Optimizing Tiaprofenic Acid Production: A Technical Breakthrough for Commercial Scale-up
The pharmaceutical industry continuously seeks robust synthetic pathways for nonsteroidal anti-inflammatory drugs (NSAIDs) that balance efficacy with manufacturing feasibility. Patent CN102432591B introduces a transformative synthesis process for 5-benzoyl-alpha-methyl-2-thiopheneacetic acid, commonly known as Tiaprofenic acid. This specific chemical entity represents a critical pharmaceutical intermediate with significant therapeutic potential for treating arthritis and post-operative pain. The disclosed methodology fundamentally shifts the production paradigm by utilizing thiophene as a starting material, undergoing a sophisticated six-step sequence involving substitution, bromination, acid catalysis, rearrangement, and Friedel-Crafts acylation. Unlike legacy methods that struggle with stability and yield, this novel approach ensures stable reaction conditions and high product quality. For global procurement teams, understanding the technical nuances of this patent is essential for securing a reliable pharmaceutical intermediates supplier capable of delivering consistent quality. The process eliminates the reliance on deteriorating catalysts and complex purification steps that have historically plagued the supply chain of this high-purity NSAID intermediate.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historical synthesis routes for Tiaprofenic acid have been fraught with significant technical and economic inefficiencies that hinder large-scale adoption. As illustrated in prior art methodologies, early attempts often relied on manganic catalysts which are prone to deterioration upon exposure to air, leading to inconsistent catalytic performance and incomplete reactions. Other conventional pathways utilized expensive raw materials like acetyl thiophene or chloroformyl ethyl formate, driving up the cost reduction in API manufacturing significantly. Furthermore, traditional oxidative steps frequently resulted in very low productive rates, often below 10%, rendering them economically unviable for commercial operations. The use of harsh conditions, such as strict explosion-proof requirements for methyl iodide reactions, introduced severe safety risks and environmental pollution concerns. These legacy processes also struggled with purification, often failing to meet pharmacopeia requirements due to difficult-to-remove byproducts. Consequently, the industry has long suffered from supply discontinuity and inflated costs associated with these inefficient synthetic strategies.
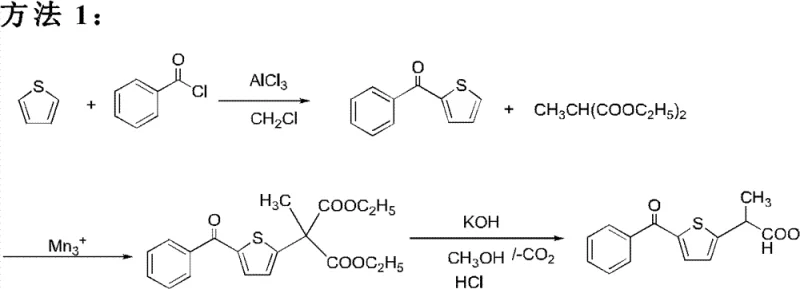
The Novel Approach
In stark contrast to the deficiencies of prior art, the novel approach detailed in the patent data leverages a streamlined and chemically elegant pathway starting from readily available thiophene. By employing Vanadium Pentoxide in FLAKES as a protonic acid agent during the initial propionylation, the process achieves optimal yields approaching 99% under controlled temperatures. The strategic introduction of a ketalization step prior to acylation serves as a protective measure, directing the subsequent Friedel-Crafts reaction to the desired position on the thiophene ring. This structural manipulation effectively bypasses the formation of unwanted regio-isomers that typically contaminate the final product in older methods. Moreover, the rearrangement step utilizes stable zinc or copper salts, eliminating the need for hazardous or unstable catalysts. The overall sequence is designed with industrial scalability in mind, utilizing standard solvents like dichloromethane and toluene that are easily recovered and recycled. This modernization of the synthetic route directly translates to enhanced supply chain reliability and substantial cost savings for downstream manufacturers.
