Advanced Homogeneous Silylation Strategy for Commercial Scale Cefpiramide Acid Production
Advanced Homogeneous Silylation Strategy for Commercial Scale Cefpiramide Acid Production
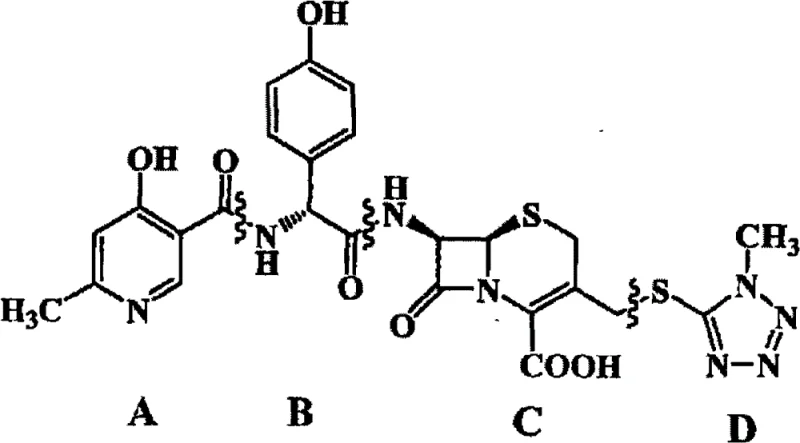
The pharmaceutical industry continuously seeks robust synthetic routes for third-generation cephalosporins to ensure consistent supply and high purity standards. Patent CN103059048A introduces a significant methodological advancement in the preparation of Cefpiramide acid, a critical active pharmaceutical ingredient (API) intermediate. This technology addresses long-standing challenges in the acylation of the cephalosporin nucleus by replacing traditional heterogeneous silylation protocols with a novel homogeneous system utilizing N,O-Bistrimethylsilylacetamide (BSA). By shifting from suspension-based reactions to clear solution chemistry, this process dramatically improves reaction completeness and downstream processing efficiency. The structural complexity of Cefpiramide acid requires precise control over stereochemistry and functional group protection, which this patent elegantly solves through optimized solvent systems and catalytic conditions. As a reliable cefpiramide acid supplier, understanding these mechanistic nuances is vital for scaling production while maintaining stringent quality specifications required by global regulatory bodies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Cefpiramide acid has relied on the use of trimethylchlorosilane (TMCS) combined with triethylamine in dichloromethane to protect the amino group of the 7-ATCA nucleus. This conventional approach suffers from inherent physical limitations, primarily because the resulting reaction mixture forms a suspension rather than a true solution. In a heterogeneous system, mass transfer is restricted, leading to incomplete silylation of the amino group, which can subsequently result in polymerization or unwanted side reactions during the acylation step. Furthermore, the workup procedure for these traditional methods is notoriously difficult; transferring the reaction mixture into aqueous systems often causes rapid oil agglomeration due to the presence of unreacted species and emulsions. This phenomenon necessitates vigorous stirring and complex filtration steps, which are detrimental to large-scale manufacturing where reproducibility is paramount. The inability to guarantee complete protection in a suspension directly correlates with lower overall yields and increased impurity profiles, posing significant risks for cost reduction in antibiotic manufacturing.
The Novel Approach
The innovative methodology disclosed in the patent utilizes N,O-Bistrimethylsilylacetamide (BSA) as the silylating agent, which fundamentally alters the physical state of the reaction medium. Upon addition of BSA to 7-ATCA in dichloromethane, the mixture transforms into a clear, homogeneous solution, indicating complete dissolution and effective protection of the reactive amino site. This transition to a homogeneous phase ensures that every molecule of the cephalosporin nucleus is equally accessible for the subsequent acylation with the activated side chain acid. The clarity of the solution serves as a visual indicator of reaction progress, allowing for tighter process control and eliminating the guesswork associated with suspended solids. Moreover, the deprotection step is streamlined; the silyl groups are easily removed via simple hydrolysis with sodium bicarbonate solution, avoiding the harsh conditions often required to break down more stubborn protecting groups. This operational simplicity translates directly into enhanced supply chain reliability, as the process becomes less sensitive to minor variations in mixing or temperature, ensuring consistent batch-to-batch quality.
