Advanced Manufacturing of Metribuzin Intermediate via Safe Catalytic Methylation
Introduction to Next-Generation Triazinone Synthesis
The global demand for high-efficiency herbicides continues to drive innovation in the manufacturing of critical agrochemical intermediates. Patent CN103333129A introduces a transformative methodology for the preparation of 4-amino-6-tertiary butyl-3-methylmercapto-1,2,4-triazine-5(4H)-ketone, a pivotal precursor in the synthesis of Metribuzin and related triazinone herbicides. This technical disclosure addresses long-standing industry challenges regarding safety, yield, and process controllability by shifting away from hazardous gaseous reagents toward a sophisticated liquid-phase catalytic system. By utilizing dimethyl sulfate as a methylating agent in the presence of a potassium iodide catalyst, the process achieves exceptional conversion rates under remarkably mild conditions. For R&D directors and supply chain strategists, this represents a significant opportunity to optimize production lines, reduce regulatory burdens associated with toxic gas handling, and secure a more reliable supply of high-purity intermediates essential for modern crop protection formulations.
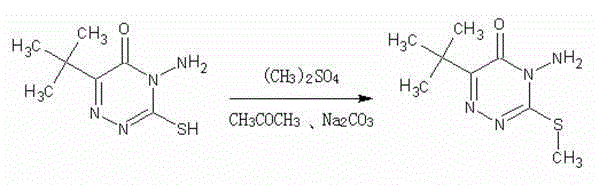
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of this key triazinone intermediate has relied on two primary pathways, both of which present substantial operational and safety liabilities. The first conventional method utilizes monobromethane (bromomethane) as the alkylating agent. As illustrated in the reaction scheme below, this approach necessitates the handling of a toxic, volatile gas that must be stored in high-pressure steel cylinders. 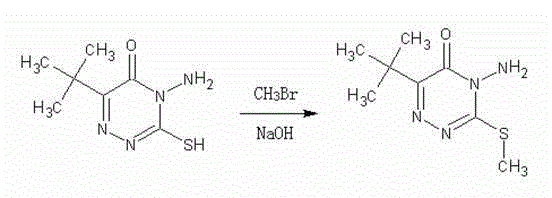
The requirement for pressurized reaction vessels significantly increases capital expenditure and maintenance costs, while the inherent toxicity of bromomethane poses severe occupational health risks and complicates leak detection protocols. Furthermore, the difficulty in precisely controlling the endpoint of a gas-liquid reaction often leads to inconsistent yields and necessitates extensive purification to meet international quality standards. The second traditional route employs a methanol and sulfuric acid system, which suffers from extremely poor efficiency, requiring reflux times of 20 to 30 hours to achieve transformation efficiencies of only around 80%. Prolonged exposure to acidic and thermal conditions frequently induces unwanted oxidation of the sulfhydryl and amino groups, resulting in dark-colored crude products laden with impurities that require multiple recrystallization steps to remediate.
The Novel Approach
In stark contrast to these legacy technologies, the novel approach detailed in the patent leverages a homogeneous catalytic system that operates at near-ambient temperatures. By substituting hazardous gaseous bromomethane with liquid dimethyl sulfate and employing potassium iodide as a nucleophilic catalyst, the reaction proceeds rapidly and selectively in an acetone solvent matrix. This method eliminates the need for high-pressure equipment, allowing the use of standard glass-lined or stainless steel reactors equipped with simple cooling systems to maintain the optimal 20-30°C range. The transition from a heterogeneous gas-liquid interface to a homogeneous liquid-phase reaction ensures superior mass transfer and heat dissipation, enabling precise control over reaction kinetics. Consequently, the production cycle is compressed from days to merely 4-5 hours, while simultaneously suppressing side reactions such as oxidation, thereby yielding a lighter-colored, higher-purity product directly from the filtration step without the need for energy-intensive distillation or complex chromatographic purification.
