages, releasing impurities that drastically lower the final product purity to below acceptable standards for sensitive battery applications.
The Novel Approach
In stark contrast, the methodology outlined in patent CN115703722A revolutionizes the production landscape by shifting the reaction medium to common alcohol solvents such as methanol, ethanol, or isopropanol. This transition allows the reaction to proceed smoothly at much more manageable temperatures ranging from -15°C to -30°C, eliminating the need for deep-freeze infrastructure. The true innovation lies in the post-reaction treatment where a corresponding alkoxide, such as sodium methoxide or sodium ethoxide, is introduced to the reaction mixture. This addition triggers a chemical transformation that decomposes the problematic dimethylamine hydrogen halide byproduct into reusable dimethylamine and a filterable inorganic salt. Consequently, the process achieves a closed-loop system where both the amine reactant and the alcohol solvent can be distilled and recycled, dramatically simplifying the purification workflow and ensuring consistent high-quality output suitable for cost reduction in electronic chemical manufacturing.
Mechanistic Insights into Alcohol-Mediated Sulfonamidation with Alkoxide Recovery
The core chemical ingenuity of this patent resides in the strategic manipulation of acid-base chemistry to drive equilibrium and facilitate separation. Initially, dimethylamine acts as both a nucleophile and an acid-binding agent in the alcohol solvent, reacting with trifluoromethyl sulfonyl halide to form the target sulfonamide and the salt byproduct, dimethylamine hydrogen halide. Under standard conditions, this salt would remain stable and contaminate the product stream. However, the introduction of the alkoxide (M+RO-) serves as a strong base that deprotonates the ammonium salt. This reaction effectively liberates free dimethylamine and regenerates the alcohol solvent, while precipitating the halide as a solid inorganic salt (M+X-). This mechanism ensures that the reaction mixture entering the distillation phase contains primarily the desired product, the solvent, and the free amine, rather than the thermally unstable salt.
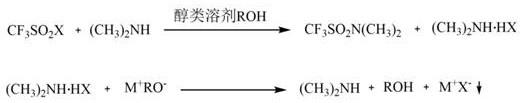
From an impurity control perspective, this mechanistic pathway is superior because it physically removes the source of contamination before thermal stress is applied. In conventional methods, heating the dimethylamine hydrogen halide causes it to break down, releasing acidic species that can degrade the sulfonamide product or create difficult-to-remove azeotropes. By converting this byproduct into a non-volatile inorganic salt like sodium fluoride or sodium chloride prior to distillation, the new method ensures that the subsequent atmospheric distillation only separates volatile components (amine and alcohol) from the non-volatile product and salt. The solid salt is removed via simple filtration, leaving a clean filtrate that yields N,N-dimethyl trifluoromethyl sulfonamide with purity levels consistently exceeding 99.5% after final rectification, meeting the stringent specifications required for high-purity OLED material and battery electrolyte applications.
How to Synthesize N,N-Dimethyl Trifluoromethyl Sulfonamide Efficiently
The operational protocol for this synthesis is designed to maximize yield while minimizing waste through a carefully controlled temperature profile and reagent addition sequence. The process begins by dissolving dimethylamine in the chosen alcohol solvent at low temperatures to form a stable solution, followed by the controlled addition of the sulfonyl halide to manage exothermicity. Once the primary substitution is complete, the critical alkoxide addition step is performed, still under cooling, to ensure the complete conversion of the byproduct salt without triggering side reactions.
- Dissolve dimethylamine in an alcohol solvent (e.g., ethanol, methanol) at -15 to -30°C to form a solution, then add trifluoromethyl sulfonyl halide while maintaining low temperature.
- After the initial reaction completes, add the corresponding alkoxide (e.g., sodium ethoxide) to decompose the dimethylamine hydrogen halide byproduct, then filter to separate solid salts.
- Distill the filtrate at normal pressure to recover dimethylamine and alcohol solvent for reuse, then rectify the residue under vacuum to obtain high-purity N,N-dimethyl trifluoromethyl sulfonamide.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented synthesis route offers transformative benefits that extend far beyond simple chemical yield improvements. The ability to recycle both the dimethylamine reactant and the alcohol solvent fundamentally alters the cost structure of production, turning what was once a linear consumption model into a circular economy within the reactor. This internal recycling capability significantly mitigates the impact of raw material price volatility, providing a buffer against market fluctuations that often plague the fine chemical sector. Moreover, the elimination of ultra-low temperature requirements reduces the dependency on specialized cryogenic equipment, lowering both capital expenditure for new plants and maintenance costs for existing facilities, thereby enhancing overall supply chain resilience.