Mechanistic Insights into BF3-Catalyzed Substitution and BSA Protection
A critical component of this synthetic route involves the high-efficiency preparation of the 7-ATCA precursor itself, which serves as the foundational building block for the final API. The patent details a superior substitution reaction where 7-Aminocephalosporanic acid (7-ACA) reacts with 1-methyl-1H-tetrazole-5-thiol (MMT) in the presence of a boron trifluoride acetonitrile complex catalyst. Unlike traditional methods employing concentrated sulfuric acid, which can degrade the sensitive beta-lactam ring, the BF3 catalyst operates under mild thermal conditions (28-32°C). This Lewis acid catalysis facilitates the nucleophilic attack of the thiol on the acetoxy methyl group at the C-3 position of the cephalosporin nucleus with high regioselectivity. The mechanism avoids the generation of excessive heat and acidic byproducts that typically lead to ring opening or epimerization, thereby preserving the stereochemical integrity of the molecule. The result is a precursor with exceptional purity (98.9% by HPLC) and a molar yield of 0.912, significantly outperforming the 0.826 yield observed in acid-catalyzed comparative examples.
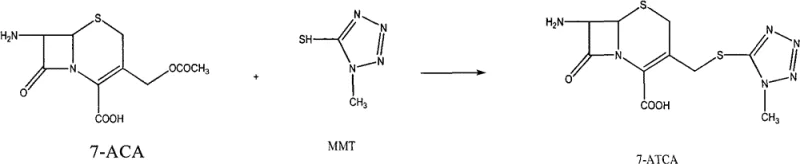
Following the synthesis of the nucleus, the protection mechanism using BSA is equally sophisticated in its simplicity. BSA acts as a potent silyl donor, transferring trimethylsilyl groups to the primary amine at the C-7 position of the 7-ATCA. This protection is crucial because the free amine is highly nucleophilic and would otherwise interfere with the activation of the side chain carboxylic acid or react with itself. By masking the amine as a silyl derivative, the molecule becomes soluble in organic solvents like dichloromethane, enabling the homogeneous reaction environment described earlier. The stability of the silyl-amine bond is balanced perfectly; it is stable enough to withstand the low-temperature acylation conditions (-60°C to -50°C) but labile enough to be cleaved rapidly upon exposure to mild aqueous base during the workup. This precise control over reactivity minimizes the formation of di-acylated byproducts or hydrolyzed beta-lactam impurities, ensuring that the final crude product achieves a purity of 96% before further purification steps.
How to Synthesize Cefpiramide Acid Efficiently
The execution of this synthesis requires strict adherence to temperature profiles and stoichiometric ratios to maximize the benefits of the homogeneous system. The process begins with the dissolution of 7-ATCA in dichloromethane, followed by the controlled addition of BSA to achieve a clear solution, confirming successful protection. Simultaneously, the side chain acid (HOPG-NAD) is activated in DMF using ethyl chloroformate and N-methylmorpholine at cryogenic temperatures to form a mixed anhydride. The coupling step involves adding the silylated 7-ATCA solution to the activated side chain, maintaining the temperature between -60°C and -50°C to prevent thermal degradation. After the acylation is complete, the reaction is warmed to 0-10°C, and the protecting groups are removed via hydrolysis with sodium bicarbonate. The detailed standardized synthesis steps see the guide below.
- Dissolve 7-ATCA in dichloromethane and treat with N,O-Bistrimethylsilylacetamide (BSA) to form a clear homogeneous silylated solution.
- Activate the side chain acid (HOPG-NAD) with ethyl chloroformate and N-methylmorpholine in DMF at low temperature (-60°C).
