Advanced Solvent-Free Synthesis of Antioxidant 1790 Intermediate for High-Performance Polymer Additives
The global demand for high-performance hindered phenol antioxidants continues to surge, driven by the expanding applications of polyurethane, polyolefin, and polystyrene in automotive and construction sectors. Within this critical supply chain, the synthesis of key intermediates such as 2,4-dimethyl-6-tert-butyl-3-chloromethyl phenol (CAS 23500-79-0) represents a pivotal technological bottleneck that directly impacts the quality and cost of the final antioxidant products. Patent CN108046999B introduces a groundbreaking solvent-free methodology that fundamentally redefines the production landscape for this essential chemical building block. By transitioning away from traditional halogenated solvent systems towards a green, surfactant-catalyzed approach, this innovation addresses the growing regulatory pressures regarding volatile organic compounds (VOCs) while simultaneously enhancing process economics. For R&D directors and procurement strategists alike, understanding the nuances of this patent is crucial for securing a reliable antioxidant intermediate supplier capable of delivering both environmental compliance and commercial viability in an increasingly stringent market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of antioxidant 1790 intermediates has relied heavily on chloromethylation reactions conducted in halogenated hydrocarbon solvents, a practice that is becoming increasingly untenable in modern chemical manufacturing. Traditional protocols, such as those described in earlier patents like CN101684067, necessitate the use of toxic solvents like chloroform or carbon tetrachloride, which pose severe health risks to operators and require complex, energy-intensive recovery systems to meet environmental standards. Furthermore, these legacy processes often operate at elevated temperatures, typically requiring heating to around 40°C or higher, which increases the thermal load on production facilities and elevates the risk of side reactions that can compromise product purity. Another significant drawback of conventional methods is the high loading of catalysts required to drive the reaction to completion; prior art often utilizes quaternary ammonium salts at concentrations ranging from 6% to 8% of the substrate mass, which not only inflates raw material costs but also complicates the downstream purification process due to the difficulty of removing residual surfactant species from the final product matrix.
The Novel Approach
In stark contrast to these legacy constraints, the novel solvent-free method disclosed in the patent data offers a paradigm shift by eliminating the solvent entirely and leveraging a sophisticated dual-surfactant catalytic system. This innovative approach allows the chloromethylation of 2,4-dimethyl-6-tert-butylphenol with paraformaldehyde to proceed efficiently at room temperature, thereby removing the need for external heating and significantly reducing the facility's energy footprint. The core of this technological advancement lies in the use of a composite catalyst comprising both ionic and nonionic surfactants, which creates a highly efficient micro-environment for the reaction to occur without the bulk medium of a solvent. This modification not only simplifies the reactor setup and operation but also drastically reduces the catalyst dosage to a mere 0.2% to 1% of the substrate weight, representing a massive reduction in chemical consumption. By integrating continuous HCl gas introduction with this mild, solvent-free regime, the process achieves conversion rates exceeding 99%, demonstrating that high efficiency and environmental stewardship are not mutually exclusive goals in fine chemical synthesis.
Mechanistic Insights into Surfactant-Catalyzed Chloromethylation
The mechanistic superiority of this solvent-free process stems from the synergistic interaction between the ionic and nonionic surfactant components, which effectively mimics a phase-transfer catalysis environment without the need for a biphasic solvent system. In the absence of a bulk organic solvent, the reactants—specifically the lipophilic phenol derivative and the hydrophilic formaldehyde source—are brought into intimate contact through the micellar structures formed by the surfactant mixture. The ionic surfactant, such as sodium dodecyl sulfate or tetraalkylammonium bromides, provides charged interfaces that facilitate the activation of the electrophilic species generated from paraformaldehyde and HCl gas. Simultaneously, the nonionic surfactant, selected from the Tween series (e.g., Tween 20, 60, or 80), enhances the solubility of the organic substrate within the reaction medium and stabilizes the transition states. This dual-action mechanism ensures that the chloromethylation occurs selectively at the ortho-position relative to the hydroxyl group, minimizing the formation of poly-chlorinated byproducts or ether linkages that often plague less controlled reactions. The result is a clean reaction profile where the primary impurity is unreacted starting material, which is easily managed by driving the conversion to near-completion levels.
