Advanced Synthesis of 2,2'-Biazaaryl Bidentate Ligands for Catalytic Applications
Advanced Synthesis of 2,2'-Biazaaryl Bidentate Ligands for Catalytic Applications
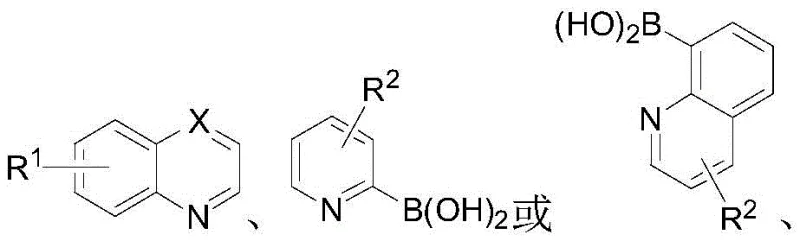
The landscape of organometallic catalysis is constantly evolving, driven by the demand for more efficient, sustainable, and cost-effective ligand systems. A pivotal advancement in this field is documented in Chinese Patent CN112778272B, which discloses a novel class of 2,2'-biazaaryl ring bidentate ligands and their preparation methods. These ligands, characterized by their unique nitrogen-containing heterocyclic structures, serve as critical components in various catalytic cycles, particularly in pharmaceutical and agrochemical synthesis. The patent introduces a groundbreaking synthetic strategy that bypasses the traditional reliance on pre-functionalized halogenated substrates, instead utilizing a direct cross-coupling approach between non-activated aromatic nitrogen heterocycles and nitrogen-containing heterocyclic arylboronic acids. This technological leap not only expands the structural diversity available to medicinal chemists but also addresses significant supply chain bottlenecks associated with complex ligand manufacturing.
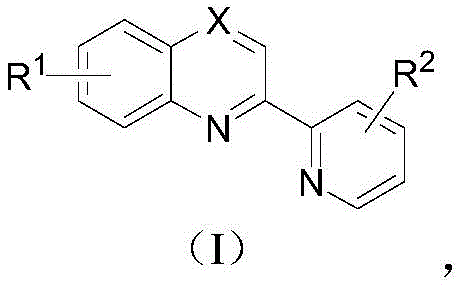
For R&D directors and procurement specialists, the implications of this technology are profound. By enabling the synthesis of these high-value ligands from cheap, commodity-grade starting materials like quinoline and pyridine boronic acids, the patent offers a pathway to drastically reduce the cost of goods sold (COGS) for downstream catalytic processes. Furthermore, the method's compatibility with aqueous conditions and mild temperatures suggests a robust safety profile and easier regulatory compliance, key factors for any reliable pharmaceutical intermediate supplier aiming to support global API production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of nitrogen-containing bidentate ligands, such as bipyridyl-type structures, has relied heavily on classical name reactions like Ullmann, Suzuki, or Stille couplings. While these methods are well-established, they suffer from inherent inefficiencies that hinder modern green chemistry initiatives. The primary bottleneck is the prerequisite for halogenated starting materials. Introducing halogen substituents (chlorine, bromine, or iodine) onto the aromatic rings prior to coupling adds at least one extra synthetic step, increasing both the time and cost of production. Moreover, these halogenation reactions often generate stoichiometric amounts of hazardous waste and require harsh reagents. From a supply chain perspective, the dependency on specific halogenated intermediates creates vulnerability; any disruption in the supply of these specialized precursors can halt the entire ligand production line. Additionally, the use of palladium catalysts in traditional Suzuki couplings, while effective, often leaves trace metal impurities that are difficult to remove to the stringent levels required for pharmaceutical applications, necessitating expensive purification protocols.
