Advanced Low-Temperature Synthesis of Dibenzoylmethane for Industrial Polymer Stabilization
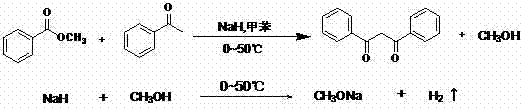
The global demand for high-performance thermoplastic stabilizers continues to surge, driven by the expanding applications of polyvinyl chloride (PVC) in medical, food packaging, and construction sectors. At the forefront of this market is Dibenzoylmethane (DBM), a critical beta-diketone compound known for its exceptional ability to improve the thermal stability and transparency of PVC resins. Patent CN103483172A introduces a groundbreaking preparation method that addresses long-standing inefficiencies in DBM manufacturing. By utilizing acetophenone and methyl benzoate as raw materials under the catalytic influence of sodium hydride, this technology enables the synthesis to proceed at remarkably mild temperatures ranging from 0 to 50°C. This innovation represents a significant leap forward for any reliable polymer additive supplier seeking to optimize production metrics while maintaining the highest standards of product quality.
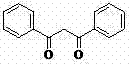
The molecular architecture of Dibenzoylmethane, characterized by its 1,3-dicarbonyl system, is fundamental to its function as a UV absorber and heat stabilizer. As illustrated in the structural representation, the conjugated system allows for effective chelation with metal ions, preventing degradation in polymer matrices. The patent highlights that this specific synthetic route not only ensures the integrity of this delicate structure but also enhances the economic viability of the manufacturing process. For procurement managers and supply chain heads, understanding the nuances of this synthesis is key to securing a stable source of high-purity dibenzoylmethane that meets international regulatory standards for non-toxic applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of dibenzoylmethane has been plagued by harsh reaction conditions that compromise both efficiency and product quality. Traditional methodologies often necessitate elevated reaction temperatures to drive the Claisen condensation forward, which inadvertently leads to a decrease in catalyst activity over time. These high-thermal environments promote the formation of undesirable side products and impurities, complicating the downstream purification process and reducing the overall yield. Furthermore, conventional routes frequently require the energy-intensive distillation of methanol byproducts to shift the equilibrium, adding significant operational costs and extending the production cycle. For a cost reduction in plastic stabilizer manufacturing strategy, these legacy methods present substantial bottlenecks that limit scalability and increase the carbon footprint of the final product.
The Novel Approach
In stark contrast, the methodology described in patent CN103483172A leverages the unique properties of sodium hydride to revolutionize the synthesis landscape. By conducting the condensation reaction in a toluene solvent at a controlled temperature range of 0 to 50°C, this novel approach effectively mitigates the thermal degradation issues associated with older techniques. The use of sodium hydride serves a dual purpose: it acts as a potent base to initiate the condensation and simultaneously reacts with the generated methanol to form sodium methoxide, which further catalyzes the reaction. This ingenious mechanism eliminates the need for high-temperature methanol removal, thereby simplifying the operational workflow and promoting the reaction towards the desired product with greater fidelity. The result is a streamlined process that delivers superior yields and purity, positioning it as an ideal solution for the commercial scale-up of complex polymer additives.
Mechanistic Insights into Sodium Hydride-Catalyzed Condensation
The core of this technological advancement lies in the sophisticated mechanistic pathway facilitated by sodium hydride (NaH). Unlike weaker bases that might struggle to deprotonate the alpha-carbon of acetophenone efficiently at low temperatures, NaH provides the necessary superbasicity to generate the enolate species rapidly. Once formed, this enolate attacks the carbonyl carbon of methyl benzoate, initiating the formation of the beta-diketone skeleton. Crucially, the patent elucidates that the methanol produced during this elimination step does not act as an inhibitor; instead, it reacts immediately with the remaining sodium hydride to produce sodium methoxide and hydrogen gas. This in-situ generation of a secondary catalyst creates a synergistic effect that maintains high reaction rates even under the mild thermal conditions of 0 to 50°C. This self-sustaining catalytic cycle ensures that the reaction proceeds to completion without the need for external driving forces like excessive heat or vacuum distillation.
From an impurity control perspective, this mechanism offers distinct advantages for producing high-purity dibenzoylmethane. The mild reaction temperature suppresses competing side reactions such as self-condensation of acetophenone or hydrolysis of the ester, which are common at higher temperatures. The evolution of hydrogen gas, which is easily collected and vented, serves as a visual indicator of reaction progress and catalyst consumption, allowing for precise process control. Following the reaction, the workup procedure involves a meticulous sequence of acid washing with hydrochloric acid, alkali washing with sodium bicarbonate, and water washing. This rigorous purification protocol effectively removes residual catalysts, unreacted starting materials, and inorganic salts. The final recrystallization step, performed in solvents like methanol or ethanol at temperatures between -10 and 25°C, ensures that the final crystalline powder achieves a purity of 99.8%, meeting the stringent specifications required for sensitive applications in food packaging and medical devices.
How to Synthesize Dibenzoylmethane Efficiently
The synthesis of dibenzoylmethane via this patented route is designed for operational simplicity and robustness, making it highly suitable for industrial implementation. The process begins with the careful charging of dry toluene and sodium hydride into a reactor under an inert nitrogen atmosphere to prevent moisture interference. Methyl benzoate is added, followed by the controlled dropwise addition of acetophenone while maintaining the temperature within the optimal 0 to 50°C window. The detailed standardized synthesis steps below outline the precise stoichiometry, mixing rates, and workup parameters required to replicate the high yields reported in the patent documentation.
