Advanced Production Method for High-Purity 1,2-N,N'-Dimethylcyclohexanediamine Intermediates
The pharmaceutical and fine chemical industries are constantly seeking robust synthetic routes that balance safety, cost-efficiency, and product purity, particularly for complex amine intermediates. Patent CN102731316B introduces a significant advancement in the production of 1,2-N,N'-dimethylcyclohexanediamine, a valuable building block in organic synthesis. This invention discloses a method that replaces hazardous reduction protocols with a safer, more controllable sequence involving N-single protection, methylation, and hydrolysis. By utilizing aromatic benzene sulfonyl chloride for protection and standard methylating agents, the process circumvents the dangers associated with strong reducing agents while ensuring high selectivity. For R&D directors and procurement managers, this represents a critical shift towards more sustainable and scalable manufacturing practices that align with modern safety standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of N,N'-dialkylated cyclohexanediamines has relied heavily on methods described in literature such as the Journal of the American Chemical Society (2002), which utilize chloroformic esters or formates followed by reduction with lithium aluminum hydride. This conventional approach presents severe drawbacks for large-scale industrial application, primarily due to the inherent dangers of handling lithium aluminum hydride, a potent and moisture-sensitive reductant that requires stringent anhydrous conditions. Furthermore, the selectivity of the reduction step is often poor, leading to complex mixtures of by-products that are difficult to separate, thereby driving up purification costs and reducing overall yield. The high cost of raw materials combined with the safety risks creates a bottleneck for reliable supply chain continuity, making it an unattractive option for commercial scale-up of complex pharmaceutical intermediates.
The Novel Approach
In stark contrast, the novel approach detailed in the patent employs a strategic protective group methodology that fundamentally alters the reaction landscape to favor safety and selectivity. By first performing an N-single protection on 1,2-diaminocyclohexane using aromatic benzene sulfonyl chloride, the reactivity of the amine groups is precisely modulated, allowing for controlled subsequent methylation. This pathway eliminates the need for dangerous hydride reductions entirely, replacing them with standard alkylation and acid hydrolysis steps that are far easier to manage in a plant setting. The result is a process that not only simplifies operations but also significantly enhances the purity profile of the final product, addressing the critical pain points of impurity control and operational hazard that plague the older methodologies.
Mechanistic Insights into Sulfonyl Protection and Methylation Strategy
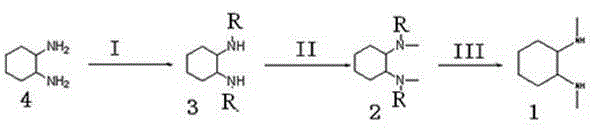
The core of this synthetic innovation lies in the precise manipulation of nucleophilicity through sulfonyl protection. In the initial step, 1,2-diaminocyclohexane reacts with Tosyl chloride in a basic medium such as pyridine or triethylamine. The reaction conditions are carefully controlled, typically maintaining temperatures below 100°C, and preferably between 10-35°C, to ensure mono-protection rather than bis-protection. This selectivity is crucial because it leaves one amine group free for the subsequent methylation while the other remains shielded by the bulky sulfonyl group. The mechanism involves the nucleophilic attack of the amine nitrogen on the sulfur atom of the sulfonyl chloride, displacing the chloride ion and forming a stable sulfonamide bond that withstands the subsequent alkylation conditions.
Following protection, the free amine undergoes N-methylation using methyl iodide or dimethyl sulphate in the presence of a base like potassium carbonate. This step introduces the methyl group specifically to the unprotected nitrogen. The final stage involves a hydrolysis reaction in the presence of sulfuric acid, where the temperature is raised to between 60-150°C. Under these acidic conditions, the sulfonamide bond is cleaved, removing the aromatic benzenesulfonyl group and regenerating the free amine functionality, thus yielding the target 1,2-N,N'-dimethylcyclohexanediamine. This mechanistic pathway ensures that methylation occurs selectively and that the final deprotection is clean, minimizing side reactions.
How to Synthesize 1,2-N,N'-Dimethylcyclohexanediamine Efficiently
To implement this synthesis effectively, operators must adhere to strict temperature controls and stoichiometric ratios during the protection and methylation phases. The process begins with the dissolution of the diamine in a suitable solvent, followed by the dropwise addition of the sulfonyl chloride solution to manage exothermicity. After the protection phase, the intermediate is isolated, often by pH adjustment and filtration, before being redissolved for the methylation step which requires cooling to prevent over-alkylation or degradation. The detailed standardized synthesis steps, including specific molar ratios, solvent volumes, and workup procedures such as extraction and distillation, are outlined in the guide below to ensure reproducibility and high yield.