Mechanistic Insights into Catalytic Rearrangement and Acylation
The core innovation of this synthesis lies in the precise control of the catalytic rearrangement and the subsequent electrophilic aromatic substitution. Following the initial bromination of propionyl thiophene, the formation of the ketal intermediate is critical for masking the ketone functionality, thereby preventing side reactions during the rearrangement phase. Under the influence of acidic catalysts such as tosic acid or sulfuric acid, the ketal undergoes a structural reorganization to form the ester intermediate. This step is pivotal as it establishes the alpha-methyl substitution pattern required for the biological activity of the final NSAID. The rearrangement is facilitated by specific metal salts like cuprous oxide or zinc chloride, which promote the migration of the ester group with high transformation efficiency. This mechanistic precision ensures that the carbon skeleton is constructed correctly before the bulky benzoyl group is introduced. Such attention to mechanistic detail is what distinguishes a laboratory curiosity from a viable commercial scale-up of complex pharmaceutical intermediates.
Impurity control is rigorously managed through the sequencing of the acylation and hydrolysis steps. In the Friedel-Crafts acylation stage, the use of Lewis acids like aluminum chloride is optimized to minimize over-acylation or polymerization of the thiophene ring. The patent specifies a careful workup procedure involving basic washes to remove unreacted benzoyl chloride and acidic byproducts, which is crucial for meeting stringent purity specifications. The final hydrolysis and acidification steps are conducted under mild conditions to prevent racemization or degradation of the sensitive carboxylic acid moiety. By avoiding the harsh oxidative conditions found in Method 2 or Method 5 of the prior art, this process significantly reduces the generation of tarry byproducts. This results in a cleaner crude product that requires less intensive recrystallization, thereby improving the overall mass balance and reducing solvent waste. For R&D directors, this level of impurity profile management is essential for ensuring regulatory compliance and batch-to-batch consistency.
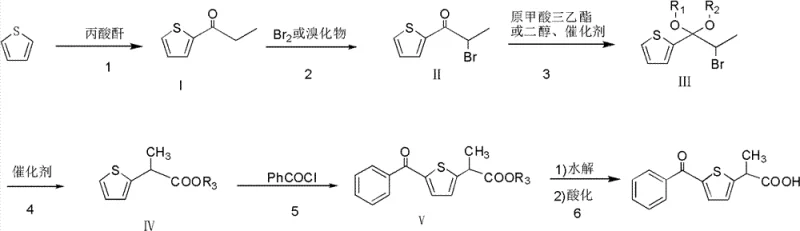
How to Synthesize 5-benzoyl-alpha-methyl-2-thiopheneacetic acid Efficiently
The execution of this synthesis requires strict adherence to the defined reaction parameters to maximize yield and safety. The process begins with the propionylation of thiophene, where temperature control between 70°C and 150°C is vital to prevent polymerization while ensuring complete conversion. Subsequent bromination must be performed under anhydrous conditions to maintain the activity of the Lewis acid catalyst. The ketalization and rearrangement steps demand precise stoichiometric ratios of glycol or orthoformate to drive the equilibrium towards the desired ester intermediate. Finally, the acylation and hydrolysis steps require careful pH monitoring to ensure complete conversion to the free acid without degrading the thiophene ring. Detailed standardized synthetic steps see the guide below.
- Propionylation of thiophene using propionic anhydride and Vanadium Pentoxide in FLAKES catalyst.
- Bromination of the propionyl thiophene intermediate using bromine or bromo-derivatives.
- Ketalization and subsequent catalytic rearrangement to form the ester intermediate.
- Friedel-Crafts acylation with Benzoyl chloride followed by hydrolysis and acidification.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented process offers compelling advantages that directly address the pain points of procurement managers and supply chain heads. The shift from expensive, specialized starting materials to commodity chemicals like thiophene and propionic anhydride drastically simplifies the raw material sourcing strategy. This fundamental change in the bill of materials reduces exposure to volatile pricing markets and ensures a more stable supply base. Furthermore, the elimination of unstable catalysts means that production batches are less likely to fail due to reagent degradation, enhancing overall operational reliability. The process safety improvements, such as avoiding explosion-prone reagents, lower insurance and facility maintenance costs, contributing to significant cost savings in the long term. These factors combine to create a more resilient supply chain capable of withstanding market fluctuations.