- Charge toluene solvent and sodium hydride into a dry reactor under nitrogen protection, followed by the addition of methyl benzoate.
- Maintain temperature between 0-50°C while dropwise adding acetophenone, allowing hydrogen gas to evolve and escape safely.
- Quench the reaction with acid, perform alkali and water washes, recover solvent, and recrystallize the crude product to achieve 99.8% purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this sodium hydride-catalyzed process translates into tangible strategic benefits that extend beyond simple chemistry. The shift to mild reaction conditions fundamentally alters the cost structure and risk profile of dibenzoylmethane production. By eliminating the need for high-temperature operations and complex distillation setups for methanol removal, the process significantly reduces industrial energy consumption. This reduction in thermal load not only lowers utility costs but also decreases the wear and tear on reactor vessels and associated equipment, leading to reduced maintenance downtime and extended asset life. Furthermore, the simplified operational steps mean that batch cycles can be completed more rapidly, enhancing the overall throughput of the manufacturing facility without requiring additional capital investment in new hardware.
- Cost Reduction in Manufacturing: The economic implications of this technology are profound, primarily driven by the elimination of expensive and energy-intensive processing steps. Traditional methods often require significant energy input to distill off methanol to drive the equilibrium; however, this novel approach utilizes the methanol byproduct to generate additional catalyst in situ. This clever chemical design removes the necessity for high-vacuum distillation equipment and the associated steam or electrical costs. Additionally, the high selectivity of the reaction minimizes the formation of byproducts, which reduces the volume of waste streams that require treatment and disposal. The combination of lower energy usage, reduced waste management costs, and higher overall yield creates a compelling value proposition for cost reduction in plastic stabilizer manufacturing, allowing suppliers to offer competitive pricing without compromising on margin.
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by complex processes that are prone to failure or variability. The robustness of this low-temperature synthesis enhances reliability by operating within a wide and manageable temperature window of 0 to 50°C. This flexibility reduces the risk of thermal runaways or equipment failures that can halt production lines. Moreover, the raw materials—acetophenone and methyl benzoate—are commodity chemicals with stable global availability, ensuring that production is not held hostage by niche precursor shortages. The ability to consistently produce off-white crystalline powder with a purity of 99.8% means that downstream customers receive a uniform product every time, reducing the need for incoming quality inspections and minimizing the risk of production stoppages at the client's end due to material variability.
- Scalability and Environmental Compliance: As regulatory pressures regarding environmental impact intensify, the scalability of this process offers a clear advantage. The reaction generates hydrogen gas as the only gaseous byproduct, which is easily managed and collected, avoiding the release of volatile organic compounds (VOCs) associated with high-temperature solvent losses. The mild conditions also align well with green chemistry principles by reducing the overall energy intensity of the manufacture. Scaling this process from laboratory benchtop to multi-ton commercial production is straightforward because it does not rely on exotic catalysts or extreme pressures that pose engineering challenges at scale. This ease of scale-up ensures that suppliers can rapidly respond to surges in market demand for high-purity dibenzoylmethane, securing the supply chain for critical industries like healthcare and food packaging.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of dibenzoylmethane using this advanced methodology. These insights are derived directly from the experimental data and beneficial effects outlined in the patent literature, providing a transparent view of the technology's capabilities. Understanding these details helps stakeholders make informed decisions about integrating this material into their supply chains.
Q: What are the primary advantages of using Sodium Hydride over traditional catalysts for DBM synthesis?
A: Sodium Hydride acts as a superbases catalyst that not only drives the condensation but also reacts with the methanol byproduct to generate sodium methoxide in situ. This dual catalytic effect eliminates the need for high-temperature methanol distillation, significantly shortening reaction time and improving overall yield.
Q: How does this process ensure high purity suitable for food-grade PVC applications?
A: The process utilizes a rigorous workup procedure involving sequential acid washing, alkali washing with sodium bicarbonate, and water washing. Followed by recrystallization in solvents like methanol or ethanol at controlled temperatures (-10 to 25°C), this method consistently achieves purity levels of 99.8%, meeting stringent requirements for non-toxic stabilizers.
Q: Is this synthesis method scalable for large-volume industrial production?
A: Yes, the reaction operates at mild temperatures (0-50°C) and atmospheric pressure with efficient gas handling for hydrogen evolution. These mild conditions reduce industrial energy consumption and equipment stress, making the process highly amenable to commercial scale-up from pilot batches to multi-ton production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Dibenzoylmethane Supplier
The technical potential of the sodium hydride-catalyzed synthesis of dibenzoylmethane is immense, offering a pathway to superior product quality and operational efficiency. At NINGBO INNO PHARMCHEM, we pride ourselves on our capability to translate such innovative patent technologies into commercial reality. As a seasoned CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of dibenzoylmethane we deliver meets the exacting standards required for high-end polymer stabilization and UV absorption applications.
We invite you to explore how our optimized manufacturing processes can enhance your product formulations and reduce your overall procurement costs. Our team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us demonstrate how we can become your trusted partner in securing a sustainable and high-quality supply of essential chemical intermediates.
