Scalable Manufacturing of High-Purity Taxane Derivatives via Novel Carbonate Formation
The pharmaceutical industry continuously seeks robust synthetic routes for complex anticancer agents, particularly taxane derivatives which serve as critical precursors for drugs like paclitaxel and docetaxel. Patent CN101448839B presents a significant technological breakthrough in the preparation of 13-(N-Boc-β-isobutylserinyl)-14-β-hydroxybaccatin III-1,14-carbonate, designated as compound (I). This specific intermediate is pivotal in the supply chain for next-generation antineoplastic therapies targeting breast, lung, and ovarian tumors. The disclosed innovation fundamentally alters the traditional manufacturing landscape by replacing hazardous gaseous reagents with safer solid surrogates and introducing a novel protecting group strategy that facilitates purification through crystallization rather than chromatography. For R&D directors and procurement specialists, this represents a dual advantage of enhanced safety profiles and substantial cost optimization potential. The method addresses long-standing bottlenecks in taxane synthesis, specifically the difficulties associated with regioselective carbonate formation and the removal of silyl-based protecting groups, thereby offering a more reliable taxane intermediate supplier pathway for global pharmaceutical manufacturers seeking to secure their API supply chains against regulatory and operational risks.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of compound (I) has relied heavily on Method B as described in prior art such as WO01/02407, which involves the use of phosgene gas for the critical 1,14-hydroxyl carbonate formation step. While this method offers good regioselectivity compared to earlier attempts, it suffers from severe scalability issues due to the inherent toxicity and handling difficulties of phosgene gas on an industrial scale. When phosgene is bubbled into the reactor to effect carbonate formation, a significant side reaction occurs where the 7-hydroxyl group also reacts, leading to the formation of impurity (II), as illustrated in the structural diagram below. This impurity possesses a compact structure very similar to the desired product, making its removal exceptionally difficult and typically requiring resource-intensive column chromatography.
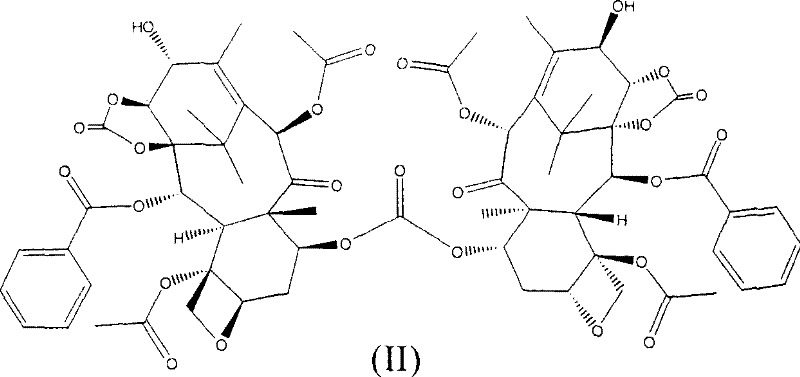
Furthermore, the conventional use of triethylsilyl (TES) groups for protecting the 7-hydroxyl position introduces another major bottleneck. Upon deprotection, triethylsilyl fluoride is generated, which cannot be completely removed by standard crystallization or low-pressure column chromatography. This necessitates complex purification protocols that are not only expensive but also generate substantial silica gel waste contaminated with toxic substances, posing environmental compliance challenges and increasing the overall cost reduction in API manufacturing efforts. The reliance on chromatography severely limits the throughput and economic viability of producing high-purity pharmaceutical intermediates at the multi-kilogram or tonnage scale required by the global market.
The Novel Approach
The present invention overcomes these critical limitations through two strategic chemical modifications that streamline the entire synthetic sequence. First, the hazardous phosgene gas is replaced by bis(trichloromethyl)carbonate, a solid reagent that acts as a safe and effective equivalent for carbonate formation. This substitution eliminates the safety risks associated with handling toxic gases and, crucially, prevents the formation of impurity (II) by controlling the reactivity profile during the carbonate cyclization step. Second, the traditional triethylsilyl protecting group is replaced with a trichloroacetyl group introduced via trichloroacetyl chloride. This seemingly small change has profound implications for downstream processing, as the resulting trichloroacetamide byproduct formed during deprotection is highly soluble in aqueous mixtures, allowing it to be washed away easily without the need for chromatography.