- Cost Reduction in Manufacturing: The adoption of this novel route eliminates the need for costly oxidants and precious metal catalysts that characterized previous methods. By utilizing abundant and inexpensive reagents, the direct material cost per kilogram of the active intermediate is substantially lowered. Additionally, the high yield in the initial propionylation step minimizes raw material waste, further optimizing the cost structure. The simplified purification process reduces the consumption of solvents and energy required for distillation and recrystallization. These cumulative efficiencies allow for a more competitive pricing model without compromising on the quality of the high-purity NSAID intermediate supplied to clients.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions ensures that production schedules can be maintained with minimal interruption. Unlike prior art methods that were sensitive to moisture or air, this process tolerates standard industrial operating environments, reducing the risk of batch failures. The use of common solvents and reagents means that supply disruptions for specific niche chemicals are unlikely to halt production. This reliability is critical for reducing lead time for high-purity pharmaceutical intermediates, allowing downstream drug manufacturers to plan their inventory more effectively. Consistent availability of this key building block supports continuous manufacturing campaigns and prevents costly downtime in the final API production lines.
- Scalability and Environmental Compliance: The process is inherently designed for suitability for industrialized production, with reaction steps that translate seamlessly from pilot plant to multi-ton reactors. The avoidance of heavy metal oxidants and toxic byproducts simplifies wastewater treatment and waste disposal protocols. This alignment with green chemistry principles facilitates easier regulatory approval and reduces the environmental footprint of the manufacturing site. The ability to scale up complex organic compounds safely ensures that supply can be ramped up quickly to meet surges in market demand. This scalability provides a strategic advantage for partners looking to secure long-term volume commitments for their global drug portfolios.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of Tiaprofenic acid intermediates. These insights are derived directly from the technical specifications and advantageous effects outlined in the patent literature. Understanding these details helps stakeholders evaluate the feasibility of integrating this supply source into their existing value chains. The answers reflect a commitment to transparency regarding process capabilities and quality standards.
Q: What are the primary advantages of the novel synthesis route for Tiaprofenic Acid?
A: The novel route described in patent CN102432591B utilizes cheap and easily obtainable raw materials like thiophene and propionic anhydride. It avoids the use of unstable catalysts found in prior art, resulting in significantly higher total yields and improved process safety suitable for industrial production.
Q: How does this process address impurity control in NSAID manufacturing?
A: The specific sequence of ketalization and rearrangement prior to acylation effectively minimizes the formation of regio-isomers such as 2-(2-benzoyl-4-thienyl) propionic acid. This ensures a cleaner impurity profile and reduces the burden on downstream purification steps.
Q: Is this synthesis method scalable for commercial supply chains?
A: Yes, the process is designed for suitability for industrialized production. It employs stable reaction conditions and standard unit operations like extraction and distillation, facilitating reliable commercial scale-up of complex pharmaceutical intermediates without requiring exotic equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-benzoyl-alpha-methyl-2-thiopheneacetic acid Supplier
At NINGBO INNO PHARMCHEM, we possess the technical expertise to translate complex patent methodologies into commercial reality. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this novel synthesis are fully realized in practice. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 5-benzoyl-alpha-methyl-2-thiopheneacetic acid meets the highest international standards. Our commitment to quality assurance means that R&D directors can rely on our materials for critical clinical and commercial applications without fear of impurity-related setbacks. We bridge the gap between innovative chemistry and reliable supply.
We invite global partners to collaborate with us to optimize their supply chains for NSAID manufacturing. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our implementation of this advanced synthesis can benefit your organization. Let us be your trusted partner in delivering high-quality chemical solutions that drive your business forward.