Mechanistic Insights into KI-Catalyzed Methylation
The efficacy of this synthetic route is rooted in the specific role of potassium iodide (KI) as a phase-transfer and nucleophilic catalyst within the acetone medium. In this mechanism, the iodide ion acts as a potent nucleophile that attacks the methyl group of dimethyl sulfate, generating methyl iodide in situ or forming a highly reactive iodide-sulfate complex that is more susceptible to nucleophilic attack by the sulfhydryl group of the triazinone substrate. This catalytic cycle significantly lowers the activation energy required for the S-methylation step compared to the uncatalyzed reaction with dimethyl sulfate alone. The use of anhydrous sodium carbonate serves a dual purpose: it acts as an acid scavenger to neutralize the methyl sulfuric acid byproduct formed during methylation, driving the equilibrium forward, and it helps maintain a slightly basic environment that enhances the nucleophilicity of the thiolate anion. This delicate balance of reagents prevents the protonation of the amino group, ensuring that methylation occurs selectively at the sulfur atom rather than the nitrogen, which is critical for maintaining the structural integrity required for herbicidal activity.
From an impurity control perspective, the mild thermal conditions (20-30°C) are paramount in preserving the chemical stability of the triazinone ring and its functional groups. In traditional acid-catalyzed processes, the combination of strong acid and high heat promotes the oxidation of the thiol (-SH) group to disulfides or sulfonic acids, and can also lead to the hydrolysis of the amide-like structures within the ring. By avoiding these harsh conditions, the novel process minimizes the formation of colored polymeric byproducts and oxidative degradation species. The stoichiometric ratio of dimethyl sulfate (1.2-1.5 equivalents) is carefully optimized to ensure complete conversion of the starting material while minimizing the presence of unreacted alkylating agent, which could otherwise complicate downstream waste treatment. The result is a crude product with a purity exceeding 96%, demonstrating that the kinetic selectivity of the KI-catalyzed pathway effectively outcompetes potential side reactions, simplifying the overall downstream processing train.
How to Synthesize 4-Amino-6-Tert-Butyl-3-Methylmercapto-1,2,4-Triazine-5(4H)-Ketone Efficiently
The implementation of this synthesis protocol requires careful attention to reagent addition rates and temperature monitoring to maximize the benefits of the catalytic system. The process begins by suspending the triazinone thiol substrate and anhydrous sodium carbonate in acetone, followed by the addition of the potassium iodide catalyst. The subsequent dropwise addition of dimethyl sulfate must be controlled to manage the exotherm, although the reaction is generally mild compared to alkylation with alkyl halides. Maintaining the temperature strictly within the 20-30°C window is crucial; temperatures that are too low may slow the catalytic turnover, while temperatures exceeding 30°C could risk minor decomposition or increased solvent loss. Following the reaction period of 4 to 5 hours, the acetone solvent is recovered via vacuum distillation, allowing for recycling and cost reduction. The residue is then treated with water to precipitate the product, which is isolated via filtration and dried under vacuum at 60°C.
- Charge the reactor with acetone, the thiol substrate, anhydrous sodium carbonate, and potassium iodide catalyst under agitation.
- Control the temperature between 20-30°C and slowly add dimethyl sulfate, maintaining this range for 4-5 hours.
- Recover acetone under vacuum, add water to precipitate the product, then filter and dry to obtain the high-purity intermediate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this catalytic methylation technology offers profound strategic advantages beyond mere technical performance. The shift from gaseous monobromethane to liquid dimethyl sulfate fundamentally alters the logistics and safety profile of the manufacturing site. Liquid reagents are significantly easier to store, transport, and meter than compressed toxic gases, reducing the reliance on specialized cylinder supply chains and high-pressure containment infrastructure. This transition translates into a substantial reduction in capital investment for plant equipment, as standard atmospheric reactors can be utilized instead of expensive autoclaves designed for high-pressure gas handling. Furthermore, the elimination of high-pressure operations lowers insurance premiums and reduces the regulatory compliance burden associated with storing large quantities of toxic gases, thereby enhancing the overall sustainability and social license to operate for the manufacturing facility.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven by multiple factors, primarily the drastic reduction in reaction time and energy consumption. By shortening the batch cycle from the traditional 20-30 hours down to just 4-5 hours, manufacturers can achieve a massive increase in asset utilization, effectively multiplying the annual production capacity of existing reactor trains without physical expansion. Additionally, the high selectivity of the reaction minimizes the loss of valuable starting materials to side products, directly improving the mass balance and reducing the cost per kilogram of the final API intermediate. The ability to recover and recycle the acetone solvent further contributes to operational cost savings, while the high crude purity (>96%) reduces the consumption of solvents and adsorbents typically required for extensive purification, leading to a leaner and more cost-effective production workflow.
