Revolutionizing HIV Protease Inhibitor Intermediate Production via Advanced Lanthanide Catalysis
The pharmaceutical landscape for antiretroviral therapies continues to evolve, driven by the urgent need for more efficient and safer manufacturing processes for HIV protease inhibitors. Patent CN101448838B introduces a groundbreaking methodology for the synthesis of difuryl alcohol intermediates, which are critical precursors in the production of potent antiviral compounds. This technology addresses long-standing challenges in the field, specifically targeting the inefficiencies and safety hazards associated with legacy synthetic routes. By leveraging lanthanide and transition metal catalysis, the disclosed process offers a pathway to high-purity intermediates that align with modern green chemistry principles. For R&D directors and procurement specialists, understanding this shift is vital, as it represents a move away from stoichiometric toxic reagents toward catalytic precision, ultimately enhancing the viability of large-scale antiviral drug production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of the requisite difuryl alcohol scaffolds, such as those described by Ghosh et al., has been plagued by significant operational and safety drawbacks. The conventional reliance on tributyltin hydride and ozone presents severe environmental and health risks, necessitating complex waste management protocols that inflate operational costs. Furthermore, these traditional methods are characterized by notoriously low yields, often hovering around 1%, which is economically unsustainable for commercial manufacturing. The requirement for extensive chromatographic purification steps not only slows down production throughput but also introduces variability in the final product quality. For supply chain heads, these factors translate into unpredictable lead times and increased vulnerability to regulatory scrutiny regarding residual heavy metals and toxic byproducts in the active pharmaceutical ingredients.
The Novel Approach
In stark contrast, the methodology outlined in CN101448838B utilizes a catalytic system involving lanthanides such as Ytterbium (Yb), Scandium (Sc), or Copper (Cu) to facilitate the cyclization of 2,3-dihydrofuran and glycolaldehyde dimers. This approach fundamentally alters the reaction kinetics, allowing for the direct formation of the difuryl alcohol core under milder conditions, typically around 50°C. The elimination of ozone and tin hydrides simplifies the safety profile of the plant operations significantly. Moreover, the integration of enzymatic resolution using immobilized lipases, such as Lipase PS-C, provides a highly selective means of isolating the desired chiral enantiomers without the need for cumbersome chiral chromatography. This combination of metal catalysis and biocatalysis creates a robust, scalable process that dramatically improves the overall mass balance and economic feasibility of producing these complex intermediates.
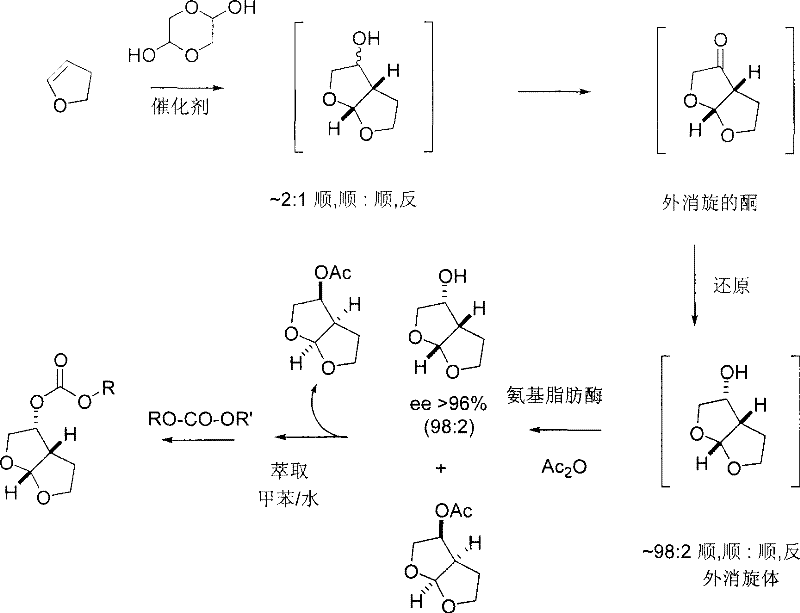
Mechanistically, the success of this novel route lies in the Lewis acidic nature of the lanthanide catalysts, which effectively activate the carbonyl species of the glycolaldehyde dimer towards nucleophilic attack by the dihydrofuran. This activation lowers the energy barrier for the cycloaddition, enabling high conversion rates even at moderate temperatures. The subsequent enzymatic kinetic resolution is equally critical; by selectively acetylating one enantiomer over the other, the process achieves optical purities exceeding 97% ee, as demonstrated in the patent examples. This level of stereocontrol is essential for the biological activity of the final HIV protease inhibitor, ensuring that the downstream synthesis proceeds with minimal formation of inactive or potentially harmful diastereomers. The ability to control impurity profiles at such an early stage reduces the burden on later purification steps, streamlining the entire manufacturing workflow.
