Revolutionizing Pregabalin Intermediate Production via Deep Eutectic Solvent Catalysis
Revolutionizing Pregabalin Intermediate Production via Deep Eutectic Solvent Catalysis
The pharmaceutical industry is currently witnessing a paradigm shift towards greener, more sustainable manufacturing processes, particularly in the synthesis of high-value active pharmaceutical ingredient (API) intermediates. A groundbreaking development in this sector is detailed in patent CN110803994B, which outlines a novel, environmentally benign synthetic route for producing 3-nitromethylene-5-methyl-hexanoic acid ethyl ester, a critical precursor for the anticonvulsant drug Pregabalin. This technology replaces traditional, hazardous organic solvents with Deep Eutectic Solvents (DES), offering a compelling solution for manufacturers seeking to optimize their supply chains while adhering to stringent environmental regulations. By leveraging the dual functionality of DES as both solvent and catalyst, this method achieves remarkable reaction efficiencies and simplifies downstream processing, positioning it as a superior alternative for the commercial scale-up of complex pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Pregabalin intermediates has been plagued by significant operational and environmental challenges that hinder efficient large-scale production. Traditional routes typically rely heavily on volatile organic compounds (VOCs) such as dichloromethane, toluene, or tetrahydrofuran, which pose severe health risks to operators and require complex, energy-intensive abatement systems to manage emissions. Furthermore, these conventional methods often necessitate the use of stoichiometric amounts of harsh acids or bases, leading to the generation of substantial quantities of saline wastewater that is costly to treat and dispose of. The cumulative effect of these factors is a manufacturing process characterized by low atom economy, extended reaction times, and fluctuating yields that struggle to meet the rigorous purity specifications demanded by global regulatory bodies. Consequently, procurement teams face inflated costs driven by solvent recovery expenses and waste management fees, while supply chain managers grapple with the instability of processes that are difficult to control and scale reliably.
The Novel Approach
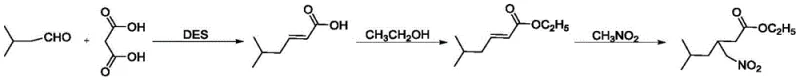
In stark contrast to these legacy methods, the technology disclosed in patent CN110803994B introduces a streamlined, three-step cascade that utilizes bio-compatible Deep Eutectic Solvents to drive the reaction forward with exceptional precision. The process initiates with a Knoevenagel condensation between isovaleraldehyde and malonic acid, followed by an esterification with ethanol, and concludes with a Michael addition involving nitromethane. What distinguishes this approach is the strategic selection of eutectic mixtures, specifically Choline Chloride-Urea and Choline Chloride-Methanesulfonic Acid, which act simultaneously as the reaction medium and the catalytic promoter. This dual role eliminates the need for additional catalysts and toxic solvents, drastically reducing the chemical footprint of the operation. The result is a robust synthetic pathway that operates under mild thermal conditions, typically around 80°C, ensuring high selectivity and minimizing the formation of unwanted by-products that complicate purification.
Mechanistic Insights into DES-Catalyzed Cascade Synthesis
The efficacy of this novel synthetic route lies in the unique physicochemical properties of the Deep Eutectic Solvents employed, which facilitate proton transfer and stabilize transition states more effectively than conventional media. In the initial step, the hydrogen bond network within the Choline Chloride-Urea mixture activates the carbonyl group of the isovaleraldehyde, promoting nucleophilic attack by the active methylene group of malonic acid. This activation lowers the energy barrier for the dehydration step, driving the equilibrium towards the formation of 5-methyl-2-hexenoic acid with high conversion rates. Subsequently, the switch to a Choline Chloride-Methanesulfonic Acid system provides the necessary acidic environment for the Fischer esterification, where the methanesulfonic acid component protonates the carboxylic acid, enhancing its electrophilicity towards the ethanol nucleophile. This precise tuning of the solvent acidity ensures that the esterification proceeds rapidly without requiring excessive temperatures that could degrade the sensitive olefinic bond.
Furthermore, the final Michael addition step benefits immensely from the basicity inherent in the Choline Chloride-Urea system, which facilitates the deprotonation of nitromethane to generate the reactive nitronate anion. This anion then attacks the beta-carbon of the alpha,beta-unsaturated ester in a highly regioselective manner, yielding the target 3-nitromethylene-5-methyl-hexanoic acid ethyl ester. Crucially, the structured solvent cage provided by the DES helps to suppress side reactions such as polymerization or over-alkylation, which are common pitfalls in nitro-alkene chemistry. From an impurity control perspective, the high polarity of the DES allows for easy separation of the organic product via simple extraction with ethyl acetate, leaving the catalyst and polar impurities in the aqueous phase. This inherent separability ensures that the final product meets stringent purity specifications with minimal downstream processing, a critical factor for R&D directors focused on impurity profiles.
How to Synthesize 3-Nitromethylene-5-Methyl-Hexanoic Acid Ethyl Ester Efficiently
Implementing this green synthesis protocol requires careful attention to the preparation and recycling of the eutectic solvents to maximize economic and environmental benefits. The process is divided into three distinct stages, each optimized for specific reaction parameters including temperature, molar ratios, and workup procedures. Operators must ensure that the DES components are thoroughly mixed and heated to form a homogeneous liquid prior to the introduction of substrates, as the integrity of the hydrogen bond network is essential for catalytic activity. Following the reaction, the product is isolated through liquid-liquid extraction, while the remaining aqueous phase containing the DES is subjected to vacuum evaporation to remove water, allowing the catalyst to be recycled for subsequent batches. For a comprehensive guide on the specific molar ratios, temperature profiles, and safety precautions required for each step, please refer to the standardized operating procedure detailed below.
