Advanced Manufacturing of Trinexapac-ethyl: Technical Insights for Global Supply Chains
Advanced Manufacturing of Trinexapac-ethyl: Technical Insights for Global Supply Chains
The global demand for efficient plant growth regulators continues to surge, driven by the need for optimized crop yields in modern agriculture. At the forefront of this sector is Trinexapac-ethyl, a potent gibberellin biosynthesis inhibitor widely used to control stem elongation in cereals and turf. A pivotal advancement in the manufacturing landscape is detailed in patent CN102101830A, which outlines a robust, high-yield synthetic pathway that addresses historical challenges in purity and scalability. This technical report analyzes the proprietary methodology disclosed in the patent, offering R&D directors and procurement specialists a deep dive into a process that promises significant operational efficiencies. By leveraging a sequence of condensation, cyclization, esterification, and rearrangement reactions, this route eliminates the need for complex purification of intermediates, thereby streamlining the production workflow. For stakeholders seeking a reliable agrochemical intermediate supplier, understanding the mechanistic underpinnings and commercial viability of this specific patent is crucial for securing long-term supply chain stability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of cyclohexane dicarboxylic acid derivatives like Trinexapac-ethyl has been plagued by inefficiencies that hinder large-scale adoption. Traditional routes often suffer from severe reaction conditions that require extreme temperatures or pressures, leading to safety concerns and increased energy consumption. Furthermore, legacy methods frequently exhibit low yields due to side reactions and incomplete conversions, necessitating extensive downstream purification processes that erode profit margins. The accumulation of impurities at each stage often requires chromatographic separation or multiple recrystallizations, which not only increases solvent waste but also extends the overall production lead time. In the context of cost reduction in plant growth regulator manufacturing, these bottlenecks represent a significant barrier to entry for generic manufacturers aiming to compete on price while maintaining quality standards.
The Novel Approach
The methodology presented in patent CN102101830A introduces a paradigm shift by optimizing reaction parameters to achieve mild conditions and superior selectivity. The process initiates with a high-pressure condensation of diethyl maleate and acetone, catalyzed by diethylamine, which proceeds with remarkable efficiency at controlled temperatures between 160°C and 165°C. Unlike conventional approaches that might isolate unstable intermediates, this novel route employs a telescoped strategy where the crude product from the condensation step is directly subjected to cyclization. This elimination of isolation steps significantly reduces material handling losses and solvent usage. The subsequent transformation involves a precise ring-closing reaction followed by esterification and a final rearrangement, all designed to maximize atom economy. By integrating these steps into a cohesive flow, the process achieves a final product purity exceeding 96% without the need for intermediate purification, marking a substantial improvement over prior art.
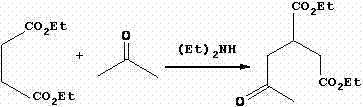
Mechanistic Insights into the Multi-Step Synthesis
The core of this synthetic route lies in the precise execution of a Michael addition followed by an intramolecular Claisen condensation, commonly known as the Dieckmann condensation. In the first stage, diethyl maleate acts as the Michael acceptor while acetone serves as the donor in the presence of a secondary amine base. This reaction forms the linear 2-acetone-1,4-succinic acid diethyl ester, which serves as the foundational scaffold for the cyclohexane ring. The success of this step relies heavily on the molar ratio of reactants, specifically maintaining an excess of acetone to drive the equilibrium forward. Following this, the introduction of sodium hydride in dimethylformamide (DMF) triggers the cyclization. The strong base deprotonates the alpha-carbon adjacent to the ketone, facilitating a nucleophilic attack on the ester carbonyl to close the six-membered ring. This generates a stable enolate species, specifically the 3-ethoxycarbonyl-5-oxocyclohexyl-1-ene-1-sodium alkoxide, which is critical for the subsequent functionalization.
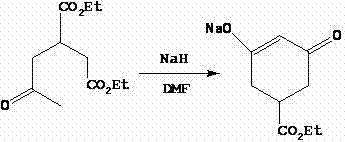
Following the ring formation, the synthesis proceeds with an acylation step where the sodium alkoxide intermediate reacts with cyclopropanecarboxylic acid chloride. This step must be conducted at low temperatures, typically between 10°C and 20°C, to prevent thermal degradation of the sensitive enolate and to control the exothermic nature of the acylation. The resulting ester retains the cyclopropane moiety essential for the biological activity of the final herbicide. The final transformation is a rearrangement reaction mediated by a cyanide source, such as cupric cyanide or potassium cyanide, in the presence of triethylamine. This step effectively migrates the double bond and stabilizes the conjugated system, yielding the final Trinexapac-ethyl structure. The choice of cyanide catalyst and the precise control of the reaction temperature at 80-90°C are paramount to ensuring high conversion rates and minimizing the formation of regio-isomers.
