Optimizing Sulfobenicillin Sodium Production: A Technical Breakthrough in Beta-Lactam Stability
Optimizing Sulfobenicillin Sodium Production: A Technical Breakthrough in Beta-Lactam Stability
The pharmaceutical industry constantly seeks robust synthetic routes for broad-spectrum antibiotics that balance high purity with industrial feasibility. A pivotal advancement in this domain is detailed in patent CN108373475B, which outlines a novel preparation method for Sulfobenicillin Sodium. This semi-synthetic penicillin is critical for treating infections caused by Pseudomonas aeruginosa and other Gram-negative bacteria, yet its production has historically been plagued by low yields and stability issues. The core innovation lies in a strategic chiral resolution followed by an anhydrous acylation process. By shifting away from traditional aqueous alkaline systems, this methodology preserves the integrity of the sensitive beta-lactam ring. For procurement leaders and technical directors, understanding this shift is vital for securing a reliable sulfobenicillin sodium supplier capable of delivering consistent quality.
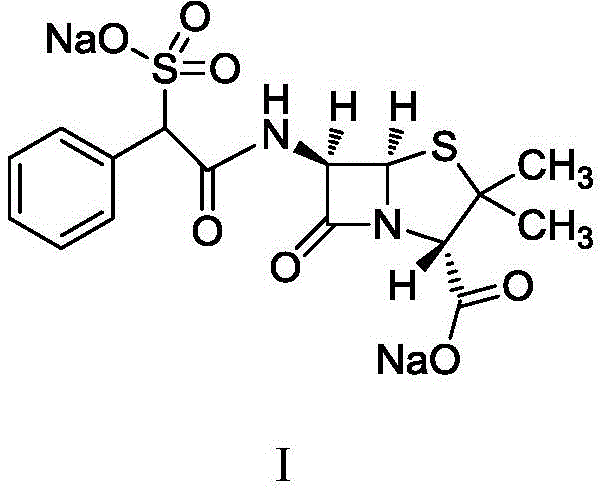
The structural complexity of Sulfobenicillin Sodium demands precise control over stereochemistry to ensure therapeutic efficacy. The molecule features a distinctive sulfonic acid group which enhances its water solubility and antibacterial spectrum. However, the presence of chiral centers necessitates rigorous control over the ratio of levorotatory and dextrorotatory isomers. The patented process addresses this by manipulating the starting materials before the final coupling reaction. This proactive approach to impurity control contrasts sharply with reactive purification methods used in older technologies. Consequently, manufacturers can achieve product purity greater than 99.0% while maintaining a molar yield exceeding 90%. Such metrics are essential for cost reduction in antibiotic manufacturing, as they minimize waste and maximize throughput per batch cycle.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of sulfobenicillin derivatives relied heavily on condensation reactions conducted in aqueous alkaline environments. While water is a convenient solvent, it poses significant risks to the stability of the beta-lactam ring found in 6-APA (6-Aminopenicillanic acid). Under alkaline conditions, the four-membered lactam ring is prone to hydrolysis, leading to the formation of inactive open-ring impurities. Furthermore, conventional resolution techniques often employed ion-exchange resins to separate chiral intermediates. These resin-based methods are not only capital intensive but also introduce operational bottlenecks due to resin saturation and regeneration requirements. The cumulative effect of these limitations is a process with lower overall yields, typically hovering around 60% to 75%, and higher production costs due to extensive downstream purification needs.
The Novel Approach
The methodology described in CN108373475B fundamentally reengineers the synthesis pathway to mitigate these risks. Instead of resolving the final product, the process resolves the side chain precursor, D,L-sulfophenylacetic acid, using L-amino acids such as L-histidine or L-lysine. This resolution step is performed in a controlled solvent system where the resolving agent can be easily recovered. Crucially, the subsequent acylation of 6-APA is conducted under strictly anhydrous conditions using organic solvents like dichloromethane or ethyl acetate. This eliminates the primary driver of beta-lactam degradation. The result is a streamlined workflow that simplifies post-processing and enhances the robustness of the reaction. For supply chain managers, this translates to commercial scale-up of complex beta-lactams that is both predictable and efficient.
