Advanced Difluorosulfonimide Synthesis: Scaling Safe Electrolyte Production for EVs
The rapid evolution of the electric vehicle (EV) and energy storage sectors has placed unprecedented demands on lithium-ion battery performance, specifically regarding energy density, cycle life, and thermal stability. As the industry seeks to overcome the inherent limitations of traditional lithium hexafluorophosphate (LiPF6) electrolytes, such as poor thermal stability and susceptibility to hydrolysis, the focus has shifted decisively towards next-generation salts like lithium bis(fluorosulfonyl)imide (LiFSI). Patent CN116283601A introduces a groundbreaking preparation method for difluorosulfonimide organic ammonium salts, which serve as critical precursors for these advanced electrolytes. This technology represents a significant leap forward in chemical synthesis, addressing the long-standing safety and efficiency bottlenecks associated with fluorinated salt production. By utilizing sulfuryl fluoride and ammonium salts in an aprotic polar solvent system, this novel approach offers a robust pathway for reliable lithium battery electrolyte intermediate supplier operations, ensuring that the supply chain can meet the surging global demand for high-performance energy storage solutions without compromising on safety or environmental standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of bis(fluorosulfonyl)imide derivatives has been plagued by complex multi-step processes that pose severe safety and economic challenges. Traditional routes often rely on chlorosulfonic acid and sulfamic acid to generate dichloro-sulfonyl-imide intermediates, which subsequently require hazardous fluorination steps using hydrogen fluoride (HF) or elemental fluorine gas under high temperature and high pressure conditions. These legacy methods not only involve highly corrosive and toxic reagents that demand expensive, specialized containment equipment but also generate significant amounts of hazardous waste, complicating environmental compliance. Furthermore, the direct reaction of sulfuryl fluoride with ammonia gas, while theoretically simpler, presents substantial operational risks due to the simultaneous handling of two toxic gases, making reaction control difficult and limiting its adoption to only a few specialized facilities. These factors collectively contribute to high production costs and supply chain vulnerabilities, hindering the widespread cost reduction in electronic chemical manufacturing necessary for the mass adoption of advanced battery technologies.
The Novel Approach
In stark contrast to these perilous conventional pathways, the method disclosed in patent CN116283601A utilizes a remarkably safer and more efficient strategy by reacting sulfuryl fluoride with solid or dissolved ammonium salts in the presence of an organic alkaline acid-binding agent. This innovation effectively bypasses the need for direct handling of toxic ammonia gas or highly corrosive hydrogen fluoride during the critical bond-forming stages. The reaction proceeds smoothly in common aprotic polar solvents like acetonitrile at mild temperatures ranging from 0°C to 60°C, drastically reducing the energy input and equipment stress compared to high-pressure fluorination processes. By shifting the paradigm from gas-gas or gas-liquid hazardous reactions to a more controllable gas-liquid-solid system, this approach significantly lowers the barrier to entry for commercial scale-up of complex fluorinated salts. The result is a streamlined process that delivers high-purity difluorosulfonimide intermediates with exceptional yields, providing a scalable foundation for the next generation of battery electrolytes.
Mechanistic Insights into Sulfuryl Fluoride Amidation
The core of this technological breakthrough lies in the elegant nucleophilic substitution and acid-base neutralization mechanism that drives the formation of the difluorosulfonimide anion. In this reaction system, sulfuryl fluoride acts as the electrophilic sulfur source, while the ammonium salt provides the necessary nitrogen nucleophile. The presence of an organic base, such as triethylamine, plays a dual critical role: it acts as an acid scavenger to neutralize the hydrogen fluoride byproduct generated during the substitution, and it facilitates the formation of the stable organic ammonium salt of the difluorosulfonimide anion. This mechanistic pathway ensures that the reaction equilibrium is driven towards the product side without the accumulation of corrosive acids that could degrade the reactor or the product. The choice of solvent is equally pivotal; polar aprotic solvents like acetonitrile stabilize the ionic transition states and the final ionic product, thereby enhancing reaction kinetics and overall conversion efficiency.
