Advanced Green Synthesis of Pregabalin Intermediate via DES and Mechanochemistry for Commercial Scale-up
The pharmaceutical industry is constantly seeking more sustainable and cost-effective pathways for the production of critical active pharmaceutical ingredient (API) precursors. A recent technological breakthrough documented in patent CN110803993B introduces a novel synthetic methodology for producing ethyl 2-carboxyethyl-3-nitromethylene-5-methylhexanoate, a pivotal intermediate in the manufacture of Pregabalin. This patent details a sophisticated two-step process that fundamentally shifts away from traditional, hazardous organic solvents towards the utilization of Deep Eutectic Solvents (DES) and mechanochemical grinding techniques. By integrating green chemistry principles directly into the core reaction design, this approach addresses long-standing challenges regarding toxicity, waste generation, and operational complexity. For R&D directors and process chemists, this represents a significant opportunity to modernize legacy production lines, ensuring compliance with increasingly stringent environmental regulations while maintaining high purity standards required for neurological therapeutics.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of complex nitro-compounds and diester intermediates like those required for Pregabalin has relied heavily on volatile organic solvents such as toluene, dichloromethane, or ethanol, often in conjunction with harsh acidic or basic catalysts. These conventional routes frequently suffer from significant drawbacks, including prolonged reaction times that bottleneck production throughput and necessitate extensive energy input for heating and refluxing. Furthermore, the post-reaction workup in traditional methods is notoriously labor-intensive, requiring multiple washing steps, drying procedures, and energy-heavy distillation processes to remove residual solvents. The environmental footprint of these legacy methods is substantial, generating large volumes of hazardous waste that incur high disposal costs and pose regulatory risks. Additionally, the use of stoichiometric amounts of catalysts that cannot be recovered leads to inflated raw material costs and complicates the purification of the final API intermediate, potentially introducing trace impurities that are difficult to remove.
The Novel Approach
In stark contrast, the methodology outlined in CN110803993B leverages the unique physicochemical properties of Deep Eutectic Solvents (DES), specifically a eutectic mixture of choline chloride and urea, to drive the initial Knoevenagel condensation. This innovative solvent system not only acts as a reaction medium but also functions as a catalyst, thereby streamlining the reagent profile. The subsequent step employs a mechanochemical grinding technique for the nitro-alkylation, effectively eliminating the need for bulk solvents in the second stage of the synthesis. 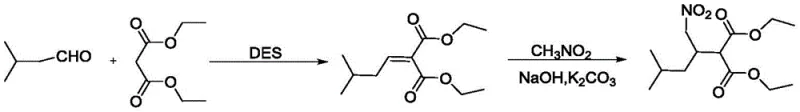 As illustrated in the reaction pathway, this dual-strategy approach results in a dramatic simplification of the operational workflow. The DES can be recovered from the aqueous phase after extraction and reused multiple times with minimal loss in efficiency, creating a closed-loop system that drastically reduces raw material consumption. The grinding method ensures rapid mixing and energy transfer at the molecular level, leading to faster reaction kinetics and superior yields compared to solution-phase counterparts, ultimately delivering a robust and scalable process for high-purity intermediate manufacturing.
As illustrated in the reaction pathway, this dual-strategy approach results in a dramatic simplification of the operational workflow. The DES can be recovered from the aqueous phase after extraction and reused multiple times with minimal loss in efficiency, creating a closed-loop system that drastically reduces raw material consumption. The grinding method ensures rapid mixing and energy transfer at the molecular level, leading to faster reaction kinetics and superior yields compared to solution-phase counterparts, ultimately delivering a robust and scalable process for high-purity intermediate manufacturing.
Mechanistic Insights into DES-Catalyzed Condensation and Mechanochemical Nitro-Alkylation
The first stage of this synthesis involves a Knoevenagel condensation between isovaleraldehyde and diethyl malonate, facilitated by the hydrogen-bonding network of the Choline Chloride-Urea DES. The urea component of the DES likely activates the carbonyl group of the aldehyde through hydrogen bonding, increasing its electrophilicity, while the basic nature of the mixture assists in the deprotonation of the active methylene group in diethyl malonate. This synergistic activation lowers the activation energy of the reaction, allowing it to proceed efficiently at a moderate temperature of 80°C. The structured solvent environment stabilizes the transition state and the resulting enolate intermediate, promoting the formation of the alpha,beta-unsaturated diester with high stereoselectivity and yield. Crucially, the polar nature of the DES facilitates the separation of the organic product upon the addition of water, as the product partitions into the organic extraction layer while the hydrophilic DES remains in the aqueous phase, ready for regeneration.
The second mechanistic phase utilizes solid-state grinding to effect the addition of nitromethane to the unsaturated intermediate. In this mechanochemical environment, the physical force of grinding brings the solid reactants (the diester intermediate, sodium hydroxide, and potassium carbonate) into intimate contact with liquid nitromethane. The bases serve to generate the nitronate anion from nitromethane directly on the solid surface, which then undergoes a Michael-type addition to the electron-deficient double bond of the diester. The absence of solvent cages allows for higher effective concentrations of reactants, driving the equilibrium towards product formation. The use of a mixed base system (NaOH and K2CO3) likely optimizes the basicity and buffering capacity within the solid matrix, preventing side reactions such as polymerization or over-nitration. This solvent-free approach not only accelerates the reaction rate but also simplifies the impurity profile, as there are no solvent-derived byproducts to contend with during the final purification stages.