- Perform Knoevenagel condensation between isovaleraldehyde and malonic acid using Choline Chloride-Urea (DES1) at 80°C to obtain 5-methyl-2-hexenoic acid.
- Conduct esterification of 5-methyl-2-hexenoic acid with absolute ethanol using Choline Chloride-Methanesulfonic Acid (DES2) at 80°C to yield the ethyl ester.
- Execute Michael addition with nitromethane using Choline Chloride-Urea (DES3) at 80°C to finalize the synthesis of 3-nitromethylene-5-methyl-hexanoic acid ethyl ester.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this DES-based technology represents a strategic opportunity to significantly reduce operational expenditures while enhancing supply security. The elimination of volatile organic solvents not only lowers the direct cost of raw materials but also removes the substantial overhead associated with solvent recovery distillation columns and hazardous waste incineration. Moreover, the ability to recycle the Deep Eutectic Solvents multiple times without significant loss of catalytic performance translates into a drastic reduction in the consumption of auxiliary chemicals, thereby stabilizing the variable costs of production. This efficiency gain is compounded by the simplified post-treatment process, which reduces the man-hours required for purification and quality control testing, allowing facilities to increase throughput without expanding their physical footprint.
- Cost Reduction in Manufacturing: The transition to a solvent-free or low-solvent regime fundamentally alters the cost structure of API intermediate manufacturing by removing the dependency on petrochemical-derived solvents. Since the DES components, such as choline chloride and urea, are inexpensive, abundant, and non-toxic bulk chemicals, the overall material cost is substantially lower compared to traditional routes that rely on specialized reagents. Additionally, the high yields reported in the patent data minimize the loss of valuable starting materials like isovaleraldehyde and nitromethane, ensuring that every kilogram of input generates maximum output value. This improved atom economy directly contributes to a more competitive pricing model for the final intermediate, providing a clear margin advantage in a cost-sensitive market.
- Enhanced Supply Chain Reliability: Relying on commodity chemicals for the catalytic system mitigates the risk of supply disruptions that often plague the procurement of specialized catalysts or exotic solvents. Choline chloride and urea are produced on a massive global scale for various industries, ensuring a stable and continuous supply line that is resilient to market fluctuations. Furthermore, the robustness of the reaction conditions means that the process is less susceptible to minor variations in raw material quality or environmental factors, leading to more consistent batch-to-batch performance. This reliability is crucial for maintaining uninterrupted production schedules and meeting the just-in-time delivery requirements of downstream pharmaceutical customers.
- Scalability and Environmental Compliance: As regulatory pressure mounts regarding industrial emissions and waste disposal, this green synthesis method offers a future-proof solution that aligns with global sustainability goals. The absence of toxic VOCs simplifies the permitting process for new manufacturing lines and reduces the liability associated with environmental compliance. The scalability of the process is further enhanced by the ease of heat management in the DES system, which allows for safe operation in large-scale reactors without the risk of thermal runaway. This combination of safety, scalability, and environmental stewardship makes the technology an ideal candidate for long-term commercial partnerships and capacity expansion projects.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthetic route. These insights are derived directly from the experimental data and beneficial effects described in the patent literature, providing a factual basis for decision-making. Understanding these details is essential for stakeholders evaluating the feasibility of integrating this technology into their existing manufacturing portfolios.
Q: What are the primary environmental advantages of this synthesis method?
A: The method utilizes Deep Eutectic Solvents (DES) composed of choline chloride and urea or methanesulfonic acid, eliminating the need for toxic volatile organic solvents. Furthermore, the DES catalysts can be recovered from the aqueous phase and reused multiple times without significant loss of activity.
Q: What yields can be expected from this DES-catalyzed route?
A: Experimental data indicates exceptionally high efficiency, with the initial Knoevenagel condensation step achieving yields up to 98%, and subsequent esterification and Michael addition steps consistently maintaining yields above 95% under optimized conditions.
Q: Is the catalyst system scalable for industrial production?
A: Yes, the process is designed for scalability. The reusability studies demonstrate that the DES catalyst maintains high catalytic activity over at least five consecutive cycles, ensuring consistent product quality and reducing raw material consumption for large-scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Nitromethylene-5-Methyl-Hexanoic Acid Ethyl Ester Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of green chemistry technologies like the one described in patent CN110803994B to redefine the standards of pharmaceutical intermediate production. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent supply of high-quality materials. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of 3-nitromethylene-5-methyl-hexanoic acid ethyl ester meets the exacting requirements of global regulatory agencies. We are committed to leveraging our technical expertise to bring this efficient, eco-friendly synthesis method to the forefront of the supply chain.
We invite forward-thinking pharmaceutical companies and chemical distributors to collaborate with us to explore the full commercial potential of this innovative process. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements and logistical needs. We encourage you to reach out today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that will drive efficiency and profitability in your API manufacturing operations.
