Advanced Grignard-Based Synthesis of Dicyclohexyl Dithiophosphinic Acid for Industrial Flotation
The chemical landscape of metallurgical flotation agents is undergoing a significant transformation driven by the need for safer, more efficient, and environmentally compliant synthesis routes. Patent CN101343287B presents a robust and innovative methodology for the production of dicyclohexyl dithiophosphinic acid, a specialized organophosphorus compound critical for the separation of lanthanides and actinides in hydrometallurgical processes. This technology addresses the longstanding challenges associated with the toxicity and volatility of precursor materials found in conventional syntheses. By leveraging a Grignard-based coupling strategy followed by a sophisticated sulfidation and purification sequence, this patent outlines a pathway that not only enhances operator safety but also streamlines the manufacturing workflow. For procurement specialists and R&D leaders in the mining chemical sector, understanding this proprietary route is essential for securing a reliable supply chain of high-performance collectors that meet increasingly rigorous global safety standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of dialkyl dithiophosphinic acids has relied heavily on methodologies that pose significant occupational health and safety risks. Traditional pathways often involve the direct utilization of secondary phosphines, which are characterized by their extreme volatility and intense, unpleasant odor. These physical properties necessitate elaborate containment systems and specialized personal protective equipment, driving up the operational expenditure for manufacturers. Furthermore, alternative routes utilizing aluminum chloride catalysis with phosphorus pentasulfide can lead to complex reaction mixtures that are difficult to purify, often resulting in lower yields and higher levels of inorganic waste. The handling of such hazardous precursors creates a bottleneck in commercial scale-up, limiting the ability of suppliers to respond flexibly to market demand while maintaining strict environmental compliance protocols regarding volatile organic compound emissions.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes a Grignard reagent derived from bromocyclohexane as the foundational building block, effectively bypassing the need for free secondary phosphines. This strategic shift allows for the formation of a stable bis(dicyclohexylphosphino) disulfide intermediate through a controlled reaction with thiophosphoryl chloride. The subsequent sulfidation step using sodium sulfide and elemental sulfur in a dioxane solvent system provides a clean and efficient conversion to the target sodium salt. This methodology not only mitigates the toxicity risks associated with earlier technologies but also simplifies the downstream processing. The ability to generate the target molecule through a sequence of well-defined, high-yielding steps represents a substantial advancement in the manufacturing of complex organophosphorus ligands, offering a clear pathway for cost reduction in mining chemical manufacturing through improved process efficiency.
Mechanistic Insights into Grignard-Mediated Phosphorus-Sulfur Coupling
The core of this synthetic innovation lies in the precise control of the phosphorus-carbon bond formation and the subsequent manipulation of the phosphorus-sulfur framework. The process initiates with the formation of cyclohexylmagnesium bromide, where the careful addition of bromocyclohexane to magnesium turnings in anhydrous diethyl ether ensures the generation of a highly reactive nucleophile. This Grignard reagent is then reacted with thiophosphoryl chloride at low temperatures, specifically maintained between 0°C and 10°C, to prevent runaway exotherms and ensure selectivity. This step results in the coupling of two phosphorus centers to form a tetra-cyclohexyl-diphosphine-disulfide dimer, a unique intermediate that serves as the reservoir for the final active species. The stability of this dimer allows for safe isolation and handling, a critical feature for industrial logistics.
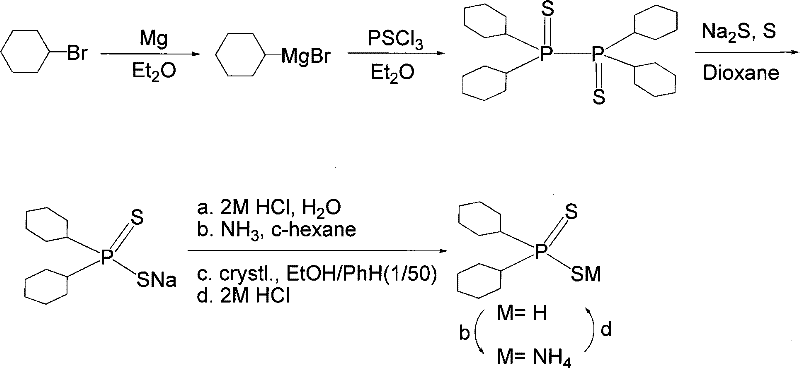
Following the formation of the dimer, the mechanism proceeds through a cleavage and sulfidation reaction. The addition of sodium sulfide nonahydrate and elemental sulfur in 1,4-dioxane under reflux conditions facilitates the breakdown of the P-P bond and the introduction of the dithio functionality. This transforms the neutral dimer into the anionic sodium dicyclohexyl dithiophosphinate. The final stage involves a sophisticated purification protocol where the crude acid is converted into an ammonium salt. This salt formation is pivotal for impurity control, as it allows for recrystallization from an ethanol and benzene mixture, effectively segregating organic byproducts. The final acidification with hydrochloric acid regenerates the free dithiophosphinic acid with high structural integrity, ensuring that the product possesses the necessary coordination chemistry properties for effective metal ion complexation in flotation applications.