The Novel Approach
The methodology presented in CN112778272B represents a paradigm shift by employing a direct C-H activation strategy. Instead of starting with halogenated pyridines or quinolines, the process utilizes non-activated aromatic nitrogen heterocycles directly. This is achieved through an iridium-catalyzed cross-coupling reaction mediated by water, which facilitates a hydrogen evolution mechanism. By eliminating the halogenation step entirely, the synthetic route is significantly shortened, leading to higher overall atom economy. The reaction conditions are remarkably mild, typically operating between 100°C and 130°C, which reduces energy consumption compared to high-temperature pyrolysis methods. Furthermore, the byproducts of this reaction are primarily hydrogen gas and boric acid, both of which are environmentally benign and easy to handle. This "green" profile not only simplifies waste treatment but also aligns perfectly with the increasingly strict environmental regulations facing the fine chemical industry today.
Mechanistic Insights into Ir-Catalyzed C-H Activation Cross-Coupling
At the heart of this innovation lies a sophisticated iridium-catalyzed mechanism that enables the functionalization of inert C-H bonds. The catalyst system, preferably utilizing dichloro(pentamethylcyclopentadienyl)iridium dimer ([Cp*IrCl2]2), operates in concert with a specific amino acid additive, L-proline. The role of L-proline is critical; it acts as a transient directing group or a proton shuttle that facilitates the cleavage of the C-H bond on the non-activated heterocycle. The reaction proceeds through a catalytic cycle where the iridium center coordinates with the nitrogen atom of the heterocycle, positioning the metal close to the target C-H bond. Subsequent oxidative addition or sigma-bond metathesis, mediated by the aqueous environment, leads to the formation of an iridium-carbon bond. This intermediate then undergoes transmetallation with the arylboronic acid species. The presence of water is not merely a solvent choice but a mechanistic necessity, participating in the hydrolysis of the boron species and the subsequent reductive elimination that releases the final 2,2'-biazaaryl product and regenerates the active catalyst. This water-mediated hydrogen evolution is a key feature that distinguishes this process from anhydrous cross-couplings, offering a safer and more sustainable reaction pathway.
From an impurity control perspective, this mechanism offers distinct advantages. Traditional halogen-based couplings often produce homocoupling byproducts (e.g., biaryls formed from two halide molecules) or dehalogenated species. In contrast, the C-H activation pathway described here is highly selective for the 2-position of the quinoline or pyridine ring due to the chelation assistance provided by the ring nitrogen. This regioselectivity minimizes the formation of isomeric impurities, resulting in a cleaner crude reaction mixture. For R&D teams, this means less time spent on optimizing chromatographic separations and higher yields of the desired bidentate ligand. The ability to tune the electronic properties of the ligand by simply varying the R1 and R2 substituents on the starting materials allows for the rapid generation of a library of ligands with tailored steric and electronic profiles, essential for optimizing specific catalytic transformations in drug discovery.
How to Synthesize 2,2'-Biazaaryl Bidentate Ligand Efficiently
The practical execution of this synthesis is designed for operational simplicity, making it accessible for both laboratory-scale optimization and pilot-plant production. The general procedure involves charging a reactor with the non-activated heterocycle and the boronic acid derivative under an inert atmosphere to prevent catalyst oxidation. The choice of solvent is flexible, with mixtures of 1,4-dioxane and water proving particularly effective, balancing the solubility of organic substrates with the mechanistic requirement for water. The addition of the iridium catalyst and L-proline additive initiates the reaction upon heating. Post-reaction workup is straightforward, involving vacuum concentration followed by liquid-liquid extraction, avoiding the need for complex filtration of inorganic salts often seen in base-mediated couplings. For detailed standard operating procedures and specific parameter optimizations, please refer to the guide below.
- Under a protective nitrogen or argon atmosphere, disperse the non-activated aromatic nitrogen heterocycle (e.g., quinoline) and nitrogen-containing heterocyclic arylboronic acid in a mixed solvent of 1,4-dioxane and water.
- Add the iridium catalyst (e.g., [Cp*IrCl2]2) and the amino acid additive (L-proline) to the reaction mixture.
- Heat the mixture to 100-130°C for 12-24 hours, then cool, concentrate, extract with organic solvent, and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route translates into tangible strategic benefits beyond mere technical novelty. The primary advantage lies in the radical simplification of the raw material portfolio. By sourcing commodity chemicals like quinoline and pyridine derivatives instead of specialized halogenated intermediates, companies can leverage existing bulk supply chains, thereby reducing exposure to price volatility and supply disruptions. The elimination of the halogenation step also removes a significant cost center, as halogenating agents and the associated waste disposal fees are substantial expenses in traditional manufacturing. Furthermore, the mild reaction conditions reduce the energy load on manufacturing facilities, contributing to lower utility costs and a smaller carbon footprint, which is increasingly important for meeting corporate sustainability goals.