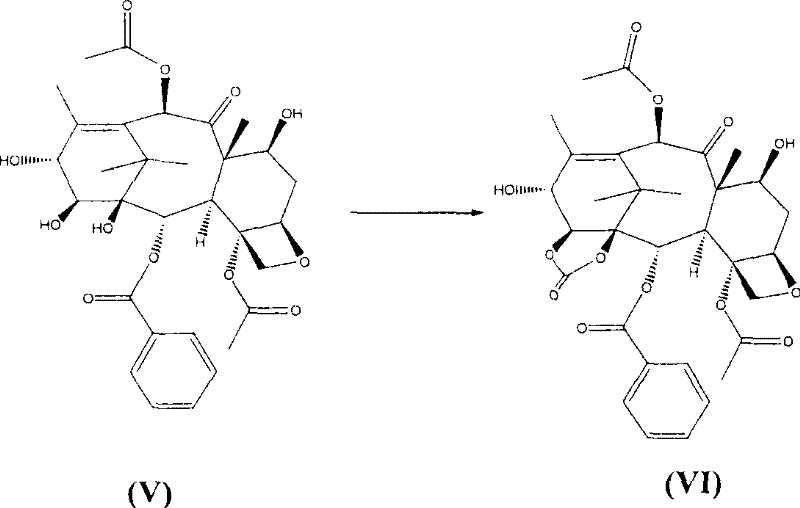
Consequently, the novel approach enables the isolation of intermediates and the final product through simple crystallization techniques, achieving purity levels exceeding 99.9%. This shift from chromatographic purification to crystallization is a game-changer for commercial scale-up of complex taxanes, drastically reducing solvent consumption, waste generation, and processing time. The ability to obtain the final compound (I) as a solid with such high purity directly from crystallization ensures a consistent quality profile that meets stringent pharmaceutical specifications, providing a robust foundation for reliable supply chain continuity.
Mechanistic Insights into Safe Carbonate Formation and Protecting Group Strategy
The core mechanistic advantage of this process lies in the controlled reactivity of bis(trichloromethyl)carbonate compared to phosgene. In the conventional phosgene method, the high reactivity and gaseous nature of the reagent often lead to non-selective acylation of available hydroxyl groups, particularly the 7-OH, resulting in the cyclic carbonate impurity (II). By utilizing bis(trichloromethyl)carbonate in dichloromethane at controlled low temperatures (e.g., -10°C to 0°C) in the presence of a base like pyridine, the reaction kinetics are modulated to favor the formation of the desired 1,14-carbonate bridge exclusively. This precision minimizes side reactions and ensures that the 7-hydroxyl group remains available for the subsequent, highly selective protection step. The use of trichloroacetyl chloride then capitalizes on the electron-withdrawing nature of the trichloromethyl group to create a protecting group that is stable under coupling conditions but labile enough to be removed under mild basic conditions.
From an impurity control perspective, the solubility characteristics of the trichloroacetyl derivative are paramount. Unlike the lipophilic triethylsilyl groups which tend to stick to silica or co-crystallize, the trichloroacetamide species generated upon treatment with ammonium hydroxide exhibits distinct solubility behavior in polar aprotic solvents mixed with water. This allows for a "precipitation and wash" protocol where the desired product precipitates while the protecting group remnants remain in the supernatant. This mechanism effectively decouples the purification efficiency from the complexity of the molecular structure, ensuring that even at large scales, the impurity profile remains clean. The final crystallization from acetone/hexane further refines the lattice, excluding any remaining trace organic impurities and yielding a product that is chemically and physically homogeneous, a critical requirement for high-purity pharmaceutical intermediates intended for oncology applications.
How to Synthesize 13-(N-Boc-β-isobutylserinyl)-14-β-hydroxybaccatin III Efficiently
The synthesis of this complex taxane derivative follows a logical sequence of protection, functionalization, and coupling steps designed to maximize yield and minimize purification burden. The process begins with the selective acetylation of the 10-hydroxyl group of the starting material, 14β-hydroxy-10-deacetylbaccatin III, utilizing acetic anhydride and a Lewis acid catalyst such as cerium chloride. This sets the stage for the critical carbonate formation using the safe bis(trichloromethyl)carbonate reagent. Following this, the 7-position is protected with trichloroacetyl chloride before the side chain is coupled using standard peptide coupling conditions with DCC and DMAP. The detailed standardized synthetic steps, including specific stoichiometric ratios, temperature profiles, and workup procedures for each transformation, are outlined in the guide below.