- Couple the silylated 7-ATCA with the activated side chain, followed by hydrolysis and pH-controlled crystallization to isolate the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this BSA-mediated process offers tangible operational improvements that extend beyond mere chemical yield. The elimination of heterogeneous reaction conditions removes a major bottleneck in manufacturing throughput, as filtration of suspensions and handling of sludge-like intermediates are time-consuming and prone to equipment fouling. By operating in a homogeneous phase, the process facilitates easier pumping and transfer of materials between reactors, reducing cycle times and labor costs associated with manual intervention. Furthermore, the improved crystallization behavior, characterized by the absence of oil agglomeration, ensures that the isolation of the final product is predictable and scalable. This predictability is a key factor in reducing lead time for high-purity pharmaceutical intermediates, as it minimizes the risk of batch failures or the need for reprocessing due to poor solid formation.
- Cost Reduction in Manufacturing: The adoption of the BF3-catalyzed route for 7-ATCA and the BSA protection strategy eliminates the need for expensive and hazardous concentrated sulfuric acid handling systems. The higher molar yields achieved in both the precursor and final coupling steps mean that less raw material is wasted per kilogram of finished product. Additionally, the simplified workup procedure reduces the consumption of solvents and washing agents, as there is no need for extensive extraction to remove emulsified oils. These cumulative efficiencies drive substantial cost savings without compromising the quality of the cefpiramide acid produced.
- Enhanced Supply Chain Reliability: The robustness of the homogeneous reaction system makes the supply chain less vulnerable to variability. Since the reaction proceeds to completion more reliably than suspension-based methods, the risk of off-spec batches is significantly mitigated. This consistency allows for tighter inventory planning and more dependable delivery schedules to downstream API manufacturers. The use of stable intermediates and straightforward crystallization protocols ensures that production can be ramped up or down based on market demand without encountering the technical difficulties often associated with scaling heterogeneous chemistries.
- Scalability and Environmental Compliance: From an environmental perspective, the replacement of strong mineral acids with Lewis acid catalysts and the reduction in solvent usage align with green chemistry principles. The process generates less acidic waste stream, simplifying effluent treatment and reducing the environmental footprint of the manufacturing facility. The ability to scale this process from laboratory to commercial production is enhanced by the clear visual indicators of reaction progress (solution clarity), allowing operators to make real-time decisions that maintain safety and quality standards throughout the scale-up phases.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation of this patented synthesis route. These insights are derived directly from the experimental data and comparative examples provided in the patent documentation, offering a transparent view of the process capabilities. Understanding these details helps stakeholders evaluate the feasibility of adopting this technology for their specific manufacturing needs.
Q: Why is N,O-Bistrimethylsilylacetamide (BSA) preferred over TMCS for 7-ATCA protection?
A: BSA creates a homogeneous clear solution in dichloromethane, whereas traditional TMCS/triethylamine systems form suspensions. This homogeneity ensures complete protection of the amino group, preventing side reactions and simplifying the subsequent workup by avoiding oil agglomeration during crystallization.
Q: How does the BF3-Acetonitrile catalyst improve 7-ATCA synthesis?
A: Using boron trifluoride acetonitrile complex instead of concentrated sulfuric acid allows for milder reaction conditions (28-32°C) and eliminates the need for harsh acidic workups. This results in a higher molar yield (0.912 vs 0.826) and improved product purity (98.9% vs 98.5%).
Q: What are the critical control points for crystallization in this process?
A: Critical control points include maintaining the hydrolysis temperature between 0-10°C and precisely adjusting the pH to 1-2 during the final acidification step. Slow addition of acid and controlled cooling are essential to prevent oiling out and ensure the formation of high-purity crystals.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cefpiramide Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced synthetic routes requires a partner with deep technical expertise and proven manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patents like CN103059048A are fully realized in practice. We maintain stringent purity specifications and operate rigorous QC labs equipped to analyze complex cephalosporin impurity profiles, guaranteeing that every batch of Cefpiramide acid meets the highest international standards. Our commitment to quality assurance means that we can consistently deliver high-purity intermediates that facilitate smoother downstream API synthesis for our global clients.
We invite you to collaborate with us to leverage these technological advancements for your supply chain. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our optimized processes can enhance your production efficiency and reduce overall manufacturing costs.