- Cost Reduction in Manufacturing: The economic impact of this technology is driven by the removal of the pre-functionalization step. In traditional routes, the synthesis of 2-halopyridines or 2-haloquinolines requires additional reagents, reactors, and purification stages, all of which add to the final cost. By bypassing this, the new method effectively compresses the supply chain, leading to substantial cost savings. Additionally, the use of earth-abundant additives like L-proline and the potential for catalyst recycling further enhance the cost-efficiency profile. The reduction in hazardous waste generation also lowers the environmental compliance costs associated with waste treatment and disposal, providing a dual financial and ecological benefit.
- Enhanced Supply Chain Reliability: Reliability is paramount in the pharmaceutical supply chain. The reliance on non-activated starting materials ensures a more robust supply base, as these chemicals are produced in massive quantities for various industries, unlike niche halogenated intermediates which may have limited suppliers. The operational simplicity of the reaction, which tolerates a wide range of substrates and uses common solvents, reduces the risk of batch failures due to sensitive reaction conditions. This robustness ensures consistent delivery schedules and reduces the lead time for high-purity pharmaceutical intermediates, allowing downstream API manufacturers to maintain leaner inventories without risking production stoppages.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces unforeseen challenges, particularly with exothermic reactions or those generating toxic gases. This iridium-catalyzed process generates hydrogen gas and boric acid as byproducts, both of which are manageable at scale with standard engineering controls. The absence of heavy metal waste streams (other than the trace iridium which can be recovered) and halogenated organic waste simplifies the environmental permitting process for new manufacturing lines. The process is inherently safer, operating at moderate temperatures and pressures, which facilitates the commercial scale-up of complex ligand manufacturing from kilogram to multi-ton scales with minimal re-engineering of the core process.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel ligand synthesis technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, aimed at clarifying the feasibility and advantages for potential partners.
Q: What are the advantages of this synthesis method over traditional Suzuki coupling?
A: Unlike traditional methods requiring pre-halogenated substrates, this patent utilizes direct C-H activation on non-activated heterocycles. This eliminates the costly and waste-generating halogenation step, significantly reducing raw material costs and environmental impact.
Q: What catalyst system is used for this cross-coupling reaction?
A: The process employs an Iridium-based catalyst system, specifically dichloro(pentamethylcyclopentadienyl)iridium dimer ([Cp*IrCl2]2), paired with L-proline as a critical additive to facilitate the hydrogen evolution reaction under water mediation.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the patent highlights mild reaction conditions (100-130°C), the use of inexpensive and readily available solvents (toluene, dioxane, water), and simple post-treatment procedures, making it highly amenable to commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,2'-Biazaaryl Ligand Supplier
The development of advanced ligand systems like the 2,2'-biazaaryl derivatives described in CN112778272B underscores the critical need for partners who can bridge the gap between academic innovation and industrial reality. NINGBO INNO PHARMCHEM stands at the forefront of this transition, offering comprehensive CDMO services tailored to the complex requirements of modern catalytic chemistry. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising results observed in the laboratory can be reliably translated into bulk manufacturing. We understand that high-purity pharmaceutical intermediates require stringent purity specifications and rigorous QC labs to verify the absence of trace metals and isomeric impurities, and our facilities are equipped to meet these exacting standards consistently.
We invite R&D directors and procurement leaders to explore how this innovative synthesis route can optimize your supply chain and reduce costs. By leveraging our expertise in iridium-catalyzed transformations and process optimization, we can help you secure a stable supply of these critical ligands. Please contact our technical procurement team to request a Customized Cost-Saving Analysis specific to your project needs. We are ready to provide specific COA data and route feasibility assessments to demonstrate how partnering with us can accelerate your development timelines and enhance your competitive edge in the global market.