- Cost Reduction in Manufacturing: The economic advantage of this process is driven primarily by the near-quantitative recovery of starting materials. By chemically regenerating dimethylamine from the byproduct salt using alkoxides, the net consumption of this key reagent is drastically lowered, leading to substantial cost savings over time. Additionally, the use of inexpensive, commodity-grade alcohol solvents instead of specialized ethers, combined with the simplified filtration step that replaces complex extractions, reduces the operational expenditure associated with solvent purchase and waste disposal. The removal of the need for -78°C cooling further slashes energy costs, making the overall manufacturing process significantly more lean and profitable.
- Enhanced Supply Chain Reliability: From a logistics and planning perspective, the robustness of this method ensures more predictable production schedules. The milder reaction conditions (-15°C to 0°C) are easier to maintain consistently across different batches and scales compared to the finicky -78°C protocols, reducing the risk of batch failures due to temperature excursions. Furthermore, the generation of solid inorganic salts as byproducts, which are easily filtered and disposed of, simplifies waste management compliance and prevents the bottlenecks often caused by difficult liquid-liquid separations. This operational stability translates directly into shorter lead times and a more dependable supply of high-purity intermediates for downstream battery manufacturers.
- Scalability and Environmental Compliance: The design of this synthesis is inherently scalable, moving seamlessly from laboratory glassware to industrial reactors without the engineering challenges posed by cryogenic gas-phase reactions. The process generates minimal hazardous waste, as the primary byproducts are benign inorganic salts and recovered solvents, aligning perfectly with modern green chemistry principles and strict environmental regulations. This eco-friendly profile not only reduces the burden on wastewater treatment facilities but also enhances the corporate sustainability metrics of the manufacturer, a critical factor for multinational corporations aiming to reduce their carbon footprint in the production of advanced battery materials.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis technology. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation, highlighting the practical advantages for industrial adoption.
Q: How does the new alcohol-based method improve product purity compared to traditional ether-based methods?
A: The novel method utilizes an alkoxide treatment step that decomposes the byproduct dimethylamine hydrogen halide back into reusable dimethylamine and inorganic salts. This prevents the thermal decomposition of the byproduct during distillation, which typically contaminates the product in older methods, thereby achieving purity levels exceeding 99.5%.
Q: What are the primary cost-saving mechanisms in this synthesis route?
A: The process significantly reduces costs by enabling the efficient recovery and recycling of both the dimethylamine reactant and the alcohol solvent. Additionally, the elimination of ultra-low temperature requirements (-78°C) reduces energy consumption and equipment complexity.
Q: Is this synthesis method suitable for large-scale industrial production?
A: Yes, the method is specifically designed for industrial scalability. It operates at milder temperatures (-15 to 0°C), uses common alcohol solvents, and simplifies post-treatment by converting corrosive byproducts into filterable inorganic salts, making it highly robust for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N,N-Dimethyl Trifluoromethyl Sulfonamide Supplier
As the global demand for safer and more efficient energy storage solutions accelerates, the need for high-quality electrolyte additives has never been more critical. NINGBO INNO PHARMCHEM stands at the forefront of this industry, leveraging advanced synthetic methodologies like the one described in CN115703722A to deliver superior chemical solutions. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the rigorous volume requirements of major battery manufacturers. We are committed to maintaining stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of N,N-dimethyl trifluoromethyl sulfonamide meets the exacting standards necessary for next-generation lithium battery performance.
We invite potential partners to engage with our technical procurement team to discuss how our optimized manufacturing processes can support your specific project needs. By collaborating with us, you gain access to a Customized Cost-Saving Analysis that demonstrates the tangible economic benefits of our efficient production routes. We encourage you to contact us today to request specific COA data and route feasibility assessments, allowing you to validate our capabilities and secure a stable, high-quality supply chain for your critical electronic chemical requirements.
