Advanced Manufacturing of 3-Cyclopentamine: A Technical Breakthrough for Commercial Scale-Up
Introduction to Next-Generation 3-Cyclopentamine Manufacturing
The pharmaceutical and fine chemical industries are constantly seeking robust synthetic routes that balance efficiency with safety, particularly for complex amine intermediates like 3-cyclopentamine. A pivotal advancement in this domain is detailed in patent CN102030658B, which discloses a novel methodology for preparing 3-cyclopentamine and its corresponding salts. This technology represents a significant departure from legacy synthesis pathways, addressing critical bottlenecks related to raw material scarcity and process safety. By leveraging a strategic cyclization of readily available chlorinated butenes with nitromethane, the process circumvents the need for hazardous azide chemistry. This shift not only enhances the safety profile of the manufacturing facility but also stabilizes the supply chain against the volatility of specialty reagent markets. Furthermore, the ability to directly synthesize stable salt forms adds value for downstream formulation processes.

The core innovation lies in the simplification of the molecular construction, transforming a multi-step, high-risk sequence into a streamlined operation. For procurement managers and supply chain directors, this translates to a reliable pharmaceutical intermediates supplier capability that is less susceptible to regulatory shutdowns or raw material shortages. The patent outlines a versatile approach where the final amine can be isolated or immediately converted into various pharmaceutically acceptable salts, such as hydrochlorides or sulfates, depending on the specific requirements of the active pharmaceutical ingredient (API) synthesis. This flexibility is crucial for contract development and manufacturing organizations (CDMOs) aiming to offer comprehensive solutions to their clients.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 3-cyclopentamine has been plagued by significant technical and economic hurdles that hindered its widespread adoption in cost-sensitive applications. The traditional pathway typically initiates with cyclopenten-1-ol, a starting material that is not only expensive but often difficult to source in the large quantities required for industrial scale-up. Following the initial protection steps, the critical transformation involves a diazotization reaction utilizing sodium azide. This specific step introduces severe safety liabilities, as organic azides are notoriously unstable and potentially explosive, necessitating specialized equipment and rigorous containment protocols. Moreover, the overall synthetic route is elongated, involving multiple isolation and purification stages that cumulatively erode the final yield. These inefficiencies result in a high cost of goods sold (COGS) and a substantial environmental footprint due to the generation of hazardous nitrogenous waste streams.
The Novel Approach
In stark contrast, the methodology described in the patent data introduces a paradigm shift by utilizing cis-1,4-dichloro-2-butene and nitromethane as the foundational building blocks. These precursors are commodity chemicals with established global supply chains, effectively eliminating the risk of raw material bottlenecks. The new process condenses the synthetic timeline by merging ring construction and functional group installation into a single, base-catalyzed cyclization event. This is followed by a straightforward reduction step to reveal the primary amine functionality. By completely avoiding the use of sodium azide and diazotization, the process removes the most dangerous element of the legacy route. This transition facilitates cost reduction in fine chemical manufacturing by lowering insurance premiums, reducing waste treatment costs, and simplifying the operational complexity required for safe production.
Mechanistic Insights into Base-Catalyzed Nitro-Cyclization
The heart of this technological breakthrough is the efficient construction of the cyclopentene ring system through a nucleophilic substitution and cyclization cascade. The reaction initiates with the deprotonation of nitromethane by a strong alkaline catalyst, such as lithium hydride, sodium hydride, or potassium tert-butoxide, generating a reactive nitronate anion. This nucleophile attacks the electrophilic carbon centers of the cis-1,4-dichloro-2-butene, displacing chloride ions in a concerted or stepwise manner that closes the five-membered ring. The choice of solvent plays a critical role in stabilizing the transition states; polar aprotic solvents like tetrahydrofuran (THF), dimethylformamide (DMF), or non-polar options like toluene are employed to optimize solubility and reaction kinetics. The temperature is carefully controlled between 25°C and 80°C to balance reaction rate with selectivity, ensuring the formation of the desired 4-nitro-cyclopentyl-1-alkenyl intermediate while minimizing polymerization or side reactions.
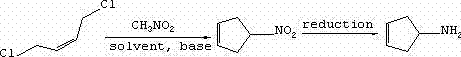
Following the cyclization, the nitro group serves as a masked amine precursor, which is unveiled through a classic metal-acid reduction system. The patent specifies the use of active metals like iron, zinc, or tin in the presence of acids such as hydrochloric acid, acetic acid, or ammonium salts. This reduction phase is highly chemoselective, converting the nitro functionality to the primary amine without affecting the internal double bond of the cyclopentene ring, which is vital for preserving the structural integrity of the target molecule. The mechanism likely proceeds through nitroso and hydroxylamine intermediates before reaching the final amine state. This robustness in impurity control ensures that the resulting 3-cyclopentamine meets high-purity specifications required for pharmaceutical applications, minimizing the burden on downstream purification units.