- Enhanced Supply Chain Reliability: Supply chain resilience is significantly bolstered by the use of readily available and stable liquid raw materials. Unlike monobromethane, which is subject to stringent transportation regulations and potential supply disruptions due to its classification as a hazardous gas, dimethyl sulfate and acetone are commodity chemicals with robust global supply networks. This accessibility ensures that production schedules are less vulnerable to raw material shortages or logistical bottlenecks. Moreover, the simplicity of the process parameters—requiring only moderate cooling rather than complex heating or pressurization systems—reduces the likelihood of unplanned downtime caused by equipment failure. This operational robustness allows for more predictable lead times and consistent delivery performance, which is critical for downstream formulators who rely on just-in-time inventory strategies to manage their own production lines.
- Scalability and Environmental Compliance: Scaling this process from pilot to commercial production is straightforward due to the excellent heat transfer characteristics of the liquid-phase reaction. The mild exotherm can be easily managed even in large-scale reactors, mitigating the safety risks often associated with scaling up exothermic alkylations. From an environmental perspective, the process generates fewer hazardous byproducts and avoids the emission of volatile toxic gases, aligning with increasingly strict global environmental regulations. The high atom economy and the potential for solvent recovery minimize the volume of chemical waste requiring treatment, lowering disposal costs and the environmental footprint of the manufacturing site. This alignment with green chemistry principles not only future-proofs the supply chain against tightening regulations but also enhances the brand value of the end-product in markets that prioritize sustainably sourced agricultural inputs.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on how this method resolves specific pain points associated with traditional triazinone manufacturing. Understanding these nuances is essential for technical teams evaluating the feasibility of technology transfer and for commercial teams assessing the long-term viability of the supply source.
Q: Why is the potassium iodide catalyzed method superior to the monobromethane route?
A: The monobromethane route requires handling toxic gas under high pressure in sealed vessels, posing significant safety risks and equipment costs. The KI-catalyzed method uses liquid dimethyl sulfate at ambient pressure and mild temperatures (20-30°C), drastically improving operational safety and ease of control.
Q: What purity levels can be achieved with this new methylation process?
A: According to patent data, this method consistently yields products with a content above 96% directly after filtration and drying, with molar yields exceeding 90%, minimizing the need for complex purification steps.
Q: How does this process impact production cycle time compared to traditional acid-catalyzed methods?
A: Traditional methanol/sulfuric acid methods require long reflux times of 20-30 hours. The novel catalytic process completes the reaction in just 4-5 hours at low temperatures, significantly increasing reactor throughput and reducing energy consumption.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Amino-6-Tert-Butyl-3-Methylmercapto-1,2,4-Triazine-5(4H)-Ketone Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced synthetic methodologies like the KI-catalyzed methylation process is critical for maintaining competitiveness in the global agrochemical market. As a premier CDMO partner, we possess the technical expertise and infrastructure to seamlessly adopt and scale such innovative pathways, ensuring that our clients benefit from the highest standards of efficiency and safety. Our facilities are equipped to handle complex heterocyclic syntheses with precision, boasting extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. We are committed to delivering high-purity intermediates that meet stringent purity specifications, supported by our rigorous QC labs which utilize state-of-the-art analytical techniques to verify every batch against the most demanding international pharmacopeial and industry standards.
We invite you to collaborate with us to leverage this superior manufacturing technology for your supply chain. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating exactly how this optimized route can improve your bottom line. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, allowing us to demonstrate our capability to be your trusted partner in the reliable supply of high-quality agrochemical intermediates.
