Advanced Synthesis of 2-Chloro-4-Aminopyridine for Scalable Agrochemical Manufacturing
The global demand for high-efficiency plant growth regulators continues to surge, driving the need for robust synthetic routes to key heterocyclic intermediates. Patent CN111056999A introduces a strategically optimized preparation method for 2-chloro-4-aminopyridine, a critical building block for cytohormone activity regulators such as N-(2-chloro-4-pyridyl) urea. This specific intermediate plays a pivotal role in modern agriculture, particularly in enhancing tuber formation and preventing aging in crops like potatoes. The disclosed technology addresses long-standing challenges in pyridine functionalization by leveraging an N-oxide mediation strategy that ensures superior regioselectivity and operational simplicity. For R&D directors and procurement specialists seeking a reliable agrochemical intermediate supplier, this patent offers a compelling blueprint for achieving high-purity 2-chloro-4-aminopyridine with minimized process complexity. By shifting away from harsh, non-selective direct substitution methods, this approach not only improves chemical yields but also aligns with modern green chemistry principles through the use of accessible reagents and straightforward workup procedures.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic pathways for accessing 4-substituted-2-chloropyridines have historically been plagued by significant technical hurdles that impede efficient commercial scale-up of complex pyridine derivatives. Direct electrophilic aromatic substitution on the pyridine ring is notoriously difficult due to the electron-deficient nature of the heterocycle, often requiring extreme temperatures and aggressive conditions that degrade sensitive functional groups. Furthermore, without a strong directing group, nitration or amination reactions frequently result in a mixture of regioisomers, such as 3-substituted and 5-substituted byproducts, which are chemically similar and extremely costly to separate via chromatography or crystallization. These purification bottlenecks drastically reduce the overall mass balance and inflate the cost of goods sold, making traditional routes economically unviable for large-volume agrochemical production. Additionally, many legacy processes rely on hazardous reagents or transition metal catalysts that introduce heavy metal impurities, necessitating expensive downstream removal steps to meet stringent pharmaceutical and agrochemical purity specifications.
The Novel Approach
The methodology outlined in CN111056999A circumvents these intrinsic electronic limitations by temporarily modifying the pyridine nitrogen into an N-oxide functionality. This strategic oxidation step fundamentally alters the electron density distribution of the ring, strongly activating the 4-position towards electrophilic attack while deactivating the 3 and 5 positions. As illustrated in the reaction scheme below, this allows for highly regioselective nitration using standard mixed acid systems, effectively channeling the reaction pathway exclusively toward the desired 4-nitro isomer. 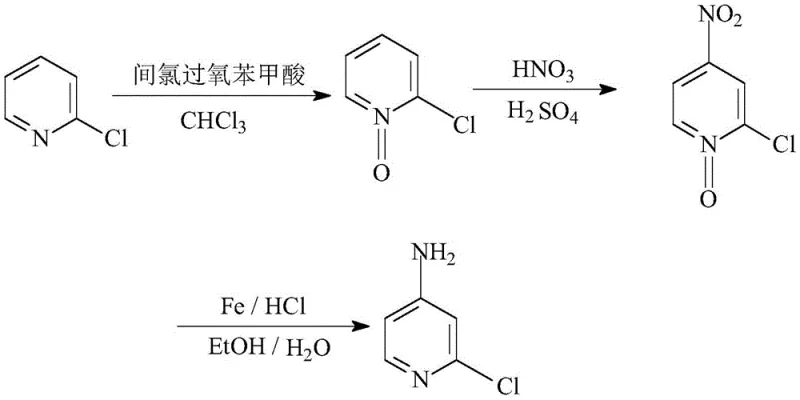 The subsequent reduction step utilizes a classic yet highly effective iron-mediated protocol that avoids the safety risks associated with catalytic hydrogenation. This novel approach transforms a potentially low-yielding, multi-isomer process into a streamlined, high-fidelity sequence. For procurement managers focused on cost reduction in agrochemical intermediates manufacturing, this route offers a distinct advantage by utilizing commodity chemicals like chloroform, m-CPBA, and iron powder, thereby insulating the supply chain from the volatility of specialized catalyst markets.