How to Synthesize Dicyclohexyl Dithiophosphinic Acid Efficiently
The execution of this synthesis requires strict adherence to anhydrous conditions during the initial Grignard formation and precise temperature control during the phosphorus coupling phase. Operators must ensure that the magnesium activation is complete before proceeding to avoid unreacted halide contamination. The subsequent sulfidation step demands careful management of the sulfur load and reflux duration to maximize the conversion of the dimer intermediate. For a comprehensive understanding of the specific molar ratios, solvent volumes, and workup procedures required to replicate this high-purity process, please refer to the standardized technical guide provided below.
- Preparation of Cyclohexylmagnesium Bromide: React bromocyclohexane with magnesium strips in anhydrous diethyl ether under reflux conditions initiated by iodine.
- Formation of Phosphorus Intermediate: React the Grignard reagent with thiophosphoryl chloride (PSCl3) at 0-10°C to form the tetra-cyclohexyl-diphosphine-disulfide dimer.
- Sulfidation and Salt Formation: Treat the dimer intermediate with sodium sulfide and sulfur powder in dioxane under reflux to generate the sodium dithiophosphinate salt.
- Purification and Acidification: Convert the crude acid to an ammonium salt, recrystallize from ethanol/benzene, and finally acidify with HCl to obtain the pure free acid.
Commercial Advantages for Procurement and Supply Chain Teams
From a supply chain perspective, the adoption of this Grignard-based synthesis route offers profound advantages in terms of raw material security and logistical stability. The primary inputs, bromocyclohexane and thiophosphoryl chloride, are commodity chemicals produced on a massive global scale, ensuring that supply disruptions are minimal compared to routes relying on niche or hazardous specialty phosphines. This abundance translates directly into enhanced supply chain reliability, allowing manufacturers to maintain consistent production schedules even during periods of market volatility. Furthermore, the elimination of highly volatile and toxic secondary phosphines reduces the regulatory burden and insurance costs associated with storage and transport, creating a leaner and more resilient operational model for the production of high-purity organophosphorus compounds.
- Cost Reduction in Manufacturing: The streamlined nature of this synthetic route eliminates the need for expensive catalysts and complex waste treatment protocols associated with aluminum chloride-based methods. By avoiding the handling of hazardous volatile phosphines, facilities can significantly reduce expenditures on specialized ventilation systems and personal protective equipment. The high efficiency of the purification step, utilizing ammonium salt recrystallization, minimizes product loss and reduces the volume of solvent waste generated per kilogram of output. These cumulative efficiencies drive down the overall cost of goods sold, making the final flotation agent more competitive in the global market without compromising on quality or performance specifications.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions, which rely on standard reflux and stirring rather than high-pressure or cryogenic setups, ensures that the manufacturing process is easily transferable across different production sites. This flexibility mitigates the risk of single-point failures in the supply network. Additionally, the stability of the key dimer intermediate allows for potential stockpiling, providing a buffer against short-term fluctuations in raw material availability. For procurement managers, this means a more predictable lead time for high-purity mining chemicals, enabling better inventory planning and reducing the need for costly emergency sourcing strategies.
- Scalability and Environmental Compliance: The process is inherently scalable, moving seamlessly from laboratory benchtop quantities to multi-ton commercial production without requiring fundamental changes to the reaction engineering. The use of closed systems for the Grignard and sulfidation steps ensures that emissions are contained, aligning with strict environmental regulations regarding sulfur and phosphorus compounds. The aqueous workup and acidification steps generate waste streams that are easier to treat compared to those containing heavy metal catalysts. This environmental compatibility not only future-proofs the manufacturing asset against tightening regulations but also enhances the corporate sustainability profile of the supply chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of dicyclohexyl dithiophosphinic acid. These insights are derived directly from the experimental data and process descriptions within the patent literature, providing a factual basis for evaluating the technology's viability. Understanding these details is crucial for technical teams assessing the feasibility of integrating this material into their existing hydrometallurgical workflows.
Q: Why is the Grignard-based route safer than traditional secondary phosphine methods?
A: Traditional methods often utilize secondary phosphines which are highly volatile, possess intense pungency, and exhibit significant toxicity. The patented Grignard route avoids handling these hazardous free phosphines, significantly improving operational safety and reducing environmental containment costs.
Q: What is the expected purity profile of the final dicyclohexyl dithiophosphinic acid?
A: The process employs a unique purification strategy involving conversion to an ammonium salt followed by recrystallization from an ethanol/benzene mixture. This step effectively removes organic impurities and unreacted intermediates, ensuring the final product meets stringent specifications required for high-efficiency metal flotation.
Q: Can this synthesis method be scaled for industrial production of flotation collectors?
A: Yes, the method utilizes readily available commodity chemicals such as bromocyclohexane and thiophosphoryl chloride. The reaction conditions, primarily involving standard reflux and controlled temperature additions, are well-suited for scale-up in standard glass-lined or stainless steel reactors without requiring exotic high-pressure equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Dicyclohexyl Dithiophosphinic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-performance flotation agents play in the efficiency of modern mineral processing operations. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the sophisticated Grignard-based synthesis described in CN101343287B can be executed with precision at an industrial level. We are committed to delivering products that meet stringent purity specifications, supported by our rigorous QC labs which utilize advanced spectroscopic methods to verify the structural integrity and absence of toxic impurities in every batch.
We invite you to collaborate with us to optimize your supply chain for metallurgical reagents. Our experts are ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and application needs. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us demonstrate how our advanced manufacturing capabilities can support your operational excellence goals.