From an impurity control perspective, the mild reaction conditions play a critical role in maintaining the integrity of the sensitive phenolic structure. Traditional high-temperature processes can induce thermal degradation or rearrangement of the tert-butyl groups, leading to complex impurity profiles that are difficult to separate and can negatively impact the color stability of the final antioxidant. By conducting the reaction at room temperature, the novel method preserves the structural fidelity of the 2,4-dimethyl-6-tert-butyl moiety, ensuring that the resulting intermediate possesses the high purity required for downstream synthesis of Antioxidant 1790. The ability to monitor reaction progress via gas chromatography (GC) until the starting phenol content drops below 1% provides a robust quality control checkpoint, guaranteeing consistent batch-to-batch reproducibility. This level of control is essential for manufacturers supplying the high-purity polymer additives market, where even trace impurities can affect the long-term aging performance of plastics and rubbers.
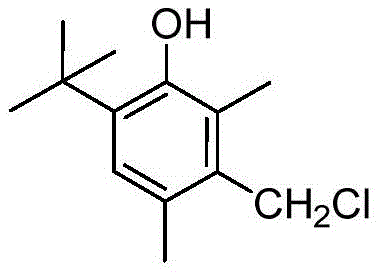
How to Synthesize 2,4-Dimethyl-6-Tert-Butyl-3-Chloromethyl Phenol Efficiently
The practical implementation of this solvent-free technology requires precise control over reagent ratios and gas flow rates to maximize the benefits of the surfactant catalysis. The process begins with the careful charging of 2,4-dimethyl-6-tert-butylphenol, paraformaldehyde, and concentrated hydrochloric acid into a stirred reaction vessel, followed by the addition of the optimized surfactant mixture. The molar ratio of phenol to paraformaldehyde is maintained between 1:1.1 and 1:1.3 to ensure complete consumption of the phenol while minimizing excess formaldehyde waste. Once the mixture is homogenized, dry HCl gas is continuously introduced at room temperature, serving both as a reactant and a driving force for the chloromethylation equilibrium. Detailed standardized synthesis steps for replicating this high-efficiency protocol are provided in the technical guide below, ensuring that production teams can safely and effectively adopt this greener methodology.
- Charge 2,4-dimethyl-6-tert-butylphenol, paraformaldehyde, concentrated hydrochloric acid, and a composite catalyst system consisting of ionic and nonionic surfactants into a reaction vessel.
- Initiate stirring and continuously introduce dry HCl gas into the system at room temperature until the starting phenol content drops below 1%.
- Allow the mixture to stand for phase separation, remove the aqueous layer for recycling, and perform reduced pressure dehydration on the organic layer to isolate the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this solvent-free synthesis route translates into tangible strategic advantages that extend far beyond simple unit price considerations. The elimination of halogenated solvents removes a major category of hazardous material from the supply chain, thereby reducing the regulatory burden associated with storage, transport, and disposal of toxic chemicals. This simplification of the material handling profile significantly de-risks the manufacturing operation, ensuring greater continuity of supply even in the face of tightening environmental regulations on volatile organic compounds. Furthermore, the drastic reduction in catalyst usage and the ability to recycle the aqueous acidic phase create a leaner production model that is inherently more resilient to fluctuations in raw material pricing. These factors combine to offer a compelling value proposition for companies seeking cost reduction in polymer additive manufacturing without compromising on the quality or reliability of their intermediate supply.
- Cost Reduction in Manufacturing: The economic impact of switching to this solvent-free process is profound, primarily driven by the complete removal of solvent purchase and recovery costs. In traditional methods, the expense of buying halogenated solvents and the energy required to distill and recover them for reuse constitute a significant portion of the operating budget; eliminating this step results in immediate and substantial savings. Additionally, the reduction in catalyst loading from the typical 6-8% down to less than 1% represents a direct decrease in raw material expenditure, compounding the financial benefits over large production volumes. The mild room temperature conditions also mean that less energy is consumed for heating and cooling, further lowering the utility costs associated with the manufacturing process and contributing to a lower overall cost of goods sold.