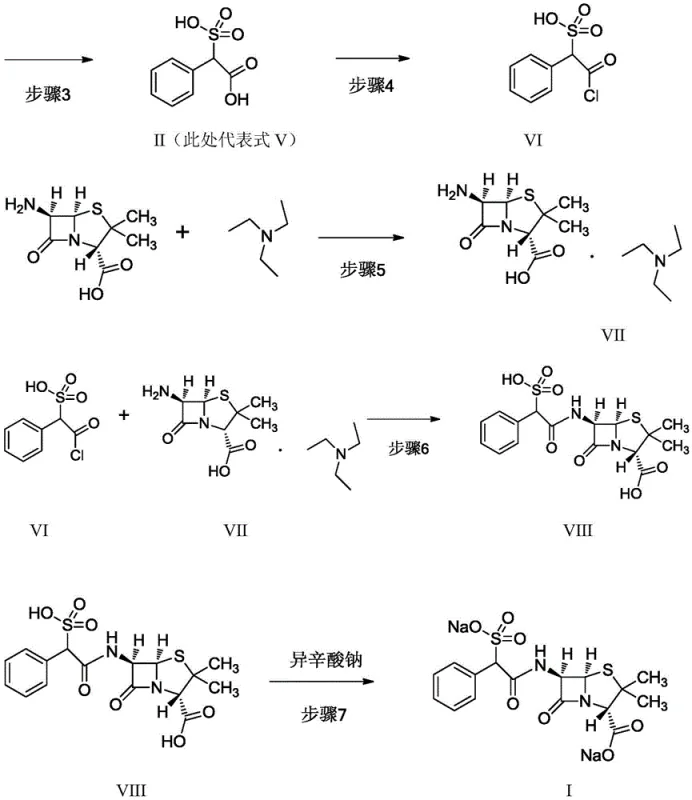
Mechanistic Insights into L-Amino Acid Mediated Chiral Resolution
The heart of this technological advancement is the chiral resolution mechanism utilizing L-amino acids. In the initial step, D,L-sulfophenylacetic acid reacts with an optically pure L-amino acid in an aqueous or alcoholic medium. Due to the difference in solubility between the diastereomeric salts formed, the D(-)-sulfophenylacetic acid L-amino acid salt preferentially crystallizes out of the solution. This solid-phase separation is highly selective and allows for the isolation of the desired D-isomer with high optical purity. Following filtration, the salt is treated with a mineral acid to liberate the free D(-)-sulfophenylacetic acid. This two-step resolution and liberation process is far more scalable than chromatographic separations. It ensures that the chiral integrity of the side chain is established before it ever encounters the sensitive 6-APA core, thereby locking in the stereochemical outcome early in the synthesis.
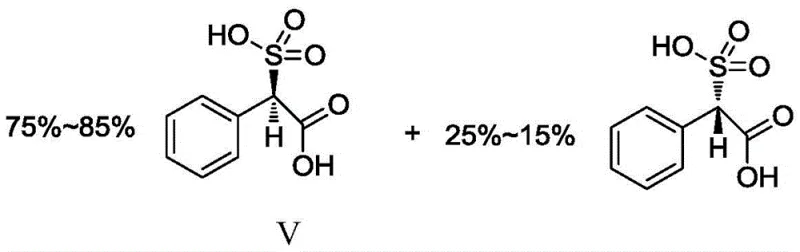
Following the isolation of the D(-)-isomer, the process employs a unique blending strategy. The pure D(-)-acid is mixed with the remaining racemic D,L-acid in a specific mass ratio, typically around 2:1. This compounding step creates a feedstock mixture with a predefined isomer ratio of approximately 78% D(-) to 22% L(+). This specific ratio aligns perfectly with pharmacopoeial requirements for the final drug substance. By adjusting the chirality at the raw material stage, the need for difficult chiral separations after the coupling reaction is entirely eliminated. The subsequent conversion of this mixture to the acid chloride and reaction with 6-APA proceeds with the isomer ratio intact. This mechanistic foresight drastically reduces the burden on quality control laboratories and ensures that every batch meets the stringent specifications for high-purity penicillin derivative standards without additional enrichment steps.
How to Synthesize Sulfobenicillin Sodium Efficiently
Implementing this synthesis route requires strict adherence to temperature controls and stoichiometric precision, particularly during the acylation phase. The process begins with the resolution of the side chain, followed by activation with thionyl chloride at sub-zero temperatures to prevent racemization. The activated acid chloride is then coupled with 6-APA, which has been pre-treated with triethylamine to form a soluble salt in an organic medium. Maintaining the reaction temperature between -15°C and -10°C is critical to suppress side reactions. Once the coupling is complete, the intermediate acid is converted to the sodium salt using sodium isooctanoate in acetone, inducing crystallization of the final product. Detailed standardized operating procedures are essential for reproducibility.
- Resolve D,L-sulfophenylacetic acid using L-amino acids like L-histidine to isolate the D(-)-isomer salt.
- Free the D(-)-acid and mix with racemic acid to achieve a precise 78: 22 D/L ratio.
- Convert the acid mixture to acid chloride under anhydrous conditions at low temperature.
- React the acid chloride with 6-APA triethylamine salt in organic solvent to form the beta-lactam core.
