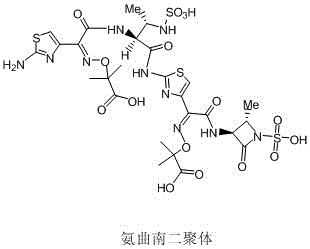
Advanced Synthesis of Aztreonam Dimer Impurity for Pharmaceutical Quality Control
The pharmaceutical industry faces relentless pressure to ensure the absolute safety and efficacy of beta-lactam antibiotics, particularly regarding the control of polymeric impurities that can trigger severe immunogenic responses. Patent CN114437059A introduces a groundbreaking preparation method for Aztreonam Dimer, a critical impurity reference standard required for the rigorous quality control of Aztreonam bulk drugs. This innovation addresses the longstanding bottleneck in obtaining high-purity dimer standards, which are essential for validating the safety profile of this widely used monobactam antibiotic. By shifting from traditional, time-consuming isolation methods to a directed synthetic approach, this technology offers a robust pathway for manufacturers to secure reliable supply chains for essential analytical standards. The method leverages readily available intermediates to construct the complex dimer structure with exceptional efficiency, marking a significant leap forward in medicinal chemistry process development.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the acquisition of Aztreonam Dimer impurities has been plagued by inefficient and labor-intensive processes that struggle to meet modern Good Manufacturing Practice (GMP) standards for reference materials. Conventional literature methods typically involve incubating Aztreonam solutions at specific pH levels for extended periods ranging from 60 to 80 hours, followed by arduous column chromatography for separation. This approach not only suffers from notoriously low product yields but also presents significant challenges in purification, often failing to achieve the high purity levels required for accurate quantitative analysis. The reliance on column chromatography is particularly detrimental in an industrial setting, as it limits batch sizes, consumes vast quantities of solvents, and introduces variability that complicates the standardization of impurity profiles. Consequently, pharmaceutical quality control laboratories have faced difficulties in sourcing consistent, high-quality dimer standards necessary for regulatory compliance.
The Novel Approach
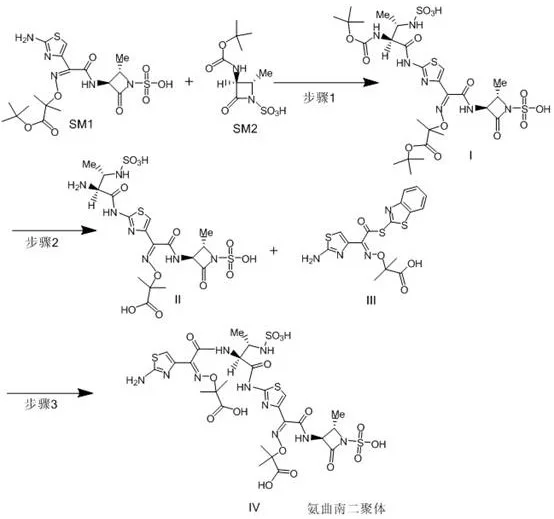
In stark contrast, the novel synthetic route disclosed in the patent utilizes a rational, step-wise construction of the dimer molecule that drastically improves both throughput and product quality. By employing Aztreonam tert-butyl ester and a BOC-protected Aztreonam main ring as starting materials, the process achieves a directed synthesis that bypasses the randomness of degradation-based methods. The entire three-step sequence operates under mild conditions, primarily at room temperature (20-35°C), which preserves the integrity of the sensitive beta-lactam rings while facilitating rapid conversion. Most critically, the purification strategy shifts from complex chromatography to simple precipitation and pulping techniques, enabling the isolation of the target compound with a purity exceeding 97%. This methodological shift transforms the production of impurity standards from a boutique laboratory exercise into a scalable, industrial-grade operation.
Mechanistic Insights into Directed Dimerization and Deprotection
The core of this technological advancement lies in the precise orchestration of coupling and deprotection reactions that mimic the natural dimerization process but with controlled stereochemistry and yield. The first step involves the nucleophilic interaction between the tert-butyl ester derivative and the protected main ring under basic catalysis, forming a stable intermediate (Compound I) that serves as the scaffold for the dimer. This is followed by a carefully controlled acid hydrolysis step at 20-30°C, which selectively removes the protecting groups without degrading the fragile beta-lactam core, yielding the key intermediate Compound II. The final condensation with the active ester (Compound III) completes the dimer structure, leveraging the reactivity of the activated carboxyl group to form the peptide-like linkage characteristic of the impurity. Each step is optimized to minimize side reactions, ensuring that the final product closely matches the structural requirements for an authentic reference standard.
Furthermore, the mechanism inherently supports superior impurity control by avoiding the harsh conditions that typically generate complex, undefined byproduct mixtures. In traditional degradation methods, the formation of the dimer is accompanied by a plethora of other hydrolysis products and stereoisomers that are difficult to separate. By building the molecule from defined precursors, this new route ensures that the impurity profile is clean and predictable, simplifying the downstream purification process. The use of specific solvents like methanol and bases like triethylamine is not arbitrary; these reagents are selected to maximize solubility during reaction while facilitating easy precipitation upon completion. This mechanistic clarity allows process chemists to troubleshoot and optimize the reaction parameters with confidence, ensuring batch-to-batch consistency that is vital for regulatory submissions.