The subsequent reduction step utilizes a classic yet highly effective iron-mediated protocol that avoids the safety risks associated with catalytic hydrogenation. This novel approach transforms a potentially low-yielding, multi-isomer process into a streamlined, high-fidelity sequence. For procurement managers focused on cost reduction in agrochemical intermediates manufacturing, this route offers a distinct advantage by utilizing commodity chemicals like chloroform, m-CPBA, and iron powder, thereby insulating the supply chain from the volatility of specialized catalyst markets.
Mechanistic Insights into N-Oxide Directed Nitration and Reduction
The core innovation of this synthesis lies in the electronic modulation provided by the pyridine N-oxide moiety during the nitration phase. In the neutral 2-chloropyridine substrate, the nitrogen atom exerts a strong electron-withdrawing inductive effect, rendering the ring resistant to electrophilic attack. However, upon oxidation to the N-oxide, the oxygen atom donates electron density into the ring system through resonance, specifically increasing electron density at the 4-position (para to the nitrogen). When exposed to the nitronium ion generated from the concentrated nitric and sulfuric acid mixture, the 4-position becomes the kinetically favored site for substitution. This electronic steering mechanism ensures that the formation of the 2-chloro-4-nitropyridine N-oxide proceeds with high specificity, minimizing the generation of difficult-to-remove isomeric impurities that typically compromise the quality of high-purity 2-chloro-4-aminopyridine. The stability of the N-oxide under these acidic conditions further contributes to the robustness of the process, allowing for controlled reaction kinetics even at elevated temperatures up to 100°C.
Following the successful installation of the nitro group, the restoration of the pyridine nitrogen and the conversion of the nitro group to an amine occur simultaneously during the reduction phase. The use of iron powder in the presence of hydrochloric acid generates nascent hydrogen in situ, which acts as the reducing agent. This heterogeneous reaction system is particularly advantageous for industrial applications because the iron salts formed are water-soluble and can be easily separated from the organic product via filtration and phase separation. The solvent system, comprising ethanol and water in a specific volume ratio, optimizes the solubility of both the organic substrate and the inorganic reagents, facilitating efficient mass transfer. This mechanistic pathway avoids the use of precious metals like palladium or platinum, which are not only expensive but also prone to poisoning by sulfur or chlorine contaminants often present in bulk chemical feedstocks. Consequently, the process demonstrates exceptional tolerance to feedstock variations, ensuring consistent batch-to-batch reproducibility essential for reducing lead time for high-purity agrochemical intermediates.
How to Synthesize 2-Chloro-4-Aminopyridine Efficiently
The execution of this synthesis requires precise control over stoichiometry and temperature to maximize the yield of each transformation while maintaining safety standards. The initial oxidation step demands careful monitoring of the peracid consumption to prevent over-oxidation or residual oxidant carryover, which could pose safety risks in subsequent steps. Following isolation of the N-oxide, the nitration must be conducted with strict temperature control during the acid addition phase to manage the exotherm, followed by heating to drive the reaction to completion.
- Oxidize 2-chloropyridine using m-chloroperoxybenzoic acid in chloroform to form 2-chloropyridine N-oxide.
- Perform nitration on the N-oxide intermediate using a mixed acid system of concentrated nitric and sulfuric acid to yield 2-chloro-4-nitropyridine N-oxide.
- Reduce the nitro-N-oxide species using iron powder and hydrochloric acid in an ethanol-water solvent system to obtain the final amine product.
Commercial Advantages for Procurement and Supply Chain Teams
For supply chain leaders evaluating potential partners, the economic and logistical implications of this patented process are profound. The reliance on widely available commodity chemicals rather than proprietary or scarce reagents creates a resilient supply chain that is less susceptible to global market disruptions. The elimination of complex purification steps, such as column chromatography, in favor of simple extraction and crystallization techniques significantly reduces processing time and solvent consumption. This operational efficiency translates directly into lower manufacturing overheads and a smaller environmental footprint, aligning with the sustainability goals of modern agrochemical corporations. Furthermore, the mild reaction conditions reduce the energy burden on production facilities, as there is no need for cryogenic cooling or ultra-high pressure equipment. These factors collectively contribute to a more predictable and cost-effective production model, enabling suppliers to offer competitive pricing without compromising on the stringent quality standards required for crop protection agents.