- Perform salt formation with sodium isooctanoate to crystallize the final Sulfobenicillin Sodium product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this patented synthesis method offers substantial strategic benefits beyond mere technical elegance. The elimination of aqueous conditions during the critical coupling step significantly reduces the generation of hydrolytic impurities, which in turn minimizes the need for expensive and time-consuming purification processes like recrystallization or chromatography. This streamlining of the manufacturing workflow leads to a drastic simplification of the post-processing phase. By removing the dependency on ion-exchange resins for chiral separation, the process avoids the high operational costs and waste disposal issues associated with resin regeneration. These efficiencies collectively contribute to significant cost savings in the overall production budget, making the supply of this antibiotic more economically sustainable in a competitive market.
- Cost Reduction in Manufacturing: The adoption of L-amino acids as resolving agents presents a clear economic advantage over traditional resin-based methods. Amino acids are generally more affordable and easier to handle on a large scale. Furthermore, the ability to recover and recycle the resolving agent through simple acidification reduces raw material consumption. The anhydrous nature of the coupling reaction also means that solvent recovery is more straightforward, as there is no water to separate from the organic phase. These factors combine to lower the variable cost per kilogram of the active pharmaceutical ingredient, providing a buffer against fluctuating raw material prices and enhancing the overall margin profile for the manufacturing operation.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by complex processes that are prone to failure or batch rejection. This new method enhances reliability by increasing the robustness of the reaction conditions. The high molar yield, consistently reported above 90%, ensures that production targets are met with fewer batches, reducing the strain on manufacturing capacity. Additionally, the use of common organic solvents like dichloromethane and ethyl acetate ensures that raw material sourcing remains stable and unaffected by niche supply constraints. This stability is crucial for reducing lead time for high-purity antibiotics, allowing manufacturers to respond more agilely to market demand spikes without compromising on quality or delivery schedules.
- Scalability and Environmental Compliance: Scaling chemical processes from the laboratory to industrial reactors often reveals hidden inefficiencies, but this route is designed with scalability in mind. The avoidance of water in the acylation step simplifies waste treatment, as the effluent streams contain less organic load and no emulsified aqueous waste. The solid-state resolution step generates less liquid waste compared to extraction-heavy methods. Moreover, the high purity of the crude product reduces the environmental footprint associated with secondary purification solvents. These attributes facilitate smoother regulatory approvals and align with modern green chemistry principles, ensuring long-term compliance with increasingly stringent environmental regulations governing pharmaceutical manufacturing facilities.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of Sulfobenicillin Sodium using this advanced methodology. These insights are derived directly from the experimental data and comparative examples provided in the patent literature. Understanding these nuances helps stakeholders make informed decisions regarding technology transfer and vendor qualification. The answers reflect the consensus on best practices for maintaining product integrity and optimizing process economics in a GMP environment.
Q: How does this method improve upon traditional aqueous synthesis?
A: Traditional methods often use aqueous alkaline systems which degrade the sensitive beta-lactam ring. This patented approach utilizes anhydrous conditions during the critical acylation step, significantly reducing hydrolysis impurities and boosting overall molar yield above 90%.
Q: What is the achieved optical purity of the final product?
A: By pre-adjusting the starting material ratio to approximately 78% D(-) and 22% L(+), the process directly yields the pharmacologically required isomer ratio without complex downstream purification, ensuring purity levels exceeding 99.0%.
Q: Is the chiral resolution agent recoverable for industrial scale?
A: Yes, unlike ion-exchange resin methods which are costly and difficult to scale, this process uses L-amino acids that can be separated via simple acidification and filtration, making the resolving agent highly recoverable and cost-effective for large-scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Sulfobenicillin Sodium Supplier
The technical potential of this synthesis route is immense, offering a pathway to superior product quality and operational efficiency. At NINGBO INNO PHARMCHEM, we possess the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to bring such innovations to life. Our facility is equipped with stringent purity specifications and rigorous QC labs to ensure that every batch of Sulfobenicillin Sodium meets the highest international standards. We understand the critical nature of beta-lactam stability and have optimized our reactor configurations to maintain the precise low-temperature conditions necessary for this chemistry. Partnering with us means gaining access to a supply chain that is both technically sophisticated and commercially resilient.
We invite you to initiate a dialogue regarding your specific supply requirements. Our team is prepared to provide a Customized Cost-Saving Analysis that demonstrates how switching to this optimized route can impact your bottom line. We encourage potential partners to contact our technical procurement team to request specific COA data and route feasibility assessments tailored to your project timelines. By collaborating closely, we can ensure a seamless integration of this high-quality intermediate into your broader pharmaceutical manufacturing portfolio.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