How to Synthesize Ethyl 2-carboxyethyl-3-nitromethylene-5-methylhexanoate Efficiently
To implement this advanced synthetic route in a laboratory or pilot plant setting, operators must strictly adhere to the optimized molar ratios and thermal profiles defined in the patent to ensure maximum efficiency and reproducibility. The process begins with the in-situ preparation of the DES catalyst, followed by the condensation reaction, and concludes with the mechanochemical finishing step. Detailed standard operating procedures regarding specific mixing speeds, grinding durations, and extraction protocols are critical for maintaining the integrity of the recyclable solvent system. Adhering to these standardized steps ensures that the theoretical benefits of the green chemistry approach are fully realized in practical application, yielding a product that meets the rigorous quality specifications demanded by downstream API synthesis.
- Prepare Deep Eutectic Solvent (DES) by mixing choline chloride and urea at 80°C, then react isovaleraldehyde with diethyl malonate in this medium.
- Isolate the intermediate ethyl 2-carboxyethyl-5-methyl-2-hexanoate via extraction and recover the DES for reuse.
- Perform a solvent-free grinding reaction of the intermediate with nitromethane, NaOH, and K2CO3 to obtain the final nitro-compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this DES-based and mechanochemical synthesis route offers profound strategic advantages that extend beyond mere technical feasibility. The primary value driver is the substantial reduction in operational expenditure (OPEX) associated with solvent procurement and waste management. By replacing expensive, regulated volatile organic compounds with commodity chemicals like choline chloride and urea, the direct material costs are significantly lowered. Furthermore, the ability to recycle the DES solvent multiple times without degradation creates a circular economy within the manufacturing process, decoupling production volume from raw material consumption and insulating the supply chain from market volatility in solvent pricing.
- Cost Reduction in Manufacturing: The elimination of bulk organic solvents in the second reaction step removes the need for large-scale solvent recovery units and the associated energy costs for distillation. Additionally, the high yields reported (consistently above 95%) minimize the loss of valuable starting materials, directly improving the overall mass balance and cost-per-kilogram of the final intermediate. The use of simple grinding equipment instead of complex high-pressure reactors also reduces capital expenditure (CAPEX) and maintenance overheads, making the technology accessible for facilities with varying levels of infrastructure investment.
- Enhanced Supply Chain Reliability: The raw materials required for this process, including isovaleraldehyde, diethyl malonate, and nitromethane, are widely available commodity chemicals with stable global supply chains. The robustness of the DES system, which tolerates minor variations in moisture and temperature better than sensitive organometallic catalysts, ensures consistent batch-to-batch quality. This reliability reduces the risk of production delays caused by reagent instability or strict storage requirements, allowing for more flexible inventory management and just-in-time manufacturing strategies.
- Scalability and Environmental Compliance: From a regulatory standpoint, this process aligns perfectly with modern green chemistry mandates, significantly reducing the facility's carbon footprint and hazardous waste output. The non-toxic nature of the DES solvent simplifies safety protocols and reduces the burden on environmental health and safety (EHS) departments. Scalability is inherently supported by the simplicity of the unit operations; the condensation can be scaled in standard stirred tanks, and the grinding step can be adapted to industrial milling equipment, facilitating a smooth transition from kilogram-scale development to multi-ton commercial production without the need for process re-engineering.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthetic pathway. These insights are derived directly from the experimental data and beneficial effects described in the patent literature, providing a clear understanding of the process capabilities. Understanding these nuances is essential for stakeholders evaluating the integration of this technology into their existing manufacturing portfolios.
Q: What are the primary advantages of using Deep Eutectic Solvents (DES) in this synthesis?
A: DES offers a non-toxic, biodegradable alternative to volatile organic compounds (VOCs), significantly reducing environmental impact and waste disposal costs while allowing for solvent recycling up to five times without significant yield loss.
Q: How does the mechanochemical grinding method improve the second reaction step?
A: The grinding method eliminates the need for bulk organic solvents in the nitro-alkylation step, drastically simplifying post-reaction workup, reducing energy consumption for solvent removal, and achieving high yields (up to 98%) in shorter reaction times.
Q: Is this synthetic route suitable for large-scale industrial production?
A: Yes, the process utilizes inexpensive, commercially available raw materials like choline chloride and urea, operates under mild conditions (80°C), and avoids complex high-pressure equipment, making it highly scalable and cost-effective for tonnage production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ethyl 2-carboxyethyl-3-nitromethylene-5-methylhexanoate Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of green synthetic methodologies like the one described in CN110803993B for the production of high-value pharmaceutical intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale innovation to industrial reality is seamless and efficient. Our state-of-the-art facilities are equipped to handle both DES-based reactions and mechanochemical processes, supported by rigorous QC labs that guarantee stringent purity specifications for every batch of Pregabalin intermediate we supply. We are committed to delivering not just a chemical product, but a comprehensive supply solution that enhances your operational efficiency.
We invite forward-thinking pharmaceutical companies to collaborate with us to leverage this cost-effective and environmentally friendly synthesis route. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out today to obtain specific COA data and route feasibility assessments, ensuring that your supply chain for neurological therapeutics is built on a foundation of innovation, reliability, and sustainable growth.