- Cost Reduction in Manufacturing: The substitution of expensive noble metal catalysts with inexpensive iron powder represents a fundamental shift in cost structure for this synthesis. By removing the dependency on palladium or rhodium, manufacturers eliminate a significant variable cost component and avoid the capital expenditure associated with hydrogenation reactors. Additionally, the high regioselectivity of the N-oxide route minimizes the loss of raw materials to byproduct formation, effectively increasing the atom economy of the process. The simplified workup procedures further reduce labor costs and waste disposal fees, as the aqueous iron waste streams are easier to treat compared to heavy metal catalyst residues. These cumulative efficiencies allow for substantial cost savings that can be passed down the value chain, enhancing the competitiveness of the final plant growth regulator in the global market.
- Enhanced Supply Chain Reliability: Sourcing strategies for chemical intermediates are increasingly focused on security of supply, and this route excels by utilizing reagents that are produced at massive scales globally. Chloroform, sulfuric acid, nitric acid, and iron powder are foundational chemicals with stable supply lines, unlike specialized ligands or organometallic complexes that may have single-source bottlenecks. This diversity of supply sources mitigates the risk of production stoppages due to raw material shortages. Moreover, the robustness of the chemistry means that the process can be easily transferred between different manufacturing sites or scaled up rapidly to meet sudden spikes in demand without extensive re-optimization. This flexibility is crucial for maintaining continuous supply to downstream formulators who depend on timely deliveries to support seasonal agricultural cycles.
- Scalability and Environmental Compliance: The transition from laboratory to industrial scale is often where new processes fail, but the described method is explicitly designed for scalability. The use of heterogeneous iron reduction is a well-understood unit operation in the fine chemical industry, allowing for straightforward engineering of large-scale reactors. The absence of high-pressure hydrogen gas eliminates a major safety hazard, simplifying regulatory compliance and insurance requirements for manufacturing plants. From an environmental perspective, the process generates less hazardous waste compared to alternative routes involving heavy metals or chlorinating agents. The ability to recycle solvents like chloroform and ethanol further enhances the green profile of the synthesis. These attributes make the technology highly attractive for companies aiming to expand their production capacity while adhering to increasingly strict environmental regulations and corporate social responsibility mandates.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis is vital for stakeholders making sourcing decisions. The following questions address common concerns regarding the feasibility, quality, and scalability of the N-oxide route for producing 2-chloro-4-aminopyridine. These insights are derived directly from the experimental data and technical disclosures within the patent documentation, providing a transparent view of the process capabilities.
Q: Why is the N-oxide route preferred for synthesizing 2-chloro-4-aminopyridine?
A: Direct nitration of 2-chloropyridine often lacks regioselectivity. Converting to the N-oxide activates the 4-position electronically, ensuring high selectivity for the 4-nitro isomer, which simplifies purification and improves overall yield.
Q: What are the cost advantages of using iron powder for reduction in this process?
A: Using iron powder and hydrochloric acid avoids the need for expensive noble metal catalysts like palladium or specialized high-pressure hydrogenation equipment. This significantly lowers capital expenditure and operational costs for large-scale production.
Q: Is this synthesis method suitable for industrial scale-up?
A: Yes, the patent explicitly states the method features mild reaction conditions, simple post-treatment, and the use of cheap, readily available raw materials, making it highly suitable for commercial scale-up of complex pyridine derivatives.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Chloro-4-Aminopyridine Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of next-generation plant growth regulators depends on access to high-quality, cost-effective intermediates. Our technical team has extensively analyzed the pathway described in CN111056999A and possesses the expertise to implement this N-oxide mediated strategy at an industrial level. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. Our facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, ensuring that every batch of 2-chloro-4-aminopyridine meets the exacting standards required for agrochemical registration and formulation. We are committed to being a partner that not only supplies chemicals but also provides technical solutions that enhance your overall product viability.
We invite you to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific project requirements. By leveraging our manufacturing capabilities, you can secure a stable supply of this critical intermediate while optimizing your cost structure. Please contact us to request a Customized Cost-Saving Analysis tailored to your volume needs. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our implementation of this technology can accelerate your time-to-market and strengthen your position in the competitive agrochemical sector.