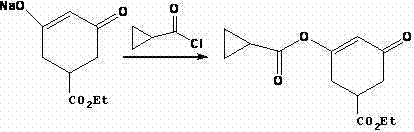
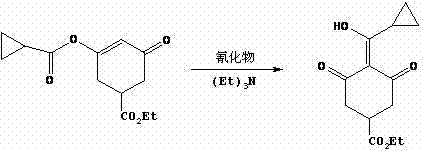
How to Synthesize Trinexapac-ethyl Efficiently
Implementing this synthesis requires strict adherence to the thermal profiles and stoichiometric ratios defined in the patent data to ensure reproducibility and safety. The process begins with loading diethyl maleate, acetone, and diethylamine into an autoclave, where the mixture is heated to initiate the condensation. Once the linear precursor is formed, the reaction mass is transferred or treated in situ with sodium hydride under an inert atmosphere to effect cyclization. The subsequent addition of the acid chloride must be dropwise to manage heat generation, followed by the final rearrangement step which requires careful monitoring via TLC to determine the endpoint. For a detailed breakdown of the specific operational parameters, including exact addition rates and workup procedures, please refer to the standardized guide below.
- Perform high-pressure condensation of diethyl maleate and acetone with diethylamine at 160-165°C to obtain 2-acetone-1,4-succinic acid diethyl ester.
- Execute ring formation using sodium hydride in DMF at 80-90°C to generate the sodium alkoxide intermediate.
- React the alkoxide with cyclopropanecarboxylic acid chloride at low temperature followed by rearrangement with cyanide and triethylamine to finalize the product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this patented synthesis route offers transformative benefits for procurement managers and supply chain directors focused on commercial scale-up of complex agrochemical intermediates. The most significant advantage is the telescoped nature of the process, which eliminates the isolation and purification of intermediates. In traditional manufacturing, each isolation step incurs costs related to filtration, drying, and solvent recovery, alongside inevitable yield losses due to mechanical transfer. By bypassing these unit operations, the overall process mass intensity (PMI) is drastically reduced, leading to substantial cost savings in raw materials and waste disposal. Furthermore, the high yields reported in the patent embodiments—often exceeding 90% for key steps—ensure that the theoretical output is closely matched by actual production, maximizing the return on investment for raw material procurement.
- Cost Reduction in Manufacturing: The elimination of intermediate purification steps directly translates to lower operational expenditures. Without the need for multiple recrystallizations or column chromatography between steps, the consumption of solvents and energy is significantly curtailed. Additionally, the use of readily available starting materials like diethyl maleate and acetone ensures that the bill of materials remains stable and predictable, shielding the supply chain from volatility associated with exotic reagents. The high atom economy of the rearrangement step further contributes to cost efficiency by minimizing waste generation.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route enhances supply continuity by reducing the risk of batch failures. The mild reaction conditions, particularly in the cyclization and acylation steps, reduce the stress on reactor equipment and lower the probability of runaway reactions or safety incidents that could halt production. Moreover, the flexibility in choosing the cyanide source (cupric, potassium, or sodium cyanide) allows manufacturers to adapt to regional availability of reagents, preventing bottlenecks caused by the shortage of a specific catalyst grade.
- Scalability and Environmental Compliance: This process is explicitly designed for industrial scalability, having been validated in autoclaves and standard reactors suitable for multi-ton production. The reduction in solvent usage and waste generation aligns with increasingly stringent environmental regulations, simplifying the permitting process for new manufacturing lines. By minimizing the discharge of organic waste and reducing the carbon footprint associated with energy-intensive purification steps, manufacturers can achieve better sustainability metrics, which is a growing requirement for partnerships with major global agrochemical corporations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These insights are derived directly from the experimental data and claims within patent CN102101830A, providing a factual basis for decision-making. Understanding these nuances is essential for technical teams evaluating the feasibility of adopting this method for reducing lead time for high-purity plant growth regulators.
Q: What are the critical reaction conditions for the cyclization step?
A: The cyclization step requires strict temperature control between 80-90°C using sodium hydride in DMF. The addition of the succinate derivative must be split into two portions to manage exothermicity and ensure complete conversion.
Q: How is product purity maintained without intermediate purification?
A: The process utilizes a telescoped synthesis strategy where crude intermediates are directly used in subsequent steps. Final purity exceeding 96% is achieved through rigorous control of the final rearrangement step and subsequent recrystallization.
Q: Is this method suitable for large-scale industrial production?
A: Yes, the patent explicitly states suitability for industrial production. The use of autoclaves for the initial condensation and standard reactors for subsequent steps allows for scalable manufacturing from 100 kgs to multi-ton quantities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Trinexapac-ethyl Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient synthesis routes in maintaining a competitive edge in the agrochemical market. Our team of expert chemists has extensively analyzed the pathway described in CN102101830A and possesses the technical capability to execute this complex sequence with precision. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and reliability. Our facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of Trinexapac-ethyl meets the highest international standards for efficacy and safety.
We invite you to collaborate with us to optimize your supply chain and reduce manufacturing costs. Our technical sales team is prepared to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact our technical procurement team to request specific COA data and comprehensive route feasibility assessments. Let us partner with you to secure a sustainable and cost-effective source of high-quality agrochemical intermediates.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