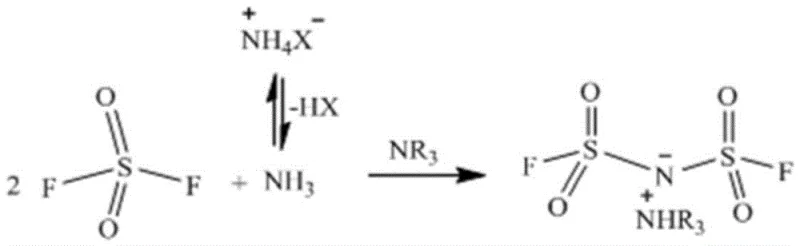
Furthermore, the control of impurities is intrinsically built into this mechanistic design through the precise stoichiometric balance of reactants. The patent data highlights that optimizing the molar ratio of sulfuryl fluoride, the organic base, and the ammonium salt is crucial for minimizing side reactions and maximizing yield. For instance, an excess of the organic base ensures complete scavenging of HF, preventing potential acid-catalyzed decomposition of the sensitive sulfonyl groups. Additionally, the mild reaction conditions prevent thermal degradation pathways that are common in high-temperature fluorination processes. This results in a cleaner crude product profile, which simplifies downstream purification steps such as distillation and washing. For R&D teams, understanding this mechanism allows for fine-tuning of reaction parameters to achieve the stringent purity specifications required for battery-grade electrolytes, ensuring long-term cycle stability and safety in the final energy storage device.
How to Synthesize Difluorosulfonimide Efficiently
To implement this advanced synthesis route effectively, manufacturers must adhere to a standardized protocol that leverages the specific advantages of the ammonium salt methodology. The process begins with the careful selection of high-purity ammonium salts, such as ammonium fluoride or ammonium chloride, which are readily available and easy to handle compared to gaseous ammonia. These salts are dissolved or suspended in a dry aprotic solvent like acetonitrile under an inert nitrogen atmosphere to prevent moisture ingress, which is critical for maintaining the integrity of the sulfuryl fluoride reagent. The subsequent addition of the organic base and the controlled introduction of sulfuryl fluoride gas require precise monitoring of temperature and pressure to maintain the optimal reaction window identified in the patent examples. Detailed standardized synthesis steps see the guide below for a comprehensive breakdown of the operational parameters.
- Prepare the reaction system by adding ammonium salt (e.g., ammonium fluoride) and an organic base (e.g., triethylamine) into an aprotic polar solvent like acetonitrile under nitrogen atmosphere.
- Introduce sulfuryl fluoride gas slowly into the reaction mixture while maintaining the temperature between 0°C and 60°C, preferably around 10°C to 30°C.
- Stir the reaction for several hours, then recover the solvent via reduced pressure distillation and purify the resulting organic phase to obtain the target ammonium salt.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthesis method translates into tangible strategic benefits that extend far beyond simple chemical yield improvements. The primary advantage lies in the drastic simplification of the raw material portfolio; by replacing hazardous gases like fluorine and hydrogen fluoride with stable solid ammonium salts and common organic bases, companies can significantly reduce the costs associated with hazardous material storage, transportation, and regulatory compliance. This shift not only lowers the total cost of ownership for the production facility but also mitigates the risk of supply disruptions caused by the limited availability of specialized toxic reagents. Moreover, the elimination of high-pressure, high-corrosion reaction steps means that manufacturers can utilize more standard chemical processing equipment, leading to substantial capital expenditure savings and faster deployment of new production lines. These factors collectively enhance the resilience and agility of the supply chain, ensuring a steady flow of high-purity lithium battery electrolyte intermediates to meet market demands.