How to Synthesize HIV Protease Inhibitor Intermediates Efficiently
The practical implementation of this synthesis involves a sequence of well-defined steps that balance chemical reactivity with operational simplicity. The process begins with the catalytic coupling of readily available starting materials to form the racemic alcohol, followed by a biocatalytic resolution to secure the correct stereochemistry. Subsequent functionalization involves coupling with protected amino acid derivatives and final salt formation to ensure stability. The detailed standardized synthesis steps for replicating this high-efficiency route are provided in the guide below, offering a clear roadmap for technical teams aiming to adopt this superior methodology.
- React 2,3-dihydrofuran with glycolaldehyde dimer in the presence of a lanthanide catalyst (e.g., Yb, Sc) to form the difuryl alcohol core.
- Perform enzymatic resolution using immobilized lipase (e.g., Lipase PS-C) to isolate the desired chiral enantiomer with high optical purity.
- Couple the resolved alcohol with protected amino acid derivatives and finalize with salt formation (e.g., adipic acid) to obtain the stable API intermediate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of the technology described in CN101448838B offers tangible strategic benefits beyond mere chemical elegance. The primary advantage lies in the substantial reduction of raw material costs and waste disposal expenses associated with removing toxic tin residues and managing ozone generation. By shifting to a catalytic regime, the process minimizes the consumption of expensive reagents and reduces the volume of hazardous waste, leading to significant cost reduction in antiviral pharmaceutical manufacturing. Furthermore, the robustness of the enzymatic resolution step ensures consistent product quality, which is a critical factor in maintaining reliable supply chains for life-saving medications. This consistency reduces the risk of batch failures and the associated delays that can disrupt global drug availability.
- Cost Reduction in Manufacturing: The elimination of stoichiometric toxic reagents like tributyltin hydride removes the need for costly heavy metal scavenging and specialized waste treatment protocols. This qualitative shift in the process chemistry directly lowers the cost of goods sold (COGS) by simplifying the downstream processing requirements. Additionally, the higher yields achieved through lanthanide catalysis mean that less starting material is required to produce the same amount of active intermediate, further optimizing the raw material budget and improving overall process economics without compromising on quality standards.
- Enhanced Supply Chain Reliability: The use of stable, commercially available lanthanide catalysts and immobilized enzymes reduces dependency on hazardous or hard-to-source reagents that often face supply constraints. This diversification of the supply base enhances the resilience of the manufacturing process against external market shocks. The simplified workflow, which avoids complex cryogenic conditions or dangerous ozonolysis steps, allows for more flexible scheduling and faster turnaround times, effectively reducing lead time for high-purity HIV protease inhibitor intermediates and ensuring a steady flow of materials to formulation sites.
- Scalability and Environmental Compliance: The patent data demonstrates successful execution at the kilogram scale, indicating that the process is ready for commercial scale-up of complex antiviral intermediates. The avoidance of ozone and tin byproducts aligns the manufacturing process with increasingly stringent environmental regulations, mitigating the risk of compliance-related shutdowns. This environmental compatibility not only safeguards the company's reputation but also facilitates smoother regulatory approvals in key markets, ensuring long-term viability and uninterrupted production capacity for essential HIV therapies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced synthetic route. These insights are derived directly from the experimental data and embodiments disclosed in the patent, providing a factual basis for decision-making. Understanding these nuances helps stakeholders evaluate the feasibility of integrating this technology into existing production lines and assess its impact on overall operational efficiency and product quality assurance.
Q: How does the new catalytic method improve upon traditional Ghosh synthesis?
A: The traditional Ghosh method relies on toxic tributyltin hydride and ozone, yielding poor results (~1%) with difficult purification. The new patent method utilizes lanthanide catalysts (Yb, Sc) for higher efficiency and avoids hazardous reagents.
Q: What represents the key advantage for supply chain stability?
A: By eliminating the need for ozonolysis and tin-based reagents, the process reduces safety risks and regulatory hurdles, ensuring more consistent batch-to-batch production and reliable delivery schedules.
Q: Is this process suitable for commercial scale-up?
A: Yes, the patent describes kilogram-scale examples (e.g., 4.45 kg glycolaldehyde dimer) demonstrating that the catalytic cyclization and enzymatic resolution steps are robust enough for industrial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable HIV Protease Inhibitor Intermediates Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting cutting-edge synthetic methodologies to meet the demands of the global pharmaceutical market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless. We are committed to delivering high-purity HIV protease inhibitor intermediates that meet stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities. Our expertise in handling complex catalytic systems and biocatalytic resolutions positions us as a strategic partner capable of navigating the intricacies of modern antiviral drug synthesis.
We invite you to collaborate with us to leverage these technological advancements for your supply chain. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific production needs. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our optimized processes can enhance your operational efficiency and secure your supply of essential pharmaceutical intermediates.
