Advancing Pharmaceutical Intermediate Manufacturing with Efficient N-Cyclopropyl Nitrone Synthesis
The landscape of organic synthesis for bioactive molecules is constantly evolving, driven by the need for more efficient and accessible pathways to complex scaffolds. A significant breakthrough in this domain is documented in Chinese Patent CN107522635B, which discloses a novel class of N-cyclopropyl nitrone derivatives and a robust preparation method thereof. Nitrones are pivotal functional groups in medicinal chemistry, serving as key intermediates for 1,3-dipolar cycloadditions and possessing intrinsic biological activity against neurodegenerative diseases and tumors. However, the historical challenge has been the difficulty in introducing the N-cyclopropyl motif, traditionally requiring complex cyclopropylamine precursors. This patent introduces a paradigm shift by utilizing a copper-catalyzed direct functionalization strategy that merges cyclopropenes with oximes. This approach not only streamlines the synthetic route but also opens new avenues for the rapid generation of diverse chemical libraries for drug discovery programs.
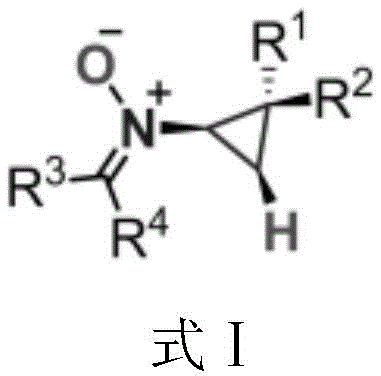
For research and development directors focusing on process chemistry, understanding the limitations of legacy methods is crucial for evaluating new technologies. Conventional routes to N-alkyl nitrones often rely on the condensation of hydroxylamines with carbonyl compounds, but introducing a cyclopropyl group specifically has been notoriously difficult due to the instability and synthetic inaccessibility of substituted cyclopropylamines. Traditional methods often suffer from harsh reaction conditions, poor atom economy, and limited substrate scope, particularly when dealing with sensitive functional groups. In contrast, the novel approach detailed in the patent utilizes a transition metal-catalyzed coupling that proceeds under remarkably mild conditions. By reacting cyclopropene compounds directly with oximes in the presence of a copper catalyst and specific ligands, the method achieves high conversion rates without the need for extreme temperatures or pressures. This represents a substantial improvement in operational simplicity and safety, allowing chemists to access previously difficult-to-synthesize ketone nitrones with greater efficiency.
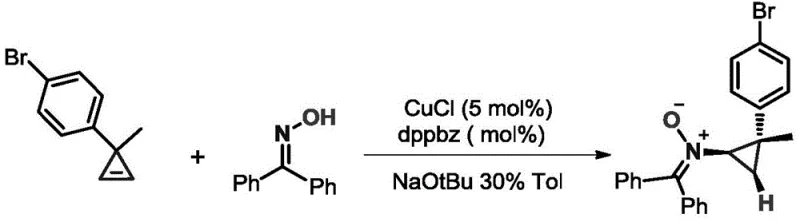
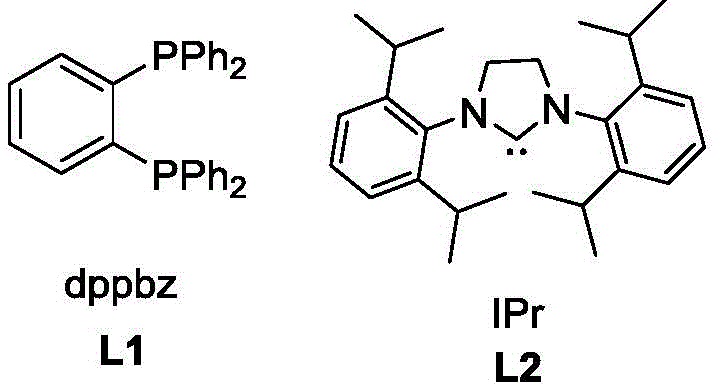
The mechanistic insights into this copper-catalyzed cyclopropenylation reveal a sophisticated interplay between the metal center and the ligand environment. The reaction likely proceeds through the formation of a copper-carbenoid or copper-nitrenoid species generated from the interaction of the copper catalyst with the strained cyclopropene ring. The choice of ligand plays a critical role in stabilizing the active catalytic species and controlling the regioselectivity of the addition to the oxime nitrogen. The patent highlights the efficacy of both phosphine ligands like dppbz and N-heterocyclic carbenes like IPr, suggesting a versatile catalytic manifold capable of accommodating electronic variations in the substrates. This mechanistic robustness is evidenced by the broad tolerance for various substituents on both the cyclopropene and the oxime, including electron-withdrawing and electron-donating groups on aromatic rings. Furthermore, the mild conditions help preserve the integrity of sensitive functional groups, minimizing the formation of side products and simplifying the impurity profile, which is a critical consideration for pharmaceutical intermediate manufacturing.