- Cost Reduction in Manufacturing: The economic impact of this process is profound, primarily driven by the removal of expensive and dangerous fluorination agents. Traditional routes requiring anhydrous hydrogen fluoride necessitate nickel-lined reactors and extensive safety infrastructure, which represent a massive financial burden. By avoiding these materials entirely, the new method allows for the use of standard stainless steel or glass-lined equipment, drastically lowering both initial investment and maintenance costs. Additionally, the high reaction yields reported in the patent data mean that less raw material is wasted, improving the overall atom economy of the process. This efficiency gain directly correlates to a lower cost per kilogram of the final product, providing a competitive edge in the price-sensitive battery materials market without sacrificing quality or performance standards.
- Enhanced Supply Chain Reliability: From a logistics perspective, the reliance on solid ammonium salts and common solvents like acetonitrile creates a much more robust supply chain compared to one dependent on toxic gases. Solid reagents are easier to transport, store, and dose accurately, reducing the likelihood of operational delays caused by delivery issues or safety inspections associated with hazardous gas cylinders. This stability is crucial for maintaining continuous production schedules, especially in regions with strict environmental and safety regulations that might restrict the transport of fluorine or ammonia gas. By securing a supply of easily accessible raw materials, manufacturers can guarantee consistent lead times for their customers, fostering stronger long-term partnerships with major battery cell producers who prioritize supply security above all else.
- Scalability and Environmental Compliance: The environmental footprint of this synthesis route is significantly smaller than that of conventional methods, aligning perfectly with the sustainability goals of the modern EV industry. The absence of highly toxic byproducts and the reduced need for extreme reaction conditions simplify waste treatment processes, making it easier for facilities to meet increasingly stringent environmental discharge standards. This compliance advantage reduces the risk of regulatory fines and production shutdowns, ensuring long-term operational continuity. Furthermore, the mild reaction conditions and straightforward workup procedures make the process highly scalable from pilot plant to full commercial production. This scalability ensures that as demand for LiFSI electrolytes grows exponentially, manufacturers can rapidly expand capacity without encountering the technical bottlenecks often associated with scaling hazardous high-pressure chemistries.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this difluorosulfonimide synthesis technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing a clear picture of what stakeholders can expect during technology transfer. Understanding these details is essential for evaluating the feasibility of integrating this route into existing manufacturing frameworks or for planning new greenfield projects dedicated to advanced electrolyte production. The answers below reflect the practical realities of scaling this chemistry while maintaining the highest standards of safety and product quality.
Q: What are the safety advantages of this new difluorosulfonimide synthesis method?
A: This method eliminates the need for highly corrosive and toxic hydrogen fluoride (HF) or fluorine gas (F2) typically used in traditional fluorination steps. By using solid or solution-phase ammonium salts instead of toxic ammonia gas directly, it significantly reduces reaction control difficulty and operational risks.
Q: How does this process impact the cost of LiFSI production?
A: The process reduces costs by simplifying the reaction steps and avoiding the need for specialized high-pressure, high-corrosion-resistant equipment required for HF handling. The use of common organic bases and solvents like acetonitrile further optimizes the raw material cost structure.
Q: What is the typical yield and purity achievable with this route?
A: Experimental data indicates high yields, often exceeding 90%, with excellent product purity. The use of specific molar ratios of sulfuryl fluoride, base, and ammonium salt allows for precise control over byproduct formation, ensuring high-quality electrolyte intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Difluorosulfonimide Supplier
As the global demand for high-performance lithium-ion batteries continues to surge, the need for reliable sources of advanced electrolyte intermediates has never been more critical. NINGBO INNO PHARMCHEM stands at the forefront of this industry, leveraging deep expertise in fluorine chemistry to deliver superior difluorosulfonimide derivatives. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the volume requirements of even the largest battery manufacturers. We are committed to maintaining stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of material meets the exacting standards required for next-generation energy storage applications. Our state-of-the-art facilities are designed to handle complex fluorinated chemistries safely and efficiently, providing a secure foundation for your supply chain.
We invite you to collaborate with us to optimize your electrolyte sourcing strategy and unlock new levels of battery performance. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific production needs, helping you identify opportunities for efficiency and margin improvement. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us help you navigate the complexities of the battery materials market with a partner dedicated to innovation, safety, and commercial success.