How to Synthesize Aztreonam Dimer Efficiently
The synthesis of Aztreonam Dimer via this patented route is designed for operational simplicity, allowing chemical manufacturers to transition from bench-scale experiments to pilot production with minimal friction. The process eliminates the need for specialized chromatography equipment, relying instead on standard reactor vessels and filtration units found in most fine chemical facilities. Operators can expect a streamlined workflow where reaction monitoring is straightforward, and work-up procedures involve basic solvent exchanges and crystallization steps. For detailed technical execution, the standardized synthesis steps are outlined below to guide your process development team.
- React Aztreonam tert-butyl ester (SM1) with BOC-protected Aztreonam main ring (SM2) under alkaline conditions at 25-35°C to obtain Compound I.
- Hydrolyze Compound I using acid at 20-30°C to remove protecting groups and yield Compound II.
- Condense Compound II with Ceftazidime acid active ester (Compound III) under alkaline conditions at 20-30°C to finalize the Aztreonam Dimer (Compound IV).
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthesis method represents a strategic opportunity to reduce costs and mitigate supply risks associated with critical quality control materials. The elimination of column chromatography is not merely a technical improvement; it is a major economic driver that significantly lowers the cost of goods sold (COGS) by reducing solvent consumption and labor hours. Traditional methods that require days of column running are inherently expensive and difficult to scale, whereas this precipitation-based approach allows for larger batch sizes and faster turnaround times. This efficiency translates directly into a more reliable supply of reference standards, ensuring that quality control laboratories never face bottlenecks due to material shortages.
- Cost Reduction in Manufacturing: The shift away from preparative chromatography results in substantial cost savings by removing one of the most resource-intensive unit operations in fine chemical synthesis. Without the need for expensive silica gel, large volumes of HPLC-grade solvents, and the associated waste disposal costs, the overall production expense is drastically simplified. Furthermore, the high yield of each step (consistently above 75%) means that raw material utilization is optimized, reducing the effective cost per gram of the final high-purity product. These factors combine to create a leaner manufacturing process that offers better margin potential for suppliers and lower costs for buyers.
- Enhanced Supply Chain Reliability: The reliance on readily available starting materials such as Aztreonam tert-butyl ester and BOC-protected intermediates ensures that the supply chain is robust and less susceptible to disruptions. Unlike methods that depend on the unpredictable degradation of the final API, this synthetic route uses stable, storable precursors that can be sourced from multiple vendors. The shortened reaction time, dropping from nearly four days to less than one day, also enhances agility, allowing manufacturers to respond quickly to sudden spikes in demand for quality control standards without maintaining excessive inventory levels.
- Scalability and Environmental Compliance: From an environmental and safety perspective, the mild reaction conditions (20-35°C) and the absence of heavy metal catalysts or hazardous reagents make this process highly compliant with modern green chemistry principles. The ability to scale up using simple filtration and pulping techniques means that production can be increased from kilograms to tons without requiring massive capital investment in new infrastructure. This scalability ensures long-term supply continuity, while the reduced solvent load simplifies waste treatment, aligning with the increasingly strict environmental regulations governing pharmaceutical intermediate manufacturing.
Frequently Asked Questions (FAQ)
To assist technical decision-makers in evaluating this technology, we have compiled answers to common questions regarding the implementation and benefits of this synthesis method. These insights are derived directly from the patent data and practical experience in scaling similar beta-lactam processes. Understanding these details is crucial for integrating this new standard into your existing quality assurance frameworks.
Q: What is the primary advantage of this new Aztreonam Dimer synthesis method?
A: The primary advantage is the significant reduction in reaction time from 60-80 hours in conventional literature methods to just 14-22 hours, alongside achieving a purity greater than 97% without the need for complex column chromatography.
Q: Why is high-purity Aztreonam Dimer critical for pharmaceutical manufacturing?
A: As polymer impurities in beta-lactam antibiotics can cause severe adverse reactions, regulatory bodies require accurate characterization. High-purity dimer standards are essential for validating the safety and quality of the final Aztreonam API.
Q: Is this synthesis method suitable for large-scale industrial production?
A: Yes, the method utilizes mild room-temperature conditions and simple purification techniques like pulping and filtration, making it highly scalable and cost-effective for industrial reference substance production compared to labor-intensive column separation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aztreonam Dimer Supplier
At NINGBO INNO PHARMCHEM, we recognize that the availability of high-quality impurity standards is the cornerstone of pharmaceutical safety and regulatory compliance. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements with consistent quality. We operate stringent purity specifications and maintain rigorous QC labs equipped to verify the structural integrity and purity of complex molecules like the Aztreonam Dimer. Our commitment to technical excellence means that every batch we produce is backed by comprehensive analytical data, giving you the confidence to proceed with your drug development and quality control programs.
We invite you to collaborate with us to leverage this advanced synthesis technology for your specific needs. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your projected volumes. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can support your supply chain goals efficiently and reliably.