Mechanistic Insights into Copper-Catalyzed Cyclopropenylation
The catalytic cycle involves the activation of the cyclopropene by the copper-ligand complex, facilitating the nucleophilic attack by the oxime nitrogen. The strain energy of the three-membered ring in the cyclopropene serves as a driving force for the reaction, allowing it to proceed rapidly even at room temperature. The use of bases such as sodium tert-butoxide aids in the deprotonation steps necessary for the turnover of the catalytic cycle. This precise control over the reaction pathway ensures that the N-O bond of the nitrone is formed selectively, avoiding competing reactions that might lead to ring-opening products or polymerization. The ability to tune the electronic properties of the catalyst system by switching between ligands L1 and L2 provides a powerful tool for optimizing yields for specific substrate combinations, ensuring high purity and consistency in the final product.
How to Synthesize N-Cyclopropyl Nitrone Derivatives Efficiently
The synthesis protocol outlined in the patent offers a straightforward procedure that is amenable to standard laboratory and pilot plant equipment. The process begins with the preparation of the catalytic system in an organic solvent, typically toluene, ensuring that all reagents are thoroughly mixed before the addition of the reactive cyclopropene component. The reaction is monitored using thin-layer chromatography to determine the endpoint, which is typically reached within a short timeframe of one to two hours. Following the reaction, standard workup procedures involving column chromatography allow for the isolation of the target nitrone derivatives in high purity. For detailed operational parameters and specific stoichiometric ratios required for different substrate classes, refer to the standardized synthesis guide below.
- Prepare the reaction vessel by adding an organic solvent such as toluene, followed by the sequential addition of the copper catalyst (e.g., CuCl), the specific ligand (dppbz or IPr), the base (e.g., NaOtBu), and the oxime substrate.
- Slowly add the cyclopropene compound to the reaction mixture while maintaining the system at room temperature to ensure controlled reactivity and safety.
- Stir the reaction mixture for 1 to 2 hours, monitoring progress via TLC, then proceed to column chromatography for purification to isolate the high-purity N-cyclopropyl nitrone derivative.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this technology offers compelling advantages that address common pain points in the sourcing of complex intermediates. The reliance on inexpensive and commercially available starting materials, such as cuprous chloride and various oximes, significantly reduces the raw material cost burden compared to methods requiring exotic reagents. The elimination of difficult-to-source cyclopropylamine precursors removes a major bottleneck in the supply chain, ensuring greater continuity and reliability of supply for downstream manufacturing processes. Additionally, the mild reaction conditions translate to lower energy consumption and reduced safety risks, which are critical factors in maintaining a sustainable and compliant production environment.
- Cost Reduction in Manufacturing: The use of earth-abundant copper catalysts instead of precious metals like palladium or rhodium drastically lowers the catalyst cost per kilogram of product. Furthermore, the high conversion rates and selectivity minimize waste generation and reduce the complexity of purification, leading to substantial overall cost savings in the manufacturing process. The avoidance of expensive protecting group strategies or multi-step sequences further contributes to a leaner and more cost-effective production model.
- Enhanced Supply Chain Reliability: By utilizing widely available commodity chemicals as starting materials, the risk of supply disruption is significantly mitigated. The robustness of the reaction conditions means that the process is less susceptible to variations in raw material quality or minor fluctuations in processing parameters, ensuring consistent output. This reliability is essential for long-term supply agreements and helps stabilize the inventory management for critical pharmaceutical intermediates.
- Scalability and Environmental Compliance: The simplicity of the reaction setup and the use of common solvents facilitate easy scale-up from laboratory to commercial production volumes. The green chemistry attributes, such as high atom economy and reduced hazardous waste, align well with increasingly stringent environmental regulations. This ensures that the manufacturing process remains compliant with global standards, reducing the regulatory burden and potential liabilities associated with chemical production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on the practical application of the method. Understanding these details helps stakeholders make informed decisions regarding the adoption of this route for their specific project needs.
Q: What are the primary advantages of this copper-catalyzed method over traditional nitrone synthesis?
A: This method bypasses the difficult synthesis of substituted cyclopropylamines, utilizing readily available cyclopropenes and oximes instead. It operates under mild room temperature conditions with high conversion rates, significantly simplifying the process compared to harsh traditional methods.
Q: Which ligands are most effective for this transformation according to the patent data?
A: The patent identifies two highly effective ligand systems: the phosphine ligand dppbz and the N-heterocyclic carbene IPr. These ligands enable the copper catalyst to achieve excellent yields and broad substrate tolerance across various aromatic and heteroaromatic systems.
Q: Is this synthesis method suitable for large-scale industrial production?
A: Yes, the use of inexpensive catalysts like cuprous chloride, combined with mild reaction conditions and short reaction times, makes this process highly scalable. The simplicity of the workup and purification further supports its viability for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Cyclopropyl Nitrone Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of advanced synthetic methodologies like the copper-catalyzed cyclopropenylation described in CN107522635B. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory discoveries are successfully translated into reliable industrial realities. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs that utilize state-of-the-art analytical instrumentation to verify the identity and purity of every batch, guaranteeing that our clients receive materials that meet the highest industry standards.
We invite pharmaceutical and agrochemical companies to leverage our expertise for the commercialization of these valuable nitrone intermediates. By partnering with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our capabilities can accelerate your development timelines and optimize your supply chain efficiency.