- Enhanced Supply Chain Reliability: Supply chain stability is greatly improved by the simplified logistics of a solvent-free operation, as there is no longer a dependency on the availability of specialized halogenated solvents which can be subject to strict quotas and supply disruptions. The use of commercially available and stable surfactants ensures that the critical catalytic components are easily sourced from multiple vendors, reducing the risk of single-source bottlenecks. Moreover, the robustness of the reaction, which tolerates slight variations in conditions while maintaining high conversion, ensures consistent output rates that allow for more accurate forecasting and inventory planning. This reliability is crucial for maintaining the production schedules of downstream antioxidant manufacturers who depend on a steady flow of high-quality intermediates.
- Scalability and Environmental Compliance: Scaling up this process is inherently safer and more straightforward because the absence of flammable or toxic solvents reduces the hazard classification of the production facility. This facilitates easier permitting and expansion of capacity to meet growing market demand for antioxidant 1790 without the need for expensive upgrades to solvent containment systems. From an environmental standpoint, the process generates significantly less hazardous waste, aligning with corporate sustainability goals and reducing the costs associated with waste treatment and disposal. The ability to recycle the aqueous phase back into the process further minimizes the environmental footprint, making this technology an ideal choice for companies aiming to enhance their green chemistry credentials while scaling up complex polymer additives.
Frequently Asked Questions (FAQ)
To assist technical teams in evaluating the feasibility of this technology for their specific applications, we have compiled answers to common inquiries regarding the operational parameters and quality outcomes of this solvent-free method. These insights are derived directly from the experimental data and technical disclosures within the patent literature, providing a factual basis for decision-making. Understanding these details is essential for assessing how this process integrates into existing manufacturing workflows and quality assurance frameworks.
Q: What are the primary advantages of the solvent-free method over traditional halogenated solvent processes?
A: The solvent-free method eliminates the need for toxic halogenated hydrocarbons like chloroform or carbon tetrachloride, drastically reducing environmental hazards and operator health risks. Furthermore, it removes the energy-intensive steps associated with solvent recovery and distillation, leading to substantial operational cost reductions and a simplified post-treatment workflow.
Q: How does the dual surfactant catalyst system improve reaction efficiency?
A: By combining ionic surfactants (such as sodium dodecyl sulfate) with nonionic surfactants (like Tween series), the process achieves a synergistic catalytic effect that allows for significantly lower catalyst loading (0.2-1%) compared to traditional single-surfactant methods (6-8%). This results in higher conversion rates exceeding 99% under mild room temperature conditions.
Q: Is the aqueous byproduct reusable in this synthesis protocol?
A: Yes, the process design includes a closed-loop capability where the separated aqueous phase can replace fresh concentrated hydrochloric acid in subsequent batches. This recycling mechanism not only minimizes liquid waste discharge but also further optimizes the consumption of raw materials, enhancing the overall sustainability profile of the manufacturing process.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Antioxidant 1790 Intermediate Supplier
As the chemical industry pivots towards more sustainable and efficient manufacturing paradigms, the ability to execute complex solvent-free syntheses at scale becomes a defining competitive advantage. NINGBO INNO PHARMCHEM stands at the forefront of this transition, leveraging our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production to bring innovations like the CN108046999B process to the global market. Our commitment to excellence is underpinned by stringent purity specifications and rigorous QC labs that ensure every batch of 2,4-dimethyl-6-tert-butyl-3-chloromethyl phenol meets the exacting standards required for high-performance polymer stabilization. We understand that consistency is key in the additives sector, and our advanced process control systems are designed to deliver the high conversion rates and low impurity profiles demonstrated in the patent data.
We invite forward-thinking organizations to collaborate with us to unlock the full potential of this green chemistry technology for their supply chains. By partnering with our technical procurement team, you can obtain a Customized Cost-Saving Analysis that quantifies the specific economic benefits of switching to our solvent-free intermediate for your operations. We encourage you to reach out today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our advanced manufacturing capabilities can drive both value and sustainability in your antioxidant production strategy.