- Selective acetylation of the 10-hydroxyl group of 14β-hydroxy-10-deacetylbaccatin III using acetic anhydride and cerium chloride.
- Formation of the 1,14-carbonate derivative using bis(trichloromethyl)carbonate at low temperature to prevent side reactions.
- Protection of the 7-hydroxyl group with trichloroacetyl chloride, followed by side-chain coupling and final deprotection via crystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented process translates directly into tangible operational efficiencies and risk mitigation. The elimination of phosgene gas removes a major regulatory and safety hurdle, simplifying facility requirements and reducing insurance and compliance costs associated with handling extremely hazardous substances. Furthermore, the shift away from column chromatography represents a massive reduction in consumable costs, as silica gel and the vast quantities of solvents required for elution are no longer needed. This process intensification allows for higher throughput in existing reactor volumes, effectively increasing capacity without capital expenditure on new equipment. The ability to purify via crystallization also shortens the batch cycle time significantly, enhancing the responsiveness of the supply chain to market demands.
- Cost Reduction in Manufacturing: The replacement of chromatographic purification with crystallization results in substantial cost savings by eliminating the need for expensive silica gel and reducing solvent recovery loads. Additionally, the use of solid bis(trichloromethyl)carbonate avoids the specialized infrastructure required for phosgene handling, lowering both CAPEX and OPEX. The high yields reported in the examples, such as 92% for the coupling step and 99% for the deprotection step, further contribute to improved material efficiency and reduced raw material costs per kilogram of final product.
- Enhanced Supply Chain Reliability: By utilizing readily available solid reagents instead of regulated toxic gases, the supply chain becomes more resilient to logistical disruptions and regulatory changes. The robustness of the crystallization-based purification ensures consistent product quality across different batches and scales, reducing the risk of batch failures or out-of-specification results that could delay shipments. This reliability is crucial for maintaining the continuous flow of materials needed for downstream API production, ensuring that drug manufacturers can meet their own production schedules without interruption.
- Scalability and Environmental Compliance: The process is inherently greener due to the reduction in hazardous waste generation, particularly the avoidance of silica gel waste contaminated with organic compounds. The simplified workup procedures involving aqueous washes and precipitations are easier to manage in large-scale wastewater treatment systems. This environmental compatibility facilitates smoother regulatory approvals for manufacturing sites and aligns with the increasing industry demand for sustainable and eco-friendly chemical processes, making it an attractive option for long-term commercial partnerships.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel taxane synthesis route. These answers are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on the practical benefits of switching from conventional phosgene-based methods to this improved protocol. Understanding these nuances is essential for technical teams evaluating the feasibility of technology transfer and for procurement teams assessing the long-term value proposition of this manufacturing strategy.
Q: Why is bis(trichloromethyl)carbonate preferred over phosgene for this synthesis?
A: Bis(trichloromethyl)carbonate is a solid surrogate for phosgene, significantly improving operational safety by eliminating the need to handle toxic gas. Furthermore, it prevents the formation of impurity (II) caused by the reaction of the 7-hydroxyl group, which is a common issue when bubbling phosgene gas into the reactor.
Q: How does the new process improve product purity compared to conventional methods?
A: The use of a trichloroacetyl protecting group at the 7-position allows for efficient removal via precipitation in water after ammonium hydroxide treatment. This avoids the difficult removal of triethylsilyl fluoride residues associated with traditional TES protection, enabling final purity levels exceeding 99.9% through simple crystallization rather than column chromatography.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process is specifically designed for scalability. By replacing hazardous gaseous reagents with solid alternatives and eliminating labor-intensive low-pressure column chromatography, the method reduces waste generation and simplifies downstream processing, making it highly viable for commercial scale-up from kilograms to metric tons.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 13-(N-Boc-β-isobutylserinyl)-14-β-hydroxybaccatin III Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the development and production of life-saving oncology drugs. Our technical team has extensively analyzed the pathway described in CN101448839B and possesses the expertise to implement this advanced synthesis route effectively. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. Our facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of 13-(N-Boc-β-isobutylserinyl)-14-β-hydroxybaccatin III meets the highest international standards for pharmaceutical intermediates.
We invite global pharmaceutical partners to collaborate with us to leverage this superior manufacturing technology. By contacting our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our adoption of this phosgene-free, chromatography-free process can optimize your supply chain and reduce overall manufacturing costs while ensuring a steady supply of high-purity materials.