- Perform N-mono-protection on 1,2-diaminocyclohexane using aromatic benzene sulfonyl chloride in pyridine or triethylamine solvent at controlled temperatures below 100°C.
- Conduct N-methylation on the protected intermediate using methyl iodide or dimethyl sulphate in the presence of potassium carbonate under ice bath conditions.
- Execute hydrolysis using sulfuric acid at elevated temperatures (60-150°C) to remove the aromatic benzenesulfonyl group, followed by neutralization and extraction.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this sulfonyl-based synthesis route offers profound logistical and economic benefits that extend beyond simple yield metrics. By eliminating the dependency on lithium aluminum hydride, manufacturers remove a major safety liability and a costly specialty reagent from their supply chain, thereby reducing the complexity of hazardous material handling and storage requirements. The use of commodity chemicals like Tosyl chloride and methyl iodide ensures a more stable and predictable sourcing landscape, mitigating the risk of supply disruptions that are common with specialized reducing agents. Furthermore, the simplified workup procedures, which rely on standard acid-base extractions and crystallizations rather than complex chromatographic separations, drastically reduce processing time and solvent consumption.
- Cost Reduction in Manufacturing: The elimination of expensive and hazardous reducing agents like lithium aluminum hydride leads to substantial cost savings in raw material procurement and waste disposal. Since the new method utilizes widely available industrial chemicals and avoids the need for specialized anhydrous infrastructure required for hydride reductions, the capital expenditure and operational overhead are significantly lowered. Additionally, the improved selectivity reduces the burden on downstream purification processes, meaning less solvent and energy are consumed per kilogram of final product, driving down the overall cost of goods sold.
- Enhanced Supply Chain Reliability: Sourcing reliability is greatly improved as the key reagents, such as aromatic benzene sulfonyl chlorides and methylating agents, are produced on a massive global scale with consistent quality. Unlike the older method which relies on reagents that may have limited suppliers or strict transport regulations due to their reactivity, this new route utilizes stable intermediates that are easier to stockpile and transport. This stability translates directly into shorter lead times for high-purity pharmaceutical intermediates, allowing manufacturers to respond more agilely to market demand fluctuations without compromising on safety or quality.
- Scalability and Environmental Compliance: The process is inherently designed for commercial scale-up, as the reaction conditions (temperature and pressure) are mild and compatible with standard stainless steel reactors. The avoidance of heavy metal waste or difficult-to-treat hydride by-products simplifies environmental compliance and wastewater treatment protocols. This makes the technology highly attractive for facilities aiming to expand production capacity from pilot scale to multi-ton annual output while adhering to increasingly stringent environmental regulations regarding chemical discharge and worker safety.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These answers are derived directly from the technical specifications and comparative data provided in the patent documentation, focusing on the practical implications for industrial adoption. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this method into existing production lines.
Q: Why is the sulfonyl protection method preferred over lithium aluminum hydride reduction?
A: The traditional method utilizing lithium aluminum hydride poses significant safety risks due to the use of strong reductants and often results in poor product selectivity. The novel sulfonyl protection route avoids dangerous reagents, offers better operational safety, and achieves superior selectivity for industrial applications.
Q: What purity levels can be achieved with this production method?
A: According to the patent data, this optimized synthetic route allows for the production of the target product with a content reaching up to 98.0%. The process includes specific crystallization and pH adjustment steps that facilitate the removal of impurities effectively.
Q: What are the key reagents required for this synthesis?
A: The core reagents include 1,2-diaminocyclohexane as the starting material, aromatic benzene sulfonyl chloride (such as Tosyl chloride) for protection, and methylating agents like methyl iodide or dimethyl sulphate. Sulfuric acid is subsequently used for the deprotection hydrolysis step.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,2-N,N'-Dimethylcyclohexanediamine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting safer and more efficient synthetic routes for high-value intermediates like 1,2-N,N'-dimethylcyclohexanediamine. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale innovation to industrial reality is seamless. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch meets the exacting standards required by global pharmaceutical and agrochemical clients who demand consistency and reliability in their supply chain.
We invite you to collaborate with us to leverage this advanced technology for your specific project needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating exactly how this optimized route can improve your margins. Please contact us today to request specific COA data and route feasibility assessments, and let us help you secure a stable, high-quality supply of this essential chemical building block.