How to Synthesize 3-Cyclopentamine Efficiently
Implementing this synthesis requires precise control over stoichiometry and reaction conditions to maximize the conversion of the dichloro-precursor. The process begins with the preparation of the nitronate species in a dry, inert atmosphere to prevent moisture interference with the strong base catalyst. Once the cyclization is complete, the workup involves simple filtration to remove inorganic salts, followed by concentration to isolate the nitro-intermediate. The subsequent reduction step is exothermic and requires careful thermal management, typically achieved by batch-wise addition of the metal powder to the reaction mixture. Detailed standardized synthetic steps see the guide below.
- Perform a cyclization reaction between cis-1,4-dichloro-2-butene and nitromethane in a solvent like THF or toluene using a strong base catalyst at 25-80°C to form 4-nitro-cyclopentyl-1-alkenyl.
- Reduce the resulting nitro-intermediate using a metal-acid reduction system, such as iron/hydrochloric acid or zinc/ammonium chloride, to yield crude 3-cyclopentamine.
- Purify the amine and optionally react with an acid like hydrochloric acid to form the stable 3-cyclopentamine salt for storage and transport.
Commercial Advantages for Procurement and Supply Chain Teams
For stakeholders responsible for the bottom line and operational continuity, the adoption of this patent-covered route offers compelling strategic benefits. The shift from rare, protected alcohols to bulk chlorinated olefins fundamentally alters the cost structure of the molecule. By removing the dependency on sodium azide, facilities can operate with lower safety overheads and reduced regulatory scrutiny regarding explosive precursors. This translates directly into a more competitive pricing model for the final intermediate. Furthermore, the simplified workflow reduces the number of unit operations, which decreases labor hours and energy consumption per kilogram of product. These factors combine to create a resilient supply chain capable of meeting fluctuating market demands without the fragility associated with complex, multi-step legacy syntheses.
- Cost Reduction in Manufacturing: The elimination of expensive starting materials like cyclopenten-1-ol and the removal of protection-deprotection sequences drastically lower the direct material costs. Additionally, the avoidance of hazardous azide chemistry reduces the capital expenditure required for specialized blast-proof reactors and extensive waste neutralization systems. The use of commodity reagents ensures that price volatility is minimized, allowing for stable long-term budgeting and significant overall cost savings in the production of high-purity 3-cyclopentamine.
- Enhanced Supply Chain Reliability: Sourcing cis-1,4-dichloro-2-butene and nitromethane is far more secure than relying on niche fine chemical suppliers for azides or protected alcohols. These feedstocks are produced at massive scales for other industries, guaranteeing availability even during global supply disruptions. This reliability reduces lead time for high-purity pharmaceutical intermediates, enabling faster turnaround times for client projects and ensuring consistent inventory levels for continuous manufacturing campaigns.
- Scalability and Environmental Compliance: The reaction conditions are mild and utilize common solvents that are easily recovered and recycled, aligning with green chemistry principles. The absence of heavy metal catalysts in the cyclization step and the use of iron or zinc in the reduction step simplify the effluent treatment process. This environmental compatibility facilitates easier permitting for commercial scale-up of complex amine intermediates, allowing manufacturers to expand capacity rapidly to meet growing market needs without encountering regulatory bottlenecks.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. Understanding these details is essential for R&D teams evaluating process feasibility and procurement officers assessing vendor capabilities. The answers are derived directly from the technical specifications and advantageous effects reported in the patent literature, providing a factual basis for decision-making.
Q: Why is the new nitro-cyclization route preferred over traditional azide methods?
A: The traditional route relies on sodium azide diazotization, which involves highly toxic and explosive reagents requiring stringent safety protocols. The new method uses commodity chemicals like nitromethane and dichlorobutene, significantly improving operational safety and reducing hazardous waste disposal costs.
Q: What are the key raw materials for this synthesis?
A: The process utilizes cis-1,4-dichloro-2-butene and nitromethane as starting materials. These are widely available industrial commodities, unlike the rare and expensive cyclopenten-1-alcohol required in older methods, ensuring a robust and continuous supply chain.
Q: Is this process suitable for large-scale manufacturing?
A: Yes, the reaction conditions are mild (25-80°C) and utilize standard solvents like THF or ethanol. The elimination of dangerous diazotization steps and the use of simple filtration for catalyst removal make it highly adaptable for multi-ton commercial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Cyclopentamine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting innovative synthetic routes to maintain competitiveness in the global pharmaceutical market. Our technical team has thoroughly analyzed the potential of the nitro-cyclization pathway described in CN102030658B and is prepared to deploy this technology for your specific project needs. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of 3-cyclopentamine meets the exacting standards required for drug substance manufacturing.
We invite you to collaborate with us to leverage these process efficiencies for your supply chain. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your volume requirements. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments, allowing you to validate the quality and economic viability of this advanced manufacturing approach for your upcoming campaigns.
